Nfe2l1) As a Hypoxia- Inducible Factor and Its Oxygen-Dependent Regulation in Vitro and in Vivo
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Activated Peripheral-Blood-Derived Mononuclear Cells
Transcription factor expression in lipopolysaccharide- activated peripheral-blood-derived mononuclear cells Jared C. Roach*†, Kelly D. Smith*‡, Katie L. Strobe*, Stephanie M. Nissen*, Christian D. Haudenschild§, Daixing Zhou§, Thomas J. Vasicek¶, G. A. Heldʈ, Gustavo A. Stolovitzkyʈ, Leroy E. Hood*†, and Alan Aderem* *Institute for Systems Biology, 1441 North 34th Street, Seattle, WA 98103; ‡Department of Pathology, University of Washington, Seattle, WA 98195; §Illumina, 25861 Industrial Boulevard, Hayward, CA 94545; ¶Medtronic, 710 Medtronic Parkway, Minneapolis, MN 55432; and ʈIBM Computational Biology Center, P.O. Box 218, Yorktown Heights, NY 10598 Contributed by Leroy E. Hood, August 21, 2007 (sent for review January 7, 2007) Transcription factors play a key role in integrating and modulating system. In this model system, we activated peripheral-blood-derived biological information. In this study, we comprehensively measured mononuclear cells, which can be loosely termed ‘‘macrophages,’’ the changing abundances of mRNAs over a time course of activation with lipopolysaccharide (LPS). We focused on the precise mea- of human peripheral-blood-derived mononuclear cells (‘‘macro- surement of mRNA concentrations. There is currently no high- phages’’) with lipopolysaccharide. Global and dynamic analysis of throughput technology that can precisely and sensitively measure all transcription factors in response to a physiological stimulus has yet to mRNAs in a system, although such technologies are likely to be be achieved in a human system, and our efforts significantly available in the near future. To demonstrate the potential utility of advanced this goal. We used multiple global high-throughput tech- such technologies, and to motivate their development and encour- nologies for measuring mRNA levels, including massively parallel age their use, we produced data from a combination of two distinct signature sequencing and GeneChip microarrays. -

Expression Profiling of KLF4
Expression Profiling of KLF4 AJCR0000006 Supplemental Data Figure S1. Snapshot of enriched gene sets identified by GSEA in Klf4-null MEFs. Figure S2. Snapshot of enriched gene sets identified by GSEA in wild type MEFs. 98 Am J Cancer Res 2011;1(1):85-97 Table S1: Functional Annotation Clustering of Genes Up-Regulated in Klf4 -Null MEFs ILLUMINA_ID Gene Symbol Gene Name (Description) P -value Fold-Change Cell Cycle 8.00E-03 ILMN_1217331 Mcm6 MINICHROMOSOME MAINTENANCE DEFICIENT 6 40.36 ILMN_2723931 E2f6 E2F TRANSCRIPTION FACTOR 6 26.8 ILMN_2724570 Mapk12 MITOGEN-ACTIVATED PROTEIN KINASE 12 22.19 ILMN_1218470 Cdk2 CYCLIN-DEPENDENT KINASE 2 9.32 ILMN_1234909 Tipin TIMELESS INTERACTING PROTEIN 5.3 ILMN_1212692 Mapk13 SAPK/ERK/KINASE 4 4.96 ILMN_2666690 Cul7 CULLIN 7 2.23 ILMN_2681776 Mapk6 MITOGEN ACTIVATED PROTEIN KINASE 4 2.11 ILMN_2652909 Ddit3 DNA-DAMAGE INDUCIBLE TRANSCRIPT 3 2.07 ILMN_2742152 Gadd45a GROWTH ARREST AND DNA-DAMAGE-INDUCIBLE 45 ALPHA 1.92 ILMN_1212787 Pttg1 PITUITARY TUMOR-TRANSFORMING 1 1.8 