Therapeutic Class Overview Incretin Mimetics
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Pharmacokinetics of Omarigliptin, a Once-Weekly Dipeptidyl Peptidase-4 Inhibitor
Available online a t www.derpharmachemica.com ISSN 0975-413X Der Pharma Chemica, 2016, 8(12):292-295 CODEN (USA): PCHHAX (http://derpharmachemica.com/archive.html) Mini-review: Pharmacokinetics of Omarigliptin, a Once-weekly Dipeptidyl Peptidase-4 Inhibitor Nermeen Ashoush a,b aClinical Pharmacy and Pharmacy Practice Department, Faculty of Pharmacy, British University in Egypt, El- Sherouk city, Cairo 11837, Egypt. bHead of Health Economics Unit, Center for Drug Research and Development (CDRD), Faculty of Pharmacy, British University in Egypt, El-Sherouk city, Cairo 11837, Egypt. _____________________________________________________________________________________________ ABSTRACT The dipeptidyl peptidase-4 (DPP-4) inhibitors are novel oral hypoglycemic drugs which have been in clinical use for the past 10 years. The drugs are safe, weight neutral and widely prescribed. There are currently many gliptins approved by FDA, namely sitagliptin, vildagliptin, saxagliptin, linagliptin, alogliptin with several more in advanced stages of development. The gliptins may possess cardiovascular protective effects and their administration may promote β-cell survival; claims currently being evaluated in clinical and preclinical studies. The gliptins are an optional second-line therapy after metformin; they are generally well tolerated with low risk of hypoglycemia. The various compounds differ with respect to their pharmacokinetic properties; however, their clinical efficacy appears to be similar. The clinical differences between the various compounds -

An in Vivo Investigation Into the Actions of the Hypothalamic Neuropeptide, QRFP
An In Vivo Investigation into the Actions of the Hypothalamic Neuropeptide, QRFP A thesis submitted to the University of Manchester for the degree of Doctor of Philosophy in the Faculty of Biology, Medicine and Health Christopher J Cook Faculty of Biology, Medicine and Health School of Medical Sciences 2017 Contents Abstract ........................................................................................................................................... 11 Declaration ........................................................................................................................................ 12 Copyright ........................................................................................................................................... 12 Acknowledgement ............................................................................................................................ 13 Chapter 1 Introduction ................................................................................................. 14 1.1 Energy homeostasis ................................................................................................................ 15 1.2 The control of food intake ...................................................................................................... 16 1.2.1 Peripheral signals regulating food intake .......................................................................... 17 1.2.2 Central aspects of food intake regulation ........................................................................ -

