HIRA Orchestrates a Dynamic Chromatin Landscape in Senescence and Is Required for Suppression of Neoplasia
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Histone Isoform H2A1H Promotes Attainment of Distinct Physiological
Bhattacharya et al. Epigenetics & Chromatin (2017) 10:48 DOI 10.1186/s13072-017-0155-z Epigenetics & Chromatin RESEARCH Open Access Histone isoform H2A1H promotes attainment of distinct physiological states by altering chromatin dynamics Saikat Bhattacharya1,4,6, Divya Reddy1,4, Vinod Jani5†, Nikhil Gadewal3†, Sanket Shah1,4, Raja Reddy2,4, Kakoli Bose2,4, Uddhavesh Sonavane5, Rajendra Joshi5 and Sanjay Gupta1,4* Abstract Background: The distinct functional efects of the replication-dependent histone H2A isoforms have been dem- onstrated; however, the mechanistic basis of the non-redundancy remains unclear. Here, we have investigated the specifc functional contribution of the histone H2A isoform H2A1H, which difers from another isoform H2A2A3 in the identity of only three amino acids. Results: H2A1H exhibits varied expression levels in diferent normal tissues and human cancer cell lines (H2A1C in humans). It also promotes cell proliferation in a context-dependent manner when exogenously overexpressed. To uncover the molecular basis of the non-redundancy, equilibrium unfolding of recombinant H2A1H-H2B dimer was performed. We found that the M51L alteration at the H2A–H2B dimer interface decreases the temperature of melting of H2A1H-H2B by ~ 3 °C as compared to the H2A2A3-H2B dimer. This diference in the dimer stability is also refected in the chromatin dynamics as H2A1H-containing nucleosomes are more stable owing to M51L and K99R substitu- tions. Molecular dynamic simulations suggest that these substitutions increase the number of hydrogen bonds and hydrophobic interactions of H2A1H, enabling it to form more stable nucleosomes. Conclusion: We show that the M51L and K99R substitutions, besides altering the stability of histone–histone and histone–DNA complexes, have the most prominent efect on cell proliferation, suggesting that the nucleosome sta- bility is intimately linked with the physiological efects observed. -

University of California, San Diego
UNIVERSITY OF CALIFORNIA, SAN DIEGO The post-terminal differentiation fate of RNAs revealed by next-generation sequencing A dissertation submitted in partial satisfaction of the requirements for the degree Doctor of Philosophy in Biomedical Sciences by Gloria Kuo Lefkowitz Committee in Charge: Professor Benjamin D. Yu, Chair Professor Richard Gallo Professor Bruce A. Hamilton Professor Miles F. Wilkinson Professor Eugene Yeo 2012 Copyright Gloria Kuo Lefkowitz, 2012 All rights reserved. The Dissertation of Gloria Kuo Lefkowitz is approved, and it is acceptable in quality and form for publication on microfilm and electronically: __________________________________________________________________ __________________________________________________________________ __________________________________________________________________ __________________________________________________________________ __________________________________________________________________ Chair University of California, San Diego 2012 iii DEDICATION Ma and Ba, for your early indulgence and support. Matt and James, for choosing more practical callings. Roy, my love, for patiently sharing the ups and downs of this journey. iv EPIGRAPH It is foolish to tear one's hair in grief, as though sorrow would be made less by baldness. ~Cicero v TABLE OF CONTENTS Signature Page .............................................................................................................. iii Dedication .................................................................................................................... -

