[Product Name] 500 Mg + 2 Mg Tablet Paracetamol + Thiocolchicoside
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

WHO Pharmaceuticals Newsletters
2021 WHO Pharmaceuticals No.1 NEWSLETTER The WHO Pharmaceuticals Newsletter provides you with the latest information on the safety of medicines WHO Vision for Medicines Safety and legal actions taken by regulatory authorities around No country left behind: the world. It also provides signals based on information worldwide pharmacovigilance for safer medicines, safer patients derived from the WHO global database of individual case safety reports, VigiBase. In addition, this edition of the Newsletter includes a The aim of the Newsletter is to disseminate regulatory summary of discussions and key recommendations of information on the safety of Advisory Committee on Safety of Medicinal Products pharmaceutical products, (ACSoMP) Seventeenth meeting. based on communications received from our network of national pharmacovigilance centres and other sources such as specialized bulletins and journals, as well as partners in WHO. The information is produced in the form of résumés in English, full texts of which may be obtained on request from: Safety and Vigilance: Medicines, EMP-HIS, World Health Organization, 1211 Geneva 27, Switzerland, Contents E -mail address: [email protected] This Newsletter is also available at: Regulatory matters http://www.who.int/medicines Safety of medicines Signal Feature WHO Pharmaceuticals Newsletter No. 1, 2021 ISBN 978-92-4-002136-5 (electronic version) ISBN 978-92-4-002137-2 (print version) © World Health Organization 2021 Some rights reserved. This work is available under the Creative Commons Attribution-NonCommercial-ShareAlike 3.0 IGO licence (CC BY-NC-SA 3.0 IGO; https://creativecommons.org/licenses/by-nc-sa/3.0/igo). Under the terms of this licence, you may copy, redistribute and adapt the work for non-commercial purposes, provided the work is appropriately cited, as indicated below. -
PMBJP Product.Pdf
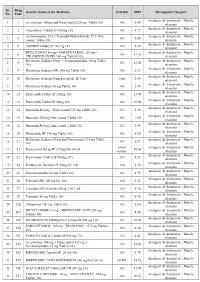
Sr. Drug Generic Name of the Medicine Unit Size MRP Therapeutic Category No. Code Analgesic & Antipyretic / Muscle 1 1 Aceclofenac 100mg and Paracetamol 325 mg Tablet 10's 10's 8.00 relaxants Analgesic & Antipyretic / Muscle 2 2 Aceclofenac Tablets IP 100mg 10's 10's 4.37 relaxants Acetaminophen 325 + Tramadol Hydrochloride 37.5 film Analgesic & Antipyretic / Muscle 3 4 10's 8.00 coated Tablet 10's relaxants Analgesic & Antipyretic / Muscle 4 5 ASPIRIN Tablets IP 150 mg 14's 14's 2.70 relaxants DICLOFENAC 50 mg+ PARACETAMOL 325 mg+ Analgesic & Antipyretic / Muscle 5 6 10's 11.30 CHLORZOXAZONE 500 mg Tablets 10's relaxants Diclofenac Sodium 50mg + Serratiopeptidase 10mg Tablet Analgesic & Antipyretic / Muscle 6 8 10's 12.00 10's relaxants Analgesic & Antipyretic / Muscle 7 9 Diclofenac Sodium (SR) 100 mg Tablet 10's 10's 6.12 relaxants Analgesic & Antipyretic / Muscle 8 10 Diclofenac Sodium 25mg per ml Inj. IP 3 ml 3 ml 2.00 relaxants Analgesic & Antipyretic / Muscle 9 11 Diclofenac Sodium 50 mg Tablet 10's 10's 2.90 relaxants Analgesic & Antipyretic / Muscle 10 12 Etoricoxilb Tablets IP 120mg 10's 10's 33.00 relaxants Analgesic & Antipyretic / Muscle 11 13 Etoricoxilb Tablets IP 90mg 10's 10's 25.00 relaxants Analgesic & Antipyretic / Muscle 12 14 Ibuprofen 400 mg + Paracetamol 325 mg Tablet 10's 15's 5.50 relaxants Analgesic & Antipyretic / Muscle 13 15 Ibuprofen 200 mg film coated Tablet 10's 10's 1.80 relaxants Analgesic & Antipyretic / Muscle 14 16 Ibuprofen 400 mg film coated Tablet 10's 15's 3.50 relaxants Analgesic & Antipyretic -