ILMN_1216721 Cdk5 CYCLIN-DEPENDENT KINASE 5 1.78 ILMN_1227009 Gas2l1 GROWTH ARREST-SPECIFIC 2 LIKE 1 1.74 ILMN_2663009 Rassf5 RAS ASSOCIATION (RALGDS/AF-6) DOMAIN FAMILY 5 1.64 ILMN_1220454 Anapc13 ANAPHASE PROMOTING COMPLEX SUBUNIT 13 1.61 ILMN_1216213 Incenp INNER CENTROMERE PROTEIN 1.56 ILMN_1256301 Rcc2 REGULATOR OF CHROMOSOME CONDENSATION 2 1.53 Extracellular Matrix 5.80E-06 ILMN_2735184 Col18a1 PROCOLLAGEN, TYPE XVIII, ALPHA 1 51.5 ILMN_1223997 Crtap CARTILAGE ASSOCIATED PROTEIN 32.74 ILMN_2753809 Mmp3 MATRIX METALLOPEPTIDASE -

Roles of NRF3 in the Hallmarks of Cancer: Proteasomal Inactivation of Tumor Suppressors
cancers Review Roles of NRF3 in the Hallmarks of Cancer: Proteasomal Inactivation of Tumor Suppressors Akira Kobayashi 1,2 1 Laboratory for Genetic Code, Graduate School of Life and Medical Sciences, Doshisha University, Kyotanabe, Kyoto 610-0394, Japan; [email protected]; Tel.: +81-774-65-6273 2 Department of Medical Life Systems, Faculty of Life and Medical Sciences, Doshisha University, Kyotanabe, Kyoto 610-0394, Japan Received: 1 September 2020; Accepted: 17 September 2020; Published: 20 September 2020 Simple Summary: This review summarizes recent advances in our understanding of the physiological roles of the NFE2-related factor 2 (NRF2)-related transcription factor NRF3 in cancer. NRF3 confers cells with six so-called “hallmarks of cancer” through upregulating gene expression of specific target genes, leading to tumorigenesis and cancer malignancy. These driver gene-like functions of NRF3 in cancer are distinct from those of NRF2. Abstract: The physiological roles of the NRF2-related transcription factor NRF3 (NFE2L3) have remained unknown for decades. The remarkable development of human cancer genome databases has led to strong suggestions that NRF3 has functional significance in cancer; specifically,high NRF3 mRNA levels are induced in many cancer types, such as colorectal cancer and pancreatic adenocarcinoma, and are associated with poor prognosis. On the basis of this information, the involvement of NRF3 in tumorigenesis and cancer malignancy has been recently proposed. NRF3 confers cancer cells with selective growth advantages by enhancing 20S proteasome assembly through induction of the chaperone gene proteasome maturation protein (POMP) and consequently promoting degradation of the tumor suppressors p53 and retinoblastoma (Rb) in a ubiquitin-independent manner. -

NFE2L1 Antibody Cat
NFE2L1 Antibody Cat. No.: 31-335 NFE2L1 Antibody Specifications HOST SPECIES: Rabbit SPECIES REACTIVITY: Human, Mouse, Rat Antibody produced in rabbits immunized with a synthetic peptide corresponding a region IMMUNOGEN: of human NFE2L1. TESTED APPLICATIONS: ELISA, WB NFE2L1 antibody can be used for detection of NFE2L1 by ELISA at 1:7862500. NFE2L1 APPLICATIONS: antibody can be used for detection of NFE2L1 by western blot at 1 μg/mL, and HRP conjugated secondary antibody should be diluted 1:50,000 - 100,000. POSITIVE CONTROL: 1) Cat. No. 1205 - Jurkat Cell Lysate PREDICTED MOLECULAR 85 kDa WEIGHT: Properties PURIFICATION: Antibody is purified by peptide affinity chromatography method. CLONALITY: Polyclonal CONJUGATE: Unconjugated PHYSICAL STATE: Liquid September 