Analysis of Trans Esnps Infers Regulatory Network Architecture
Analysis of trans eSNPs infers regulatory network architecture Anat Kreimer Submitted in partial fulfillment of the requirements for the degree of Doctor of Philosophy in the Graduate School of Arts and Sciences COLUMBIA UNIVERSITY 2014 © 2014 Anat Kreimer All rights reserved ABSTRACT Analysis of trans eSNPs infers regulatory network architecture Anat Kreimer eSNPs are genetic variants associated with transcript expression levels. The characteristics of such variants highlight their importance and present a unique opportunity for studying gene regulation. eSNPs affect most genes and their cell type specificity can shed light on different processes that are activated in each cell. They can identify functional variants by connecting SNPs that are implicated in disease to a molecular mechanism. Examining eSNPs that are associated with distal genes can provide insights regarding the inference of regulatory networks but also presents challenges due to the high statistical burden of multiple testing. Such association studies allow: simultaneous investigation of many gene expression phenotypes without assuming any prior knowledge and identification of unknown regulators of gene expression while uncovering directionality. This thesis will focus on such distal eSNPs to map regulatory interactions between different loci and expose the architecture of the regulatory network defined by such interactions. We develop novel computational approaches and apply them to genetics-genomics data in human. We go beyond pairwise interactions to define network motifs, including regulatory modules and bi-fan structures, showing them to be prevalent in real data and exposing distinct attributes of such arrangements. We project eSNP associations onto a protein-protein interaction network to expose topological properties of eSNPs and their targets and highlight different modes of distal regulation. -
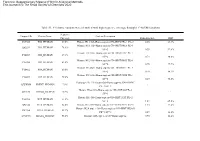
Table S1. 49 Histone Variants Were Identified with High Sequence Coverage Through LC-MS/MS Analysis Electronic Supplementary
Electronic Supplementary Material (ESI) for Analytical Methods. This journal is © The Royal Society of Chemistry 2020 Table S1. 49 histone variants were identified with high sequence coverage through LC-MS/MS analysis Sequence Uniprot IDs Protein Name Protein Description Coverage Ratio E2+/E2- RSD P07305 H10_HUMAN 67.5% Histone H1.0 OS=Homo sapiens GN=H1F0 PE=1 SV=3 4.85 23.3% Histone H1.1 OS=Homo sapiens GN=HIST1H1A PE=1 Q02539 H11_HUMAN 74.4% SV=3 0.35 92.6% Histone H1.2 OS=Homo sapiens GN=HIST1H1C PE=1 P16403 H12_HUMAN 67.1% SV=2 0.73 80.6% Histone H1.3 OS=Homo sapiens GN=HIST1H1D PE=1 P16402 H13_HUMAN 63.8% SV=2 0.75 77.7% Histone H1.4 OS=Homo sapiens GN=HIST1H1E PE=1 P10412 H14_HUMAN 69.0% SV=2 0.70 80.3% Histone H1.5 OS=Homo sapiens GN=HIST1H1B PE=1 P16401 H15_HUMAN 79.6% SV=3 0.29 98.3% Testis-specific H1 histone OS=Homo sapiens GN=H1FNT Q75WM6 H1FNT_HUMAN 7.8% \ \ PE=2 SV=3 Histone H1oo OS=Homo sapiens GN=H1FOO PE=2 Q8IZA3 H1FOO_HUMAN 5.2% \ \ SV=1 Histone H1t OS=Homo sapiens GN=HIST1H1T PE=2 P22492 H1T_HUMAN 31.4% SV=4 1.42 65.0% Q92522 H1X_HUMAN 82.6% Histone H1x OS=Homo sapiens GN=H1FX PE=1 SV=1 1.15 33.2% Histone H2A type 1 OS=Homo sapiens GN=HIST1H2AG P0C0S8 H2A1_HUMAN 99.2% PE=1 SV=2 0.57 26.8% Q96QV6 H2A1A_HUMAN 58.0% Histone H2A type 1-A OS=Homo sapiens 0.90 11.2% GN=HIST1H2AA PE=1 SV=3 Histone H2A type 1-B/E OS=Homo sapiens P04908 H2A1B_HUMAN 99.2% GN=HIST1H2AB PE=1 SV=2 0.92 30.2% Histone H2A type 1-C OS=Homo sapiens Q93077 H2A1C_HUMAN 100.0% GN=HIST1H2AC PE=1 SV=3 0.76 27.6% Histone H2A type 1-D OS=Homo sapiens P20671 -

The Landscape of Somatic Mutations in Epigenetic Regulators Across 1,000 Paediatric Cancer Genomes
