Ethinyl Estradiol and Norgestimate
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

ORTHO TRI-CYCLEN® TABLETS ORTHO-CYCLEN® TABLETS (Norgestimate/Ethinyl Estradiol)
PHYSICIANS' PACKAGE INSERT ORTHO TRI-CYCLEN® TABLETS ORTHO-CYCLEN® TABLETS (norgestimate/ethinyl estradiol) Patients should be counseled that this product does not protect against HIV infection (AIDS) and other sexually transmitted diseases. DESCRIPTION Each of the following products is a combination oral contraceptive containing the progestational compound norgestimate and the estrogenic compound ethinyl estradiol. ORTHO TRI-CYCLEN 21 Tablets and ORTHO TRI-CYCLEN 28 Tablets. Each white tablet contains 0.180 mg of the progestational compound, norgestimate (18,19-Dinor- 17-pregn-4-en-20-yn-3-one,17-(acetyloxy)-13-ethyl-, oxime,(17α)-(+)-) and 0.035 mg of the estrogenic compound, ethinyl estradiol (19-nor-17α-pregna,1,3,5(10)-trien-20-yne-3,17-diol). Inactive ingredients include lactose, magnesium stearate, and pregelatinized starch. Each light blue tablet contains 0.215 mg of the progestational compound norgestimate (18,19- Dinor-17-pregn-4-en-20-yn-3-one,17-(acetyloxy)-13-ethyl-,oxime,(17α)-(+)-) and 0.035 mg of the estrogenic compound, ethinyl estradiol (19-nor-17α-pregna,1,3,5(10)-trien-20-yne-3,17-diol). Inactive ingredients include FD & C Blue No. 2 Aluminum Lake, lactose, magnesium stearate, and pregelatinized starch. Each blue tablet contains 0.250 mg of the progestational compound norgestimate (18,19-Dinor-17- pregn-4-en-20-yn-3-one, 17-(acetyloxy)-13-ethyl-,oxime,(17α)-(+)-) and 0.035 mg of the estrogenic compound, ethinyl estradiol (19-nor-17α-pregna,1,3,5(10)-trien-20-yne-3,17-diol). Inactive ingredients include FD & C Blue No. 2 Aluminum Lake, lactose, magnesium stearate, and pregelatinized starch. -

Estrogen and Progestin Hormone Doses in Combined Birth Control Pills
Estrogen and Progestin Hormone Doses in Combined Birth Control Pills Estrogen level Pill Brand Name Progestin Dose (mg) ethinyl estradiol (micrograms) 20 mcgm Alesse® levonorgestrel 0.10 Levlite® levonorgestrel 0.10 Loestrin 1/20® Fe norethindrone 1.00 acetate Mircette® desogestrel 0.15 Ortho Evra® norelgestromin 0.15 (patch) (norgestimate metabolite) phasic Estrostep® Fe norethindrone 1.0/1.0/1.0 20/30/35 mcgm acetate 30 mcgm Levlen® levonorgestrel 0.15 Levora® levonorgestrel 0.15 Nordette® levonorgestrel 0.15 Lo/Ovral® norgestrel 0.30 Desogen® desogestrel 0.15 Ortho-Cept® desogestrel 0.15 Loestrin® 1.5/30 norethindrone 1.50 acetate Yasmin® drospirenone 3.0 phasic Triphasil® levonorgestrel 0.05/0.075/0.125 30/40/30 mcgm Tri-Levlen® levonorgestrel 0.05/0.075/0.125 Trivora® levonorgestrel 0.05/0.075/0.125 35 mcgm Ortho-Cyclen® norgestimate 0.25 Ovcon-35® norethindrone 0.40 Brevicon® norethindrone 0.50 Modicon® norethindrone 0.50 Necon® norethindrone 1.00 Norethin® norethindrone 1.00 Norinyl® 1/35 norethindrone 1.00 Ortho-Novum® 1/35 norethindrone 1.00 Demulen® 1/35 ethynodiol diacetate 1.00 Zovia® 1/35E ethynodiol diacetate 1.00 phasic Ortho-Novum® norethindrone 0.50/1.00 35/35 mcgm 10/11 Jenest® norethindrone 0.50/1.00 phasic Ortho-Tri-Cyclen® norgestimate 0.15/0.215/0.25 35/35/35 mcgm Ortho-Novum® norethindrone 0.50/0.75/1.00 7/7/7 Tri-Norinyl® norethindrone 0.50/1.00/0.50 50 mcgm Necon® 1/50 norethindrone 1.00 Norinyl® 1/50 norethindrone 1.00 Ortho-Novum® 1/50 norethindrone 1.00 Ovcon-50® norethindrone 1.00 Ovral® norgestrel 0.50 Demulen® 1/50 ethynodiol diacetate 1.00 Zovia® 1/50E ethynodiol diacetate 1.00 Which pills have higher progestin side efects or cause more acne and hair growth? Each progestin has a diferent potency, milligram per milligram, in terms of progesterone efect to stop menstrual bleeding or androgen efect to stimulate acne and hair growth. -