27, 2021 1 https://www.prosci-inc.com/nfe2l1-antibody-31-335.html Purified antibody supplied in 1x PBS buffer with 0.09% (w/v) sodium azide and 2% BUFFER: sucrose. CONCENTRATION: batch dependent For short periods of storage (days) store at 4˚C. For longer periods of storage, store STORAGE CONDITIONS: NFE2L1 antibody at -20˚C. As with any antibody avoid repeat freeze-thaw cycles. Additional Info OFFICIAL SYMBOL: NFE2L1 ALTERNATE NAMES: NFE2L1, FLJ00380, LCR-F1, NRF1, TCF11 ACCESSION NO.: NP_003195 PROTEIN GI NO.: 4505379 GENE ID: 4779 USER NOTE: Optimal dilutions for each application to be determined by the researcher. Background and References NFE2L1 activates erythroid-specific, globin gene expression. This gene encodes a protein that is involved in globin gene expression in erythrocytes. Confusion has occurred in bibliographic databases due to the shared symbol of NRF1 for this gene, NFE2L1, and for BACKGROUND: 'nuclear respiratory factor 1' which has an official symbol of NRF1. -

Human Small Maf Proteins Form Heterodimers with CNC Family Transcription Factors and Recognize the NF-E2 Motif
Oncogene (1997) 14, 1901 ± 1910 1997 Stockton Press All rights reserved 0950 ± 9232/97 $12.00 Human small Maf proteins form heterodimers with CNC family transcription factors and recognize the NF-E2 motif Tsutomu Toki1, Jugou Itoh2, Jun'ichi Kitazawa1, Koji Arai1, Koki Hatakeyama3, Jun-itsu Akasaka4, Kazuhiko Igarashi5, Nobuo Nomura6, Masaru Yokoyama1, Masayuki Yamamoto5 and Etsuro Ito1 1Department of Pediatrics, 2Medicine, School of Medicine; 3Department of Biology, Faculty of Sciences, Hirosaki University, Hirosaki 036; 4Department of Biochemistry, Tohoku University School of Medicine, Sendai 980-77; 5Center for TARA and Institute of Basic Medical Sciences, University of Tsukuba, Tsukuba 305; 6Kazusa DNA Institute, Kisarazu 292, Japan The transcription factor NF-E2, a heterodimeric protein Talbot et al., 1990; Talbot and Grosveld, 1991; complex composed of p45 and small Maf family Kotkow and Orkin, 1995). Recent analyses demon- proteins, is considered crucial for the regulation of strated that NF-E2 is composed of two subunits erythroid gene expression and platelet formation. To (Andrews et al., 1993a,b; Igarashi et al., 1994). The facilitate the characterization of NF-E2 functions in large p45 subunit belongs to a family of basic leucine- human cells, we isolated cDNAs encoding two members zipper (bZip) proteins that is closely related to the of the small Maf family, MafK and MafG. The human Drosophila Cap`n'colar (the CNC family) factor mafK and mafG genes encode proteins of 156 and 162 (Mohler et al., 1991). It cannot bind to the NF-E2 amino acid residues, respectively, whose deduced amino sequence as a homodimer, but does do after forming acid sequences show approximately 95% identity to their heterodimers with chicken small Maf family proteins, respective chicken counterparts. -

Distinct Roles of Jun : Fos and Jun : ATF Dimers in Oncogenesis