ARTICLE Received 24 Sep 2013 | Accepted 12 Mar 2014 | Published 8 Apr 2014 DOI: 10.1038/ncomms4630 The landscape of somatic mutations in epigenetic regulators across 1,000 paediatric cancer genomes Robert Huether1,*, Li Dong2,*, Xiang Chen1, Gang Wu1, Matthew Parker1, Lei Wei1, Jing Ma2, Michael N. Edmonson1, Erin K. Hedlund1, Michael C. Rusch1, Sheila A. Shurtleff2, Heather L. Mulder3, Kristy Boggs3, Bhavin Vadordaria3, Jinjun Cheng2, Donald Yergeau3, Guangchun Song2, Jared Becksfort1, Gordon Lemmon1, Catherine Weber2, Zhongling Cai2, Jinjun Dang2, Michael Walsh4, Amanda L. Gedman2, Zachary Faber2, John Easton3, Tanja Gruber2,4, Richard W. Kriwacki5, Janet F. Partridge6, Li Ding7,8,9, Richard K. Wilson7,8,9, Elaine R. Mardis7,8,9, Charles G. Mullighan2, Richard J. Gilbertson10, Suzanne J. Baker10, Gerard Zambetti6, David W. Ellison2, Jinghui Zhang1 & James R. Downing2 Studies of paediatric cancers have shown a high frequency of mutation across epigenetic regulators. Here we sequence 633 genes, encoding the majority of known epigenetic regulatory proteins, in over 1,000 paediatric tumours to define the landscape of somatic mutations in epigenetic regulators in paediatric cancer. Our results demonstrate a marked variation in the frequency of gene mutations across 21 different paediatric cancer subtypes, with the highest frequency of mutations detected in high-grade gliomas, T-lineage acute lymphoblastic leukaemia and medulloblastoma, and a paucity of mutations in low-grade glioma and retinoblastoma. The most frequently mutated genes are H3F3A, PHF6, ATRX, KDM6A, SMARCA4, ASXL2, CREBBP, EZH2, MLL2, USP7, ASXL1, NSD2, SETD2, SMC1A and ZMYM3. We identify novel loss-of-function mutations in the ubiquitin-specific processing protease 7 (USP7) in paediatric leukaemia, which result in decreased deubiquitination activity. -

Differences for Ectopic Versus Eutopic Cells
556 RBMO VOLUME 39 ISSUE 4 2019 ARTICLE Chemosensitivity and chemoresistance in endometriosis – differences for ectopic versus eutopic cells BIOGRAPHY Andres Salumets is Professor of Reproductive Medicine at the University of Tartu, and Scientific Head at the Competence Centre on Health Technologies, Tartu, Estonia. He has been involved in assisted reproduction for 20 years, first as an embryologist and later as a researcher. His major interests are endometriosis, endometrial biology and implantation. Darja Lavogina1,2,*, Külli Samuel1, Arina Lavrits1,3, Alvin Meltsov1, Deniss Sõritsa1,4,5, Ülle Kadastik6, Maire Peters1,4, Ago Rinken2, Andres Salumets1,4,7, 8 KEY MESSAGE Akt/PKB inhibitor GSK690693, CK2 inhibitor ARC-775, MAPK pathway inhibitor sorafenib, proteasome inhibitor bortezomib, and microtubule-depolymerizing toxin MMAE showed higher cytotoxicity in eutopic cells. In contrast, 10 µmol/l of the anthracycline toxin doxorubicin caused cellular death in ectopic cells more effectively than in eutopic cells, underlining the potential of doxorubicin in endometriosis research. ABSTRACT Research question: Endometriosis is a common gynaecological disease defined by the presence of endometrium-like tissue outside the uterus. This complex disease, often accompanied by severe pain and infertility, causes a significant medical and socioeconomic burden; hence, novel strategies are being sought for the treatment of endometriosis. Here, we set out to explore the cytotoxic effects of a panel of compounds to find toxins with different efficiency in eutopic versus ectopic cells, thus highlighting alterations in the corresponding molecular pathways. Design: The effect on cellular viability of 14 compounds was established in a cohort of paired eutopic and ectopic endometrial stromal cell samples from 11 patients. -

DNA Methylation Changes in Down Syndrome Derived Neural Ipscs Uncover Co-Dysregulation of ZNF and HOX3 Families of Transcription