Safety and Efficacy of Omarigliptin (MK-3102), a Novel Once-Weekly
2106 Diabetes Care Volume 38, November 2015 fi Wayne H.-H. Sheu,1 Ira Gantz,2 Safety and Ef cacy of Omarigliptin Menghui Chen,2 Shailaja Suryawanshi,2 Arpana Mirza,2 Barry J. Goldstein,2 (MK-3102), a Novel Once-Weekly Keith D. Kaufman,2 and Samuel S. Engel2 DPP-4 Inhibitor for the Treatment of Patients With Type 2 Diabetes Diabetes Care 2015;38:2106–2114 | DOI: 10.2337/dc15-0109 OBJECTIVE This study was conducted to determine the optimal dose of omarigliptin, a once- weekly (q.w.) dipeptidyl peptidase IV (DPP-4) inhibitor, for the treatment of patients with type 2 diabetes and evaluate the long-term safety of that dose. RESEARCH DESIGN AND METHODS In a multicenter, double-blind, 12-week, dose-range finding study, 685 oral antihy- perglycemic agent-na¨ıve or washed-out subjects with type 2 diabetes were random- ized to one of five once-weekly doses of omarigliptin (0.25 mg, 1 mg, 3 mg, 10 mg, or 25 mg) or placebo. The primary efficacy end point was change from baseline in HbA1c, and secondary end points were 2-h postmeal glucose (PMG) and fasting plasma glucose (FPG). Analysis included all patients who received at least one dose of the study medication. Subjects who completed the base study were eligible to enter a 66-week extension study. RESULTS Once-weekly treatment for 12 weeks with omarigliptin provided dose-related reduc- 1Division of Endocrinology and Metabolism, De- partment of Internal Medicine, Taichung Veterans EMERGING TECHNOLOGIES AND THERAPEUTICS tions in HbA , 2-h PMG, and FPG. -
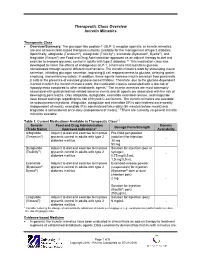
Therapeutic Class Overview Incretin Mimetics
Therapeutic Class Overview Incretin Mimetics Therapeutic Class Overview/Summary: The glucagon-like peptide-1 (GLP-1) receptor agonists, or incretin mimetics, are one of two incretin-based therapies currently available for the management of type 2 diabetes. Specifically, albiglutide (Tanzeum®), dulaglutide (Trulicity®), exenatide (Bydureon®, Byetta®), and liraglutide (Victoza®) are Food and Drug Administration-approved as an adjunct therapy to diet and exercise to improve glycemic control in adults with type 2 diabetes.1-5 This medication class was developed to mimic the effects of endogenous GLP-1, a hormone that maintains glucose homeostasis through several different mechanisms. The incretin mimetics work by stimulating insulin secretion, inhibiting glucagon secretion, improving β cell responsiveness to glucose, delaying gastric emptying, and enhancing satiety. In addition, these agents increase insulin secretion from pancreatic β cells in the presence of elevated glucose concentrations. Therefore, due to the glucose-dependent manner in which the incretin mimetics work, the medication class is associated with a low risk of hypoglycemia compared to other antidiabetic agents.6 The incretin mimetics are most commonly associated with gastrointestinal-related adverse events and all agents are associated with the risk of developing pancreatitis. Only albiglutide, dulaglutide, exenatide extended-release, and liraglutide have boxed warnings regarding the risk of thyroid C-cell tumors. The incretin mimetics are available as subcutaneous injections. Albiglutide, dulaglutide and exenatide ER is administered once-weekly (independent of meals), exenatide IR is administered twice-daily (60 minutes before meals) and liraglutide is administered once-daily (independent of meals).1-5 There are currently no generic incretin mimetics available. Table 1. -

Pleiotropic Mechanisms for the Glucose-Lowering Action of DPP-4 Inhibitors
2196 Diabetes Volume 63, July 2014 Bilal Omar and Bo Ahrén Pleiotropic Mechanisms for the Glucose-Lowering Action of DPP-4 Inhibitors Diabetes 2014;63:2196–2202 | DOI: 10.2337/db14-0052 Dipeptidyl peptidase (DPP)-4 inhibition is a glucose- developed on the basis that the gut-derived glucagon-like lowering treatment for type 2 diabetes. The classical peptide (GLP)-1 is a potent antidiabetic hormone due to mechanism for DPP-4 inhibitors is that they inhibit DPP-4 its ability to stimulate insulin secretion and inhibit gluca- activity in peripheral plasma, which prevents the in- gon secretion. DPP-4 inhibition prevents the inactivation activation of the incretin hormone glucagon-like peptide of GLP-1 and, therefore, raises the circulating intact (ac- (GLP)-1 in the peripheral circulation. This in turn in- tive) GLP-1 levels above physiological levels that have creases circulating intact GLP-1, which results in stimu- antidiabetic actions. lated insulin secretion and inhibited glucagon secretion, After initial preclinical development, the first clinical in turn increasing glucose utilization and diminishing proof-of-concept study for DPP-4 inhibition was reported hepatic glucose production, which, through reduction in in the early 2000s (2). DPP-4 inhibition was first ap- postprandial and fasting glucose, reduces HbA .How- 1c proved for clinical use in 2006 with the DPP-4 inhibitor ever, recent experimental studies in mainly rodents but sitagliptin, and thereafter, several other DPP-4 inhibitors also to a limited degree in humans have found additional have been introduced into clinical practice (3). They are all mechanisms for DPP-4 inhibitors that may contribute to their glucose-lowering action. -