Principios Activos Fotosensibles
PRINCIPIOS ACTIVOS FOTOSENSIBLES PROTEGE A TUS PRODUCTOS DE LA FOTODEGRADACIÓN PRINCIPIOS ACTIVOS FOTOSENSIBLES Adenosine Carbamazepine Digitoxin Heptacaine Methaqualone-1-oxide Phenothiazines Sulfisomidine Tinidazole Vitamin D Adrenaline Carbisocaine Digoxin Hexachlorophane Methotrexate Phenylbutazone Sulpyrine Tolmetin Vitamin E Adriamycin Carboplatin Dihydroergotamin Hydralazine Metronidazole Phenylephrine Suprofen Tretinoin Vitamin K1 Amidopyrin Carmustine Dihydropyridines Mifepristone Phenytoin Triamcinolone Vitamin K2 Hydrochlorothiazide Suramin Amidinohydrazones Cefotaxime Diltiazem Hydrocortisone Minoxidil Physostigmine Triamterene Warfarin Amiloride Cefuroxime axetil Diosgenin Mitomycin C Piroxicam Tauromustine (TCNU) Trifluoperazine Hydroxychloroquine Terbutaline 4-Aminobenzoic acid Cephaeline Diphenhydramine Hypochloride Mitonafide Pralidoxime Trimethoprim Aminophenazone Cephalexine Dipyridamole Ibuprofen Mitoxantrone Prednisolone Tetracyclines Ubidecarenone Thiazide Aminophylline Cephradine Dithranol Imipramin Molsidomine Primaquine Vancomycin Thiocolchicoside Aminosalicylic acid Chalcone Dobutamin Indapamide Morphine Proguanil Vinblastine Thioridazine Amiodarone Chloramphenicol Dopamine Indomethacin Nalidixic acid Promazine Vitamin A Thiorphan Amodiaquine Chlordiazepoxide Dothiepin Indoprofen Naproxen Promethazine Vitamin B (see also Thiothixene Amonafide Chloroquine Doxorubicin Isoprenaline Neocarzinostatin Proxibarbital riboflavine) Tiaprofenic acid Amphotericin B Chlorpromazine DTIC Isopropylamino- Nimodipine Psoralen -

Myofascial Pain Syndrome: a Treatment Review Mehul J
Himmelfarb Health Sciences Library, The George Washington University Health Sciences Research Commons Anesthesiology and Critical Care Medicine Faculty Anesthesiology and Critical Care Medicine Publications 6-2013 Myofascial pain syndrome: A treatment review Mehul J. Desai George Washington University Vikramjeet Saini Shawnjeet Saini Follow this and additional works at: https://hsrc.himmelfarb.gwu.edu/smhs_anesth_facpubs Part of the Anesthesia and Analgesia Commons Recommended Citation Desai, M. J., Saini, V., & Saini, S. (2013). Myofascial Pain Syndrome: A Treatment Review. Pain and Therapy, 2(1), 21–36. This Journal Article is brought to you for free and open access by the Anesthesiology and Critical Care Medicine at Health Sciences Research Commons. It has been accepted for inclusion in Anesthesiology and Critical Care Medicine Faculty Publications by an authorized administrator of Health Sciences Research Commons. For more information, please contact [email protected]. Pain Ther (2013) 2:21–36 DOI 10.1007/s40122-013-0006-y REVIEW Myofascial Pain Syndrome: A Treatment Review Mehul J. Desai • Vikramjeet Saini • Shawnjeet Saini To view enhanced content go to www.paintherapy-open.com Received: September 27, 2012 / Published online: February 12, 2013 Ó The Author(s) 2013. This article is published with open access at Springerlink.com ABSTRACT and nonpharmacologic treatments, the authors aim to provide clinicians with a more Myofascial pain syndrome (MPS) is defined as comprehensive knowledge of the interventions pain that originates from myofascial trigger for myofascial pain. points in skeletal muscle. It is prevalent in regional musculoskeletal pain syndromes, either alone or in combination with other pain Keywords: Analgesics; Anticonvulsants; generators. The appropriate evaluation and Muscle relaxants; Myofascial pain; management of myofascial pain is an important Nonpharmacological treatment; Pain; part of musculoskeletal rehabilitation, and Treatment regional axial and limb pain syndromes. -