Laan et al. Clinical Epigenetics (2020) 12:9 https://doi.org/10.1186/s13148-019-0803-1 RESEARCH Open Access DNA methylation changes in Down syndrome derived neural iPSCs uncover co- dysregulation of ZNF and HOX3 families of transcription factors Loora Laan1†, Joakim Klar1†, Maria Sobol1, Jan Hoeber1, Mansoureh Shahsavani2, Malin Kele2, Ambrin Fatima1, Muhammad Zakaria1, Göran Annerén1, Anna Falk2, Jens Schuster1 and Niklas Dahl1* Abstract Background: Down syndrome (DS) is characterized by neurodevelopmental abnormalities caused by partial or complete trisomy of human chromosome 21 (T21). Analysis of Down syndrome brain specimens has shown global epigenetic and transcriptional changes but their interplay during early neurogenesis remains largely unknown. We differentiated induced pluripotent stem cells (iPSCs) established from two DS patients with complete T21 and matched euploid donors into two distinct neural stages corresponding to early- and mid-gestational ages. Results: Using the Illumina Infinium 450K array, we assessed the DNA methylation pattern of known CpG regions and promoters across the genome in trisomic neural iPSC derivatives, and we identified a total of 500 stably and differentially methylated CpGs that were annotated to CpG islands of 151 genes. The genes were enriched within the DNA binding category, uncovering 37 factors of importance for transcriptional regulation and chromatin structure. In particular, we observed regional epigenetic changes of the transcription factor genes ZNF69, ZNF700 and ZNF763 as well as the HOXA3, HOXB3 and HOXD3 genes. A similar clustering of differential methylation was found in the CpG islands of the HIST1 genes suggesting effects on chromatin remodeling. Conclusions: The study shows that early established differential methylation in neural iPSC derivatives with T21 are associated with a set of genes relevant for DS brain development, providing a novel framework for further studies on epigenetic changes and transcriptional dysregulation during T21 neurogenesis. -

Functional Annotations of Single-Nucleotide Polymorphism
CLINICAL RESEARCH e-ISSN 1643-3750 © Med Sci Monit, 2020; 26: e922710 DOI: 10.12659/MSM.922710 Received: 2020.01.08 Accepted: 2020.02.20 Functional Annotations of Single-Nucleotide Available online: 2020.03.30 Published: 2020.05.25 Polymorphism (SNP)-Based and Gene-Based Genome-Wide Association Studies Show Genes Affecting Keratitis Susceptibility Authors’ Contribution: BCDEF 1 Yue Xu* 1 Department of Ophthalmology, First Affiliated Hospital of Soochow University, Study Design A BCDEF 2 Xiao-Lin Yang* Suzhou, Jiangsu, P.R. China Data Collection B 2 Center for Genetic Epidemiology and Genomics, School of Public Health, Medical Statistical Analysis C BCD 1 Xiao-Long Yang College of Soochow University, Suzhou, Jiangsu, P.R. China Data Interpretation D BC 1 Ya-Ru Ren Manuscript Preparation E BC 1 Xin-Yu Zhuang Literature Search F Funds Collection G ADE 2 Lei Zhang ADE 1 Xiao-Feng Zhang * Yue Xu and Xiao-Lin Yang contributed equally Corresponding Authors: Xiao-Feng Zhang, e-mail: [email protected], Lei Zhang, e-mail: [email protected] Source of support: Departmental sources Background: Keratitis is a complex condition in humans and is the second most common cause of legal blindness worldwide. Material/Methods: To reveal the genomic loci underlying keratitis, we performed functional annotations of SNP-based and gene- based genome-wide association studies of keratitis in the UK Biobank (UKB) cohort with 337 199 subjects of European ancestry. Results: The publicly available SNP-based association results showed a total of 34 SNPs, from 14 distinct loci, associated with keratitis in the UKB. Gene-based association analysis identified 2 significant genes:IQCF3 (p=2.0×10–6) and SOD3 (p=2.0×10–6). -