Incretin Medications
New Medications for Diabetes BARRETT CHAPIN, MD, FACP, FACE, CDE Diabetes medications - Timeline TIME 1922 1955 1995 1996 1997 New Diabetes medications - Overview Incretin medications GLP-1 Agonists - 2005 DPP-4 Inhibitors - 2006 SGLT-2 Inhibitors - 2013 Insulins Short-acting / Inhailed (Afrezza) - 2014 Long-acting U-300 Glargine (Toujeo) – 2015 U-200 Humalog – 2015 Degludec (Tresiba) and 70/30 Degludec/Aspart (Ryzodeg) 9/2015 New Diabetes medications - Incretins Incretin medications GLP-1 Agonists DPP-4 Inhibitors Incretin - Physiology and Action In response to food: Gastric Inhibitory polypeptide (GIP) Secreted into the Blood stream. Glucagon-like peptide-1 (GLP-1) Incretin Physiology and Action of GLP-1 and GIP Function • Stimulates Insulin release from Glucagon-like the pancreas (only when the peptide-1 (GLP-1) glucose is elevated) both • Inhibits Glucagon release from Gastric Inhibitory the pancreas (which stops the polypeptide (GIP) liver from making glucose, when the glucose is elevated) Incretin Physiology and Action of GLP-1 and GIP Dipepitdyl Pepidase-4 Enzyme Glucagon-like (DPP-4) peptide-1 (GLP-1) Breaks down GLP-1 and GIP. Inactive Degradation Half life of GLP-1 and GIP is just Products Gastric Inhibitory a few minutes polypeptide (GIP) Incretin medication - Categories GLP-1 DPP-4 Inhibitors Agonists Dipeptidyl Pepidase-4 Enzyme Glucagon-like (DPP-4) peptide-1 (GLP-1) Breaks down GLP-1 and GIP. Inactive Degradation Half life of GLP-1 and GIP is just Products Gastric Inhibitory a few minutes polypeptide (GIP) DPP-4 Inhibitors -

Incretin Hormones in Obesity and Related Cardiometabolic Disorders: the Clinical Perspective
nutrients Review Incretin Hormones in Obesity and Related Cardiometabolic Disorders: The Clinical Perspective Joanna Michałowska * , Ewa Miller-Kasprzak and Paweł Bogda ´nski Department of Obesity and Metabolic Disorders Treatment and Clinical Dietetics, Pozna´nUniversity of Medical Sciences, Szamarzewskiego St. 82/84, 60-569 Pozna´n,Poland; [email protected] (E.M.-K.); [email protected] (P.B.) * Correspondence: [email protected] Abstract: The prevalence of obesity continues to grow rapidly worldwide, posing many public health challenges of the 21st century. Obese subjects are at major risk for serious diet-related noncommunicable diseases, including type 2 diabetes mellitus, cardiovascular disease, and non- alcoholic fatty liver disease. Understanding the mechanisms underlying obesity pathogenesis is needed for the development of effective treatment strategies. Dysregulation of incretin secretion and actions has been observed in obesity and related metabolic disorders; therefore, incretin-based therapies have been developed to provide new therapeutic options. Incretin mimetics present glucose-lowering properties, together with a reduction of appetite and food intake, resulting in weight loss. In this review, we describe the physiology of two known incretins—glucose-dependent insulinotropic polypeptide (GIP) and glucagon-like peptide-1 (GLP-1), and their role in obesity and related cardiometabolic disorders. We also focus on the available and incoming incretin-based medications that can be used in the treatment of the above-mentioned conditions. Keywords: obesity; GLP-1; GIP; incretins; incretin-based therapy; type 2 diabetes mellitus; non- alcoholic fatty liver disease; cardiovascular disease Citation: Michałowska, J.; Miller-Kasprzak, E.; Bogda´nski,P. Incretin Hormones in Obesity and Related Cardiometabolic Disorders: 1. -

GIP: an Inconsequential Incretin Or Not?
Editorials EDITORIAL (SEE SOLOMON ET AL., P. 1561) GIP: An Inconsequential Incretin or Not? he “incretin effect” refers to the en- control subjects (4). Rather, it appears tion. A total of 29 older obese men and hancement of insulin secretion in that -cell insulin secretory responses to women with either NGT (n ϭ 16) or new- T response to an oral glucose load rel- incretin hormones are impaired in pa- ly-diagnosed type 2 diabetes (n ϭ 13) ative to that of an isoglycemic intravenous tients with type 2 diabetes. With GLP-1 participated in a 3-month weight loss and glucose challenge (1). In humans, the in- this defect is modest and can be overcome exercise intervention. At baseline and af- cretin effect is mediated by two peptide with infusions that achieve higher GLP-1 ter the intervention, oral glucose toler- hormones, glucagon-like peptide 1 levels. In contrast, even infusions that ance tests were performed to measure (GLP-1) and glucose-dependent insulino- achieve supraphysiological GIP concen- nutrient-induced insulin secretion and tropic polypeptide (GIP), which are se- trations fail to elicit a significant insulin GIP responses and hyperinsulinemic- creted from enteroendocrine cells in secretory response in patients with type 2 euglycemic glucose clamps were per- response to nutrients entering the gut. diabetes (5). Thus, GLP-1 infusions formed to measure insulin action. They GLP-1 secretion from L-cells, found in quickly normalize blood glucose levels in observed that the lifestyle intervention, highest density in the distal ileum but also patients with type 2 diabetes, whereas which decreased body weight by around throughout the small and large intestine, GIP infusions do not (6). -