Treatment for Acute Pain: an Evidence Map Technical Brief Number 33
Technical Brief Number 33 R Treatment for Acute Pain: An Evidence Map Technical Brief Number 33 Treatment for Acute Pain: An Evidence Map Prepared for: Agency for Healthcare Research and Quality U.S. Department of Health and Human Services 5600 Fishers Lane Rockville, MD 20857 www.ahrq.gov Contract No. 290-2015-0000-81 Prepared by: Minnesota Evidence-based Practice Center Minneapolis, MN Investigators: Michelle Brasure, Ph.D., M.S.P.H., M.L.I.S. Victoria A. Nelson, M.Sc. Shellina Scheiner, PharmD, B.C.G.P. Mary L. Forte, Ph.D., D.C. Mary Butler, Ph.D., M.B.A. Sanket Nagarkar, D.D.S., M.P.H. Jayati Saha, Ph.D. Timothy J. Wilt, M.D., M.P.H. AHRQ Publication No. 19(20)-EHC022-EF October 2019 Key Messages Purpose of review The purpose of this evidence map is to provide a high-level overview of the current guidelines and systematic reviews on pharmacologic and nonpharmacologic treatments for acute pain. We map the evidence for several acute pain conditions including postoperative pain, dental pain, neck pain, back pain, renal colic, acute migraine, and sickle cell crisis. Improved understanding of the interventions studied for each of these acute pain conditions will provide insight on which topics are ready for comprehensive comparative effectiveness review. Key messages • Few systematic reviews provide a comprehensive rigorous assessment of all potential interventions, including nondrug interventions, to treat pain attributable to each acute pain condition. Acute pain conditions that may need a comprehensive systematic review or overview of systematic reviews include postoperative postdischarge pain, acute back pain, acute neck pain, renal colic, and acute migraine. -

Federal Register / Vol. 60, No. 80 / Wednesday, April 26, 1995 / Notices DIX to the HTSUS—Continued
20558 Federal Register / Vol. 60, No. 80 / Wednesday, April 26, 1995 / Notices DEPARMENT OF THE TREASURY Services, U.S. Customs Service, 1301 TABLE 1.ÐPHARMACEUTICAL APPEN- Constitution Avenue NW, Washington, DIX TO THE HTSUSÐContinued Customs Service D.C. 20229 at (202) 927±1060. CAS No. Pharmaceutical [T.D. 95±33] Dated: April 14, 1995. 52±78±8 ..................... NORETHANDROLONE. A. W. Tennant, 52±86±8 ..................... HALOPERIDOL. Pharmaceutical Tables 1 and 3 of the Director, Office of Laboratories and Scientific 52±88±0 ..................... ATROPINE METHONITRATE. HTSUS 52±90±4 ..................... CYSTEINE. Services. 53±03±2 ..................... PREDNISONE. 53±06±5 ..................... CORTISONE. AGENCY: Customs Service, Department TABLE 1.ÐPHARMACEUTICAL 53±10±1 ..................... HYDROXYDIONE SODIUM SUCCI- of the Treasury. NATE. APPENDIX TO THE HTSUS 53±16±7 ..................... ESTRONE. ACTION: Listing of the products found in 53±18±9 ..................... BIETASERPINE. Table 1 and Table 3 of the CAS No. Pharmaceutical 53±19±0 ..................... MITOTANE. 53±31±6 ..................... MEDIBAZINE. Pharmaceutical Appendix to the N/A ............................. ACTAGARDIN. 53±33±8 ..................... PARAMETHASONE. Harmonized Tariff Schedule of the N/A ............................. ARDACIN. 53±34±9 ..................... FLUPREDNISOLONE. N/A ............................. BICIROMAB. 53±39±4 ..................... OXANDROLONE. United States of America in Chemical N/A ............................. CELUCLORAL. 53±43±0 -