Hira Peracha a Thesis
DIAGNOSIS METHOD FOR MUCOPOLYSACCHARIDOSES by Hira Peracha A thesis submitted to the Faculty of the University of Delaware in partial fulfillment of the requirements for the degree of Bachelor of Arts in Biological Sciences with Distinction Spring 2018 © 2018 Hira Peracha All Rights Reserved i DIAGNOSIS METHOD FOR MUCOPOLYSACCHARIDOSES by Hira Peracha Approved: __________________________________________________________ Shunji Tomatsu, MD, Ph.D. Professor in charge of thesis on behalf of the Advisory Committee Approved: __________________________________________________________ Deni Galileo, Ph.D. Professor in charge of thesis on behalf of the Advisory Committee Approved: __________________________________________________________ Jessica Tanis, Ph.D. Committee member from the Department of Department Name Approved: __________________________________________________________ Barbara Settles, Ph.D. Committee member from the Board of Senior Thesis Readers Approved: __________________________________________________________ Michael Chajes, Ph.D. Chair of the University Committee on Student and Faculty Honors ii “Education is the key to unlocking the world, a passport to freedom.” -Oprah Winfrey iii ACKNOWLEDGMENTS I dedicate my thesis to my late grandfather, who taught me the value of life, hard work and sacrifice- especially in sickness and in health. I would like to express my deepest acknowledgements to my family. I could not have achieved all of my dreams and goals in life without my parents; my mom and my dad, whose sacrifices, love and support have been endless and infinite. Thank you for raising me to believe that anything was possible, and for spending your entire lives to better mine. To my younger siblings, Saarah, Hafsah and Arif, who have been the light in the dark times in my life and have always brought joy and happiness in my life. -

M2139: Molecular Analysis for Gliomas
Molecular Analysis for Gliomas Policy Number: AHS – M2139 – Molecular Analysis Prior Policy Name and Number, as applicable: for Gliomas • AHS – M2139 – Analysis of MGMT Promoter Methylation for Malignant Gliomas Initial Presentation Date: 06/16/2021 Revision Date: N/A I. Policy Description Glioma refers to tumors resulting from metaplastic transformation of glial tissue of the nervous system. Tumors have historically been classified by the retained histologic features of the three types of glial cells: astrocytes, oligodendrocytes, and ependymal cells. Tumors of each type can vary widely in aggressiveness, response to treatment, and prognosis (Louis, Schiff, & Batchelor, 2020). Molecular genetic features were added to histopathologic appearance in the current WHO classification to yield more biologically homogeneous and narrowly defined diagnostic entities for greater diagnostic accuracy, improved patient management, more accurate determinations of prognosis, and better treatment response (Louis et al., 2016). II. Related Policies Policy Number Policy Title AHS-M2109 Molecular Panel Testing of Cancers for Diagnosis, Prognosis, and Identification of Targeted Therapy III. Indications and/or Limitations of Coverage Application of coverage criteria is dependent upon an individual’s benefit coverage at the time of the request 1. MGMT promoter methylation testing IS MEDICALLY NECESSARY for prognosis of malignant gliomas. 2. IDH1 and IDH2 testing IS MEDICALLY NECESSARY for prognosis of malignant gliomas. M2139 Molecular Analysis for Gliomas Page 1 of 15 3. Testing for the co-deletion of 1p and 19q by either fluorescence in situ hybridization (FISH) or polymerase chain reaction (PCR) for the characterization of gliomas and/or to guide treatment decisions IS MEDICALLY NECESSARY. 4. ATRX mutation testing via EITHER immunohistochemistry OR gene sequencing IS MEDICALLY NECESSARY for the prognosis of malignant gliomas. -