Oncogene (2001) 20, 2453 ± 2464 ã 2001 Nature Publishing Group All rights reserved 0950 ± 9232/01 $15.00 www.nature.com/onc Distinct roles of Jun : Fos and Jun : ATF dimers in oncogenesis Hans van Dam*,1 and Marc Castellazzi2 1Department of Molecular Cell Biology, Leiden University Medical Center, Sylvius Laboratories, PO Box 9503, 2300 RA Leiden, The Netherlands; 2Unite de Virologie Humaine, Institut National de la Sante et de la Recherche MeÂdicale (INSERM-U412), Ecole Normale SupeÂrieure, 46 alleÂe d'Italie, 69364 Lyon Cedex 07, France Jun : Fos and Jun : ATF complexes represent two classes dimers with emphasis on their roles in oncogenic of AP-1 dimers that (1) preferentially bind to either transformation in avian model systems. Previous heptameric or octameric AP-1 binding sites, and (2) are reviews on AP-1 and cell transformation include dierently regulated by cellular signaling pathways and references: (Angel and Karin, 1991; Wisdom, 1999; oncogene products. To discriminate between the func- Vogt, 1994; Karin et al., 1997; van Dam and van der tions of Jun : Fos, Jun: ATF and Jun : Jun, mutants were Eb, 1994; Hagmeyer et al., 1995). developed that restrict the ability of Jun to dimerize either to itself, or to Fos(-like) or ATF(-like) partners. Introduction of these mutants in chicken embryo Jun : Fos and Jun : ATF transcription factors: dimeric ®broblasts shows that Jun : Fra2 and Jun : ATF2 dimers complexes with variable composition and activities play distinct, complementary roles in in vitro oncogenesis by inducing either anchorage independence or growth AP-1 sub-units: members of the bZip protein family factor independence, respectively. -

Maff Is an Antiviral Host Factor That Suppresses Transcription from Hepatitis B Virus Core Promoter
bioRxiv preprint doi: https://doi.org/10.1101/2020.07.29.227793; this version posted March 24, 2021. The copyright holder for this preprint (which was not certified by peer review) is the author/funder. All rights reserved. No reuse allowed without permission. 1 Title 2 MafF is an antiviral host factor that suppresses transcription from Hepatitis B Virus 3 core promoter 4 Running title 5 MafF restricts viral replication 6 Authors 7 Marwa K. Ibrahim1,2, Tawfeek H. Abdelhafez1,2, Junko S. Takeuchi1,3,4, Kosho Wakae1, 8 Masaya Sugiyama5, Masataka Tsuge6, Masahiko Ito7, Koichi Watashi1, Mohamed El Kassas8, 9 Takanobu Kato1, Asako Murayama1, Tetsuro Suzuki7, Kazuaki Chayama9, Kunitada 10 Shimotohno10, Masamichi Muramatsu1#, Hussein H. Aly1#, Takaji Wakita1 11 Affiliation 12 1Department of Virology II, National Institute of Infectious Diseases, 1-23-1 Toyama, Shinjuku-ku, 13 Tokyo 162-8640, Japan 14 2Department of Microbial Biotechnology, Division of Genetic Engineering and Biotechnology 15 Research, National Research Centre, 33 EL Bohouth St. (formerly El Tahrir St.), Dokki, Giza, P. O. 16 12622, Egypt 17 3Center for Clinical Sciences, National Center for Global Health and Medicine, 1-21-1 18 Toyama Shinjuku-ku, Tokyo 162-8655, Japan 19 4Organization for the Strategic Coordination of Research and Intellectual Properties, Meiji 20 University, 1-1-1, Higashi-Mita, Tama-ku, Kawasaki, Kanagawa 214-8571, Japan 21 5Genome Medical Sciences Project, National Center for Global Health and Medicine, 1-7-1 Kohnodai, 22 Ichikawa, Chiba, 272-8516, Japan 23 6Department of Gastroenterology and Metabolism, Graduate School of Biomedical and Health 24 Science, Hiroshima University, 1-2-3 Kasumi, Minami-ku, Hiroshima, 734-8551, Japan 25 7Department of Virology and Parasitology, Hamamatsu University School of Medicine, 1-20-1 26 Handayama, Higashi-ku, Hamamatsu, Shizuoka, 431-3192, Japan 27 8Endemic Medicine Department, Faculty of Medicine, Helwan University, Cairo, P. -