THIOCOLCHICOSIDE AS MUSCLE RELAXANT: a REVIEW Introduction
Available Online through International Journal of Pharmacy and Biological Sciences (eISSN: 2230-7605) www.ijpbs.com IJPBS |Volume 1| Issue 3 |JULY-SEPT |2011|364-371 THIOCOLCHICOSIDE AS MUSCLE RELAXANT: A REVIEW A.R Umarkar*, S.R Bavaskar & P.N.Yewale. Shree.Sureshdada Jain Institute of Pharmaceutical & Education and Research Jamner Dist: Jalgaon 424206 *Corresponding Author Email: [email protected] Review Article RECEIVED ON 29-08-2011 ACCEPTED ON 25-09-2011 ABSTRACT Thiocolchicoside, is a synthetic sulphur derivative of colchicoside, a naturally occurring glucosidecontained in the Colchicum autumnale plant Thiocolchicoside has a selective affinity for g-amino-butyric acid (GABA) receptors and acts on themuscular contracture by activating the GABA-nergicinhibitory pathways thereby acting as a potent mus-cle relaxant Thiocolchicoside (Muscoril, Myoril, Neoflax) is a muscle relaxant with anti-inflammatory and analgesic effects. It acts as a competitive GABAA receptor antagonist and also inhibits glycine receptors with similar potency and nicotinic acetylcholine receptors to a much lesser extent. It has powerful convulsant activity and should not be used in seizure- prone individuals. Mode of action includes modulation of chemokine and prostanoid production and inhibition of neutrophil and endothelial cell adhesion molecules by which it interferes with the initiation and amplification of the joint inflammation. THC is a muscle relaxant given by oral in the treatment of arthritis in a usual dose equivalent to 8 mg first day to 12-16mg /day -

The Role of Biotransformation in Drug Discovery and Development
b Meta olis g m & ru D T o f x o i Journal of Drug Metabolism and l c a o Kebamo et al., J Drug Metab Toxicol 2015, 6:5 n l o r g u y o J Toxicology DOI: 10.4172/2157-7609.1000196 ISSN: 2157-7609 ReviewResearch Article Article OpenOpen Access Access The Role of Biotransformation in Drug Discovery and Development Selamu Kebamo1, Shibiru Tesema2 and Bekesho Geleta3* 1Department of Pharmacology, College of Medical and Health Science, Wollega University, Ethiopia 2Deapartment of Pharmacy, College of Public Health and Medicine Science, Jimma University, Jimma, Ethiopia 3Traditional and Modern Medicine Research, Ethiopian Public Health Institute, Addis Ababa, Ethiopia Abstract Biotransformation is a process by which organic compounds are transformed from one form to another, aided by organisms such as bacteria, fungi and enzymes. It plays a major role and determines the fate of the prospective drugs. Biotransformation must take place only after the drugs reach their specific target site and produce the desired effects. In addition, the nature of the metabolites produced from the drug, must be thoroughly studied; otherwise, the drugs would be rejected during the screening process. Hence, drug metabolism is a major criterion in the high- throughput screening of prospective drugs. Biotransformation has an important role in the determination of the pharmacokinetic parameters like oral bioavailability, drug-drug interaction, clearance and the half-life of the entity within the cell. It is very essential in the toxicity studies too. Biotransformation is used as a valuable strategy to build molecules, similar to parent drug in the drug discovery programme. -