The Effect of Insulin Dysregulation and Breed on HPA Axis Function and Plasma Cortisol Binding Dynamics in Ponies and Horses
The Effect of Insulin Dysregulation and Breed on HPA Axis Function and Plasma Cortisol Binding Dynamics in Ponies and Horses Hart KA, Vaughn S, Norton NA Department of Large Animal Internal Medicine, University of Georgia College of Veterinary Medicine, Athens, GA Mechanisms resulting in breed predispositions to insulin dysregulation (ID) and endocrinopathic laminitis are poorly characterized. The adrenal steroid cortisol antagonizes insulin, and free, biologically active cortisol can be increased in ID. Breed-related differences in serum free cortisol fraction (FCF) could contribute to ID in pre-disposed breeds such as ponies, but FCF has not been quantified in ID- predisposed-breeds. The objective of this study was to compare FCF between horses and ponies during health and ID. We hypothesized: 1) in health, FCF is higher in ponies than horses; and 2) FCF is further increased in ponies with ID. Serum total cortisol (TC), ACTH, FCF and insulin were measured in 36 horses (age 1-24 years) and 31 mixed-breed ponies (age 4-27 years). Animals were sampled before morning feeding in their normal routine, and ID defined as fasted insulin > 20 μIU/ml or non-fasted insulin > 60 μIU/ml for animals sampled on pasture. Data were compared with Mann-Whitney tests and Spearman correlation analysis (P<0.05). TC and FCF were comparable in healthy horses and ponies, but ACTH and insulin concentrations were 1.3-1.6-fold higher in ponies (P=0.001-0.041). In animals with ID, TC was similar but FCF and insulin were increased 1.6-fold and 3.2-fold respectively in ponies (n=9) compared to horses (n=11, P=0.01-0.049), and FCF and insulin were positively correlated (P=0.04, r=0.45, 95%C.I=0.014- 0.746). -

Disruption of PC1 3 Expression in Mice Causes Dwarfism and Multiple Neuroendocrine Peptide Processing Defects
Disruption of PC1͞3 expression in mice causes dwarfism and multiple neuroendocrine peptide processing defects Xiaorong Zhu*, An Zhou†, Arunangsu Dey*, Christina Norrbom*‡, Raymond Carroll§, Chunling Zhang§, Virginie Laurent¶, Iris Lindberg¶, Randi Ugleholdt‡, Jens J. Holst‡, and Donald F. Steiner*§ʈ *Department of Biochemistry and Molecular Biology, University of Chicago, and §The Howard Hughes Medical Institute, 5841 South Maryland Avenue, Chicago, IL 60637; †Legacy Research and Technology Center, Neurobiology, 1225 NE Second Avenue, Portland, OR 97232; ¶Department of Biochemistry, Louisiana State University Medical Center, 1901 Perdido Street, New Orleans, LA 70112; and ‡Department of Medical Physiology, The Panum Institute, University of Copenhagen, Blegdamsvej 3, DK-2200 Copenhagen N, Denmark Contributed by Donald F. Steiner, June 12, 2002 The subtilisin-like proprotein convertases PC1͞3 (SPC3) and PC2 (SPC2) structurally similar and often functionally overlap, each has are believed to be the major endoproteolytic processing enzymes of definite substrate site preferences. Combined with differences in the regulated secretory pathway. They are expressed together or their expression pattern in NE tissues, these convertases may separately in neuroendocrine cells throughout the brain and dis- process the same precursor protein to release different products persed endocrine system in both vertebrates and invertebrates. having divergent or even opposing functions. An excellent Disruption of the gene-encoding mouse PC1͞3 has now been accom- example is proglucagon, which is processed by PC2 in the islet plished and results in a syndrome of severe postnatal growth impair- alpha cells to release mainly glucagon, whereas in the intestinal ment and multiple defects in processing many hormone precursors, L cells, it is believed to be processed by PC1͞3 to release including hypothalamic growth hormone-releasing hormone (GHRH), glucagon-like peptide (GLP)-1 and GLP-2 (3, 4). -