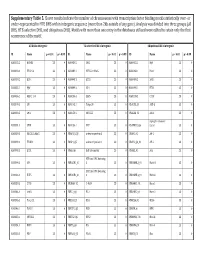
Supplementary Table 5. Clover Results Indicate the Number Of
Supplementary Table 5. Clover results indicate the number of chromosomes with transcription factor binding motifs statistically over‐ or under‐represented in HTE DHS within intergenic sequence (more than 2kb outside of any gene). Analysis was divided into three groups (all DHS, HTE‐selective DHS, and ubiquitous DHS). Motifs with more than one entry in the databases utilized were edited to retain only the first occurrence of the motif. All DHS x Intergenic TEselective DHS x Intergenic Ubiquitous DHS x Intergenic ID Name p < 0.01 p > 0.99 ID Name p < 0.01 p > 0.99 ID Name p < 0.01 p > 0.99 MA0002.2 RUNX1 23 0 MA0080.2 SPI1 23 0 MA0055.1 Myf 23 0 MA0003.1 TFAP2A 23 0 MA0089.1 NFE2L1::MafG 23 0 MA0068.1 Pax4 23 0 MA0039.2 Klf4 23 0 MA0098.1 ETS1 23 0 MA0080.2 SPI1 23 0 MA0055.1 Myf 23 0 MA0099.2 AP1 23 0 MA0098.1 ETS1 23 0 MA0056.1 MZF1_1‐4 23 0 MA0136.1 ELF5 23 0 MA0139.1 CTCF 23 0 MA0079.2 SP1 23 0 MA0145.1 Tcfcp2l1 23 0 V$ALX3_01 ALX‐3 23 0 MA0080.2 SPI1 23 0 MA0150.1 NFE2L2 23 0 V$ALX4_02 Alx‐4 23 0 myocyte enhancer MA0081.1 SPIB 23 0 MA0156.1 FEV 23 0 V$AMEF2_Q6 factor 23 0 MA0089.1 NFE2L1::MafG 23 0 V$AP1FJ_Q2 activator protein 1 23 0 V$AP1_01 AP‐1 23 0 MA0090.1 TEAD1 23 0 V$AP4_Q5 activator protein 4 23 0 V$AP2_Q6_01 AP‐2 23 0 MA0098.1 ETS1 23 0 V$AR_Q6 half‐site matrix 23 0 V$ARX_01 Arx 23 0 BTB and CNC homolog MA0099.2 AP1 23 0 V$BACH1_01 1 23 0 V$BARHL1_01 Barhl‐1 23 0 BTB and CNC homolog MA0136.1 ELF5 23 0 V$BACH2_01 2 23 0 V$BARHL2_01 Barhl2 23 0 MA0139.1 CTCF 23 0 V$CMAF_02 C‐MAF 23 0 V$BARX1_01 Barx1 23 0 MA0144.1 Stat3 23 0 -

The Expression of Genes Contributing to Pancreatic Adenocarcinoma Progression Is Influenced by the Respective Environment – Sagini Et Al
The expression of genes contributing to pancreatic adenocarcinoma progression is influenced by the respective environment – Sagini et al Supplementary Figure 1: Target genes regulated by TGM2. Figure represents 24 genes regulated by TGM2, which were obtained from Ingenuity Pathway Analysis. As indicated, 9 genes (marked red) are down-regulated by TGM2. On the contrary, 15 genes (marked red) are up-regulated by TGM2. Supplementary Table 1: Functional annotations of genes from Suit2-007 cells growing in pancreatic environment Categoriesa Diseases or p-Valuec Predicted Activation Number of genesf Functions activationd Z-scoree Annotationb Cell movement Cell movement 1,56E-11 increased 2,199 LAMB3, CEACAM6, CCL20, AGR2, MUC1, CXCL1, LAMA3, LCN2, COL17A1, CXCL8, AIF1, MMP7, CEMIP, JUP, SOD2, S100A4, PDGFA, NDRG1, SGK1, IGFBP3, DDR1, IL1A, CDKN1A, NREP, SEMA3E SERPINA3, SDC4, ALPP, CX3CL1, NFKBIA, ANXA3, CDH1, CDCP1, CRYAB, TUBB2B, FOXQ1, SLPI, F3, GRINA, ITGA2, ARPIN/C15orf38- AP3S2, SPTLC1, IL10, TSC22D3, LAMC2, TCAF1, CDH3, MX1, LEP, ZC3H12A, PMP22, IL32, FAM83H, EFNA1, PATJ, CEBPB, SERPINA5, PTK6, EPHB6, JUND, TNFSF14, ERBB3, TNFRSF25, FCAR, CXCL16, HLA-A, CEACAM1, FAT1, AHR, CSF2RA, CLDN7, MAPK13, FERMT1, TCAF2, MST1R, CD99, PTP4A2, PHLDA1, DEFB1, RHOB, TNFSF15, CD44, CSF2, SERPINB5, TGM2, SRC, ITGA6, TNC, HNRNPA2B1, RHOD, SKI, KISS1, TACSTD2, GNAI2, CXCL2, NFKB2, TAGLN2, TNF, CD74, PTPRK, STAT3, ARHGAP21, VEGFA, MYH9, SAA1, F11R, PDCD4, IQGAP1, DCN, MAPK8IP3, STC1, ADAM15, LTBP2, HOOK1, CST3, EPHA1, TIMP2, LPAR2, CORO1A, CLDN3, MYO1C, -