Botulinum Toxin
Botulinum toxin From Wikipedia, the free encyclopedia Jump to: navigation, search Botulinum toxin Clinical data Pregnancy ? cat. Legal status Rx-Only (US) Routes IM (approved),SC, intradermal, into glands Identifiers CAS number 93384-43-1 = ATC code M03AX01 PubChem CID 5485225 DrugBank DB00042 Chemical data Formula C6760H10447N1743O2010S32 Mol. mass 149.322,3223 kDa (what is this?) (verify) Bontoxilysin Identifiers EC number 3.4.24.69 Databases IntEnz IntEnz view BRENDA BRENDA entry ExPASy NiceZyme view KEGG KEGG entry MetaCyc metabolic pathway PRIAM profile PDB structures RCSB PDB PDBe PDBsum Gene Ontology AmiGO / EGO [show]Search Botulinum toxin is a protein and neurotoxin produced by the bacterium Clostridium botulinum. Botulinum toxin can cause botulism, a serious and life-threatening illness in humans and animals.[1][2] When introduced intravenously in monkeys, type A (Botox Cosmetic) of the toxin [citation exhibits an LD50 of 40–56 ng, type C1 around 32 ng, type D 3200 ng, and type E 88 ng needed]; these are some of the most potent neurotoxins known.[3] Popularly known by one of its trade names, Botox, it is used for various cosmetic and medical procedures. Botulinum can be absorbed from eyes, mucous membranes, respiratory tract or non-intact skin.[4] Contents [show] [edit] History Justinus Kerner described botulinum toxin as a "sausage poison" and "fatty poison",[5] because the bacterium that produces the toxin often caused poisoning by growing in improperly handled or prepared meat products. It was Kerner, a physician, who first conceived a possible therapeutic use of botulinum toxin and coined the name botulism (from Latin botulus meaning "sausage"). -

Management of Frozen Shoulder: a Systematic Review and Cost-Effectiveness Analysis
Health Technology Assessment 2012; Vol. 16: No.111 Acknowledgements Health Technology Assessment 2012; Vol. 16: No. 11 ISSNContribution 1366-5278 of authors ISSN 1366-5278 ReferencesAbstractAppendix 7.2 Sodium hyaluronate Appendix 7.3 Physical therapy GlossaryAppendixAppendix 1 7.4 Acupuncture (with or without physical therapy) SearchAppendix strategies 7.5 Manipulation for effectiveness under review anaesthesia 179 ListBIOSIS ofAppendix abbreviations Previews 7.6 Distension CumulativeAppendix Index7.7 Capsular to Nursing release and Allied Health Literature (CINAHL) ExecutiveCochrane summary Central Register of Controlled Trials (CENTRAL) Clinicaltrials.govBackgroundAppendix 8 CochraneObjectivesStudy quality Database of Systematic Reviews (CDSR) 369 ConferenceMethodsControlled Proceedings trials Citation Index: Science Management of frozen shoulder: DatabaseResultsObservational of Abstracts studies of Reviews of Effects (DARE) EMBASEConclusions a systematic review and HealthFundingAppendix Management 9 Information Consortium (HMIC) HealthWinBUGS Technology code Assessment (HTA) database 373 cost-effectiveness analysis Chapter 1 Latin American and Caribbean Health Sciences Literature (LILACS) BackgroundAppendix 10 Manual, Alternative and Natural Therapy (MANTIS) TheMixed-treatment decision problem comparison 375 NHS Economic Evaluation Database (NHS EED) FrozenNetwork shoulder 1: studies of any intervention (i.e. conservative and invasive) and any quality National Technical Information Service (NTIS) DiagnosisNetwork 2:and studies management of any intervention that were of good or satisfactory quality (i.e. method E Maund, D Craig, S Suekarran, AR Neilson, PASCAL Previousof randomisation systematic was reviews adequate and outcome assessment was blinded) Physiotherapy Evidence Database (PEDro) K Wright, S Brealey, L Dennis, L Goodchild, FocusNetwork of the 3: studiessynthesis of conservative treatments of any quality PREMEDLINE Network 4: studies of conservative treatments that were of good or satisfactory quality (i.e. -