Anti-Histone H2B Antibody (ARG41373)
Product datasheet [email protected] ARG41373 Package: 100 μl anti-Histone H2B antibody Store at: -20°C Summary Product Description Rabbit Polyclonal antibody recognizes Histone H2B Tested Reactivity Hu, Ms, Rat Tested Application FACS, ICC/IF, IHC-P, IP, WB Host Rabbit Clonality Polyclonal Isotype IgG Target Name Histone H2B Antigen Species Human Immunogen Synthetic peptide derived from Human Histone H2B. Conjugation Un-conjugated Alternate Names Histone H2B type 1-K; H2BFAiii; H2BFT; H2B/S; H2B K; H2BK; HIRA-interacting protein 1 Application Instructions Application table Application Dilution FACS 1:50 ICC/IF 1:50 - 1:200 IHC-P 1:50 - 1:200 IP 1:30 WB 1:5000 - 1:20000 Application Note * The dilutions indicate recommended starting dilutions and the optimal dilutions or concentrations should be determined by the scientist. Calculated Mw 14 kDa Observed Size ~ 14 kDa Properties Form Liquid Purification Affinity purified. Buffer PBS (pH 7.4), 150 mM NaCl, 0.02% Sodium azide and 50% Glycerol. Preservative 0.02% Sodium azide Stabilizer 50% Glycerol Storage instruction For continuous use, store undiluted antibody at 2-8°C for up to a week. For long-term storage, aliquot www.arigobio.com 1/2 and store at -20°C. Storage in frost free freezers is not recommended. Avoid repeated freeze/thaw cycles. Suggest spin the vial prior to opening. The antibody solution should be gently mixed before use. Note For laboratory research only, not for drug, diagnostic or other use. Bioinformation Gene Symbol HIST1H2BK Gene Full Name histone cluster 1, H2bk Background Histones are basic nuclear proteins that are responsible for the nucleosome structure of the chromosomal fiber in eukaryotes. -

Molecular Effects of Isoflavone Supplementation Human Intervention Studies and Quantitative Models for Risk Assessment
Molecular effects of isoflavone supplementation Human intervention studies and quantitative models for risk assessment Vera van der Velpen Thesis committee Promotors Prof. Dr Pieter van ‘t Veer Professor of Nutritional Epidemiology Wageningen University Prof. Dr Evert G. Schouten Emeritus Professor of Epidemiology and Prevention Wageningen University Co-promotors Dr Anouk Geelen Assistant professor, Division of Human Nutrition Wageningen University Dr Lydia A. Afman Assistant professor, Division of Human Nutrition Wageningen University Other members Prof. Dr Jaap Keijer, Wageningen University Dr Hubert P.J.M. Noteborn, Netherlands Food en Consumer Product Safety Authority Prof. Dr Yvonne T. van der Schouw, UMC Utrecht Dr Wendy L. Hall, King’s College London This research was conducted under the auspices of the Graduate School VLAG (Advanced studies in Food Technology, Agrobiotechnology, Nutrition and Health Sciences). Molecular effects of isoflavone supplementation Human intervention studies and quantitative models for risk assessment Vera van der Velpen Thesis submitted in fulfilment of the requirements for the degree of doctor at Wageningen University by the authority of the Rector Magnificus Prof. Dr M.J. Kropff, in the presence of the Thesis Committee appointed by the Academic Board to be defended in public on Friday 20 June 2014 at 13.30 p.m. in the Aula. Vera van der Velpen Molecular effects of isoflavone supplementation: Human intervention studies and quantitative models for risk assessment 154 pages PhD thesis, Wageningen University, Wageningen, NL (2014) With references, with summaries in Dutch and English ISBN: 978-94-6173-952-0 ABSTRact Background: Risk assessment can potentially be improved by closely linked experiments in the disciplines of epidemiology and toxicology.