Micrornas As Modulators of Smoking-Induced Gene Expression Changes in Human Airway Epithelium
MicroRNAs as modulators of smoking-induced gene expression changes in human airway epithelium Frank Schembria,1, Sriram Sridharb,1, Catalina Perdomoc, Adam M. Gustafsond, Xiaoling Zhangd, Ayla Ergune, Jining Lua, Gang Liua, Xiaohui Zhanga, Jessica Bowersf, Cyrus Vazirib, Kristen Ottc, Kelly Sensingerf, James J. Collinse, Jerome S. Brodya, Robert Gettsf, Marc E. Lenburga,b,d, and Avrum Spiraa,b,d,2 aPulmonary Center, bDepartment of Pathology and Laboratory Medicine, and cGenetics and Genomics Graduate Program, Boston University School of Medicine, Boston, MA 02118; dBioinformatics Program, Boston University School of Engineering, Boston, MA 02215; eDepartment of Biomedical Engineering and Center for BioDynamics and Howard Hughes Medical Institute, Boston University, Boston, MA 02215; and fGenisphere, Inc., Hatfield, PA 19440 Edited by Charles R. Cantor, Sequenom Inc., San Diego, CA, and approved November 21, 2008 (received for review July 3, 2008) We have shown that smoking impacts bronchial airway gene Hundreds of human miRNAs have been cloned (11), with each of expression and that heterogeneity in this response associates with them predicted to regulate hundreds of genes by complementary smoking-related disease risk. In this study, we sought to determine binding to the 3Ј-untranslated region (UTR) of target mRNAs (12). whether microRNAs (miRNAs) play a role in regulating the airway A number of miRNAs have been shown to play important roles in gene expression response to smoking. We examined whole-ge- developmental timing, cell death, carcinogenesis, and stress responses in nome miRNA and mRNA expression in bronchial airway epithelium vitro (13–16), such as hypoxia (17). Whereas miRNA expression has and found 28 miRNAs to been examined in lung tumors (18–24), neither the expression of (20 ؍ from current and never smokers (n be differentially expressed (P < 0.05) with the majority being miRNA in bronchial epithelium nor the effect of smoking on bronchial down-regulated in smokers. -
Reprogramming of Lysosomal Gene Expression by Interleukin-4 and Stat6 Brignull Et Al
Reprogramming of lysosomal gene expression by interleukin-4 and Stat6 Brignull et al. Brignull et al. BMC Genomics 2013, 14:853 http://www.biomedcentral.com/1471-2164/14/853 Brignull et al. BMC Genomics 2013, 14:853 http://www.biomedcentral.com/1471-2164/14/853 RESEARCH ARTICLE Open Access Reprogramming of lysosomal gene expression by interleukin-4 and Stat6 Louise M Brignull1†, Zsolt Czimmerer2†, Hafida Saidi1,3, Bence Daniel2, Izabel Villela4,5, Nathan W Bartlett6, Sebastian L Johnston6, Lisiane B Meira4, Laszlo Nagy2,7 and Axel Nohturfft1* Abstract Background: Lysosomes play important roles in