Non-Steroidal Anti-Inflammatory Drugs for Acute Low Back Pain Van Der Gaag, Wendelien H; Roelofs, Pepijn Ddm; Enthoven, Wendy Tm; Van Tulder, Maurits W; Koes, Bart W
VU Research Portal Non-steroidal anti-inflammatory drugs for acute low back pain van der Gaag, Wendelien H; Roelofs, Pepijn Ddm; Enthoven, Wendy Tm; van Tulder, Maurits W; Koes, Bart W published in The Cochrane database of systematic reviews 2020 DOI (link to publisher) 10.1002/14651858.CD013581 document version Publisher's PDF, also known as Version of record document license Article 25fa Dutch Copyright Act Link to publication in VU Research Portal citation for published version (APA) van der Gaag, W. H., Roelofs, P. D., Enthoven, W. T., van Tulder, M. W., & Koes, B. W. (2020). Non-steroidal anti-inflammatory drugs for acute low back pain. The Cochrane database of systematic reviews, 2020(4), 1-151. [CD013581]. https://doi.org/10.1002/14651858.CD013581 General rights Copyright and moral rights for the publications made accessible in the public portal are retained by the authors and/or other copyright owners and it is a condition of accessing publications that users recognise and abide by the legal requirements associated with these rights. • Users may download and print one copy of any publication from the public portal for the purpose of private study or research. • You may not further distribute the material or use it for any profit-making activity or commercial gain • You may freely distribute the URL identifying the publication in the public portal ? Take down policy If you believe that this document breaches copyright please contact us providing details, and we will remove access to the work immediately and investigate your claim. E-mail address: [email protected] Download date: 24. -

Marrakesh Agreement Establishing the World Trade Organization
No. 31874 Multilateral Marrakesh Agreement establishing the World Trade Organ ization (with final act, annexes and protocol). Concluded at Marrakesh on 15 April 1994 Authentic texts: English, French and Spanish. Registered by the Director-General of the World Trade Organization, acting on behalf of the Parties, on 1 June 1995. Multilat ral Accord de Marrakech instituant l©Organisation mondiale du commerce (avec acte final, annexes et protocole). Conclu Marrakech le 15 avril 1994 Textes authentiques : anglais, français et espagnol. Enregistré par le Directeur général de l'Organisation mondiale du com merce, agissant au nom des Parties, le 1er juin 1995. Vol. 1867, 1-31874 4_________United Nations — Treaty Series • Nations Unies — Recueil des Traités 1995 Table of contents Table des matières Indice [Volume 1867] FINAL ACT EMBODYING THE RESULTS OF THE URUGUAY ROUND OF MULTILATERAL TRADE NEGOTIATIONS ACTE FINAL REPRENANT LES RESULTATS DES NEGOCIATIONS COMMERCIALES MULTILATERALES DU CYCLE D©URUGUAY ACTA FINAL EN QUE SE INCORPOR N LOS RESULTADOS DE LA RONDA URUGUAY DE NEGOCIACIONES COMERCIALES MULTILATERALES SIGNATURES - SIGNATURES - FIRMAS MINISTERIAL DECISIONS, DECLARATIONS AND UNDERSTANDING DECISIONS, DECLARATIONS ET MEMORANDUM D©ACCORD MINISTERIELS DECISIONES, DECLARACIONES Y ENTEND MIENTO MINISTERIALES MARRAKESH AGREEMENT ESTABLISHING THE WORLD TRADE ORGANIZATION ACCORD DE MARRAKECH INSTITUANT L©ORGANISATION MONDIALE DU COMMERCE ACUERDO DE MARRAKECH POR EL QUE SE ESTABLECE LA ORGANIZACI N MUND1AL DEL COMERCIO ANNEX 1 ANNEXE 1 ANEXO 1 ANNEX