Report 752 by Category Description
PAs by Category Description Sorted by Descending Count Date Range: 04/01/2006 through 06/30/2006 Number Number Category of Category of Description PAs Description PAs Cetirizine HCl 792 Ziprasidone HCl 43 Duloxetine HCl 784 Norelgestromin-Ethinyl Estradiol 42 Methylphenidate HCl 646 Nicotine 41 Venlafaxine HCl 620 Levofloxacin 41 Atomoxetine HCl 472 Carisoprodol 41 Quetiapine Fumarate 430 Albuterol 40 Gabapentin 422 Amylase-Lipase-Protease 40 Nutritional Supplements 378 Famotidine 40 Montelukast Sodium 326 Levothyroxine Sodium 39 Zolpidem Tartrate 288 Enoxaparin Sodium 37 Amphetamine-Dextroamphetamine 286 Norgestimate-Ethinyl Estradiol (Triphasic) 37 Aripiprazole 271 Tretinoin 37 Desloratadine 191 Modafinil 36 Fexofenadine HCl 188 Pioglitazone HCl 36 Topiramate 186 Citalopram Hydrobromide 36 Polyethylene Glycol 3350 182 Budesonide (Inhalation) 36 Fentanyl 175 Epoetin Alfa 33 Eszopiclone 174 Etanercept 33 Esomeprazole Magnesium 172 Botulinum Toxin Type A 32 Celecoxib 157 Somatropin 31 Pregabalin 148 Metformin HCl 31 Escitalopram Oxalate 142 Oxycodone w/ Acetaminophen 31 Sertraline HCl 135 Morphine Sulfate 30 Risperidone 127 Levetiracetam 30 Bupropion HCl 118 Clonazepam 30 Tiotropium Bromide Monohydrate 112 Phenobarbital 30 Oxycodone HCl 110 Drospirenone-Ethinyl Estradiol 29 Ezetimibe 105 Rosiglitazone Maleate 29 Clopidogrel Bisulfate 103 Valsartan 29 Ondansetron HCl 97 Memantine HCl 28 Olanzapine 93 Sumatriptan Succinate 28 Temazepam 92 Buprenorphine HCl 28 Oxcarbazepine 82 B-Complex w/ C & Folic Acid 28 Rabeprazole Sodium 74 Ranitidine -

Oral Contraceptives
ORAL CONTRACEPTIVES STRENGTH DRUG ESTROGEN PROGESTIN (ESTROGEN/PROGESTIN) MONOPHASIC Aviane 28, Lessina, Lutera, Orsythia, Ethinyl estradiol Levonorgestrel 20mcg/0.1mg Sronyx† Beyaz*, YAZ Ethinyl estradiol Drospirenone 20mcg/3mg [Gianvi, Loryna, Vestura]† Brevicon, Modicon Ethinyl estradiol Norethindrone 35mcg/0.5mg [Necon 0.5/35, Nortrel 0.5/35]† Desogen Ethinyl estradiol Desogestrel 30mcg/0.15mg [Apri, Emoquette, Reclipsen, Solia]† Femcon Fe, Ovcon 35 Ethinyl estradiol Norethindrone 35mcg/0.4mg [Balziva, Briellyn, Philith, Vyfemla Zenchent, Zenchent Fe]† Loestrin 21 1/20, Loestrin Fe 1/20, Ethinyl estradiol Norethindrone acetate 20mcg/1mg Loestrin 24 Fe, Minastrin 24 Fe [Gildess Fe 1/20, Junel 1/20, Junel Fe 1/20, Larin 1/20, Larin Fe 1/20, Microgestin 1/20, Microgestin Fe 1/20]† Loestrin 21 1.5/30, Loestrin Fe 1.5/30 Ethinyl estradiol Norethindrone acetate 30mcg/1.5mg [Gildess Fe 1.5/30, Junel 1.5/30, Junel Fe 1.5/30, Microgestin 1.5/30, Microgestin Fe 1.5/30]† Lo/Ovral Ethinyl estradiol Norgestrel 30mcg/0.3mg [Cryselle, Low-Ogestrel]† Lybrel Ethinyl estradiol Levonorgestrel 20mcg/0.09mg [Amethyst]† [Altavera, Levora, Marlissa, Portia]† Ethinyl estradiol Levonorgestrel 30mcg/0.15mg Norinyl 1/35, Ortho-Novum 1/35 Ethinyl estradiol Norethindrone 35mcg/1mg [Alyacen 1/35, Cyclafem 1/35, Dasetta 1/35, Necon 1/35, Nortrel 1/35]† Necon 1/50, Norinyl 1/50 Mestranol Norethindrone 50mcg/1mg Ogestrel 0.5/50† Ethinyl estradiol Norgestrel 50mcg/0.5mg Ortho-Cyclen Ethinyl estradiol Norgestimate 35mcg/0.25mg [MonoNessa, Previfem, Sprintec]† Ovcon -

EUROPEAN PHARMACOPOEIA 10.0 Index 1. General Notices
EUROPEAN PHARMACOPOEIA 10.0 Index 1. General notices......................................................................... 3 2.2.66. Detection and measurement of radioactivity........... 119 2.1. Apparatus ............................................................................. 15 2.2.7. Optical rotation................................................................ 26 2.1.1. Droppers ........................................................................... 15 2.2.8. Viscosity ............................................................................ 27 2.1.2. Comparative table of porosity of sintered-glass filters.. 15 2.2.9. Capillary viscometer method ......................................... 27 2.1.3. Ultraviolet ray lamps for analytical purposes............... 15 2.3. Identification...................................................................... 129 2.1.4. Sieves ................................................................................. 16 2.3.1. Identification reactions of ions and functional 2.1.5. Tubes for comparative tests ............................................