Pharmacology, Physiology, and Mechanisms of Incretin Hormone Action
Cell Metabolism Review Pharmacology, Physiology, and Mechanisms of Incretin Hormone Action Jonathan E. Campbell1 and Daniel J. Drucker1,* 1Department of Medicine, Samuel Lunenfeld Research Institute, Mount Sinai Hospital, University of Toronto, Toronto, ON M5G 1X5, Canada *Correspondence: [email protected] http://dx.doi.org/10.1016/j.cmet.2013.04.008 Incretin peptides, principally GLP-1 and GIP, regulate islet hormone secretion, glucose concentrations, lipid metabolism, gut motility, appetite and body weight, and immune function, providing a scientific basis for utilizing incretin-based therapies in the treatment of type 2 diabetes. Activation of GLP-1 and GIP receptors also leads to nonglycemic effects in multiple tissues, through direct actions on tissues expressing incretin receptors and indirect mechanisms mediated through neuronal and endocrine pathways. Here we contrast the pharmacology and physiology of incretin hormones and review recent advances in mechanisms coupling incretin receptor signaling to pleiotropic metabolic actions in preclinical studies. We discuss whether mechanisms identified in preclinical studies have potential translational relevance for the treatment of human disease and highlight controversies and uncertainties in incretin biology that require resolution in future studies. Introduction to Incretin Biology tase 1/3 (PC1/3). Original concepts of L cells as predominantly Incretins are gut hormones that potentiate insulin secretion after unihormonal or bihormonal have evolved to reflect evidence meal ingestion in a glucose-dependent manner. The two best- that enteroendocrine L cells exhibit a molecular profile overlap- studied incretins, glucose-dependent insulinotropic polypeptide ping with other gut endocrine cell types and coexpress multiple (GIP) and glucagon-like peptide-1 (GLP-1), exert their insulino- peptide hormones, with diversity of peptide hormone coexpres- tropic actions through distinct G-protein-coupled receptors sion changing along the length of the gastrointestinal tract (Habib highly expressed on islet b cells. -

Gut-Derived Incretin Hormones and New Therapeutic Approaches
Reviews/Commentaries/ADA Statements PERSPECTIVES ON THE NEWS Gut-Derived Incretin Hormones and New Therapeutic Approaches ZACHARY T. BLOOMGARDEN, MD showing in somatostatin-, insulin-, and glucagon-infused dogs (thus not allowing GLP-1 to change insulin or glucagon lev- els) that peripherally administering the his is the first of a series of articles on from nil to 100%. Conversely, when glu- peptide increased hepatic glucose uptake presentations at the American Dia- cose was infused peripherally versus in 60%, while administration either via the Ϫ Ϫ T betes Association Annual Meeting, the portal vein at 2.5 mg ⅐ kg 1 ⅐ min 1, portal vein or hepatic artery let to a 150% Orlando, Florida, 4–8 June 2004, ad- arterial glucose levels were identical but increase in hepatic glucose uptake, sug- dressing an important theme of the meet- there was a rapid drop from hepatic glu- gesting the effect to be in the liver rather ing: new therapeutic approaches based on cose output to uptake with portal deliv- than specifically requiring GLP-1 levels to gut-derived incretin hormones. ery, whereas a much slower fall was seen increase in the portal venous system. Io- in hepatic glucose output occurring with nut et al. (abstract 1412) similarly showed peripheral delivery, with consequent lack a glucose-lowering effect of portal vein Physiology of glucagon-like peptide of increased peripheral glucose uptake GLP-1 infusion. 1 action following intraportal administration. Pe- Cherrington concluded that the por- Alan Cherrington (Nashville, TN) de- ripheral glucose administration led to tal vein and liver may contain nutrient- scribed a series of studies of peripheral most glucose uptake being peripheral.