multiple aspects of physiology, but the problem of how the transcription of lysosomal genes is coordinated remains incompletely understood. The goal of this study was to illuminate the physiological contexts in which lysosomal genes are coordinately regulated and to identify transcription factors involved in this control. Results: As transcription factors and their target genes are often co-regulated, we performed meta-analyses of array-based expression data to identify regulators whose mRNA profiles are highly correlated with those of a core set of lysosomal genes. Among the ~50 transcription factors that rank highest by this measure, 65% are involved in differentiation or development, and 22% have been implicated in interferon signaling. The most strongly correlated candidate was Stat6, a factor commonly activated by interleukin-4 (IL-4) or IL-13. Publicly available chromatin immunoprecipitation (ChIP) data from alternatively activated mouse macrophages show that lysosomal genes are overrepresented among Stat6-bound targets. Quantification of RNA from wild-type and Stat6-deficient cells indicates that Stat6 promotes the expression of over 100 lysosomal genes, including hydrolases, subunits of the vacuolar H+ ATPase and trafficking factors. -

Identification of Dna Sequence Variants in the Estrogen
IDENTIFICATION OF DNA SEQUENCE VARIANTS IN THE ESTROGEN RECEPTOR PATHWAY IN BREAST CANCER by Seyed Amir Bahreini BSc in Cell & Mol Biology, University of Isfahan, Iran, 2010 Submitted to the Graduate Faculty of Department of Human Genetics Graduate School of Public Health in partial fulfillment of the requirements for the degree of Doctor of Philosophy University of Pittsburgh 2016 UNIVERSITY OF PITTSBURGH Graduate School of Public Health This dissertation was presented by Seyed Amir Bahreini It was defended on April 6, 2016 and approved by Dissertation Advisor: Steffi Oesterreich, PhD Professor Department of Pharmacology and Chemical Biology School of Medicine University of Pittsburgh Committee Members: Candace M. Kammerer, PhD Professor Department of Human Genetics Graduate School of Public Health University of Pittsburgh Adrian V. Lee, PhD Professor Department of Pharmacology and Chemical Biology School of Medicine University of Pittsburgh Ryan L. Minster, PhD Professor Department of Human Genetics Graduate School of Public Health University of Pittsburgh ii Copyright © by Seyed Amir Bahreini 2016 iii Steffi Oesterreich, PhD IDENTIFICATION OF DNA SEQUENCE VARIANTS IN THE ESTROGEN RECEPTOR PATHWAY IN BREAST CANCER Seyed Amir Bahreini, PhD University of Pittsburgh, 2016 ABSTRACT Breast cancer is of public health importance with an increasing incidence over the past decade. Estrogen Receptor (ER) activity is critical for promoting majority of breast cancers. Inhibiting ER is one of the most successful targeted therapies in oncology. Studies have suggested that genomic variation in ER binding sites and ESR1 gene may be responsible for endocrine treatment response and cancer progression. We investigated the role of single nucleotide variants (SNVs) in the ER pathway in breast cancer, including clinically relevant mutations in ER gene and regulatory variants in ER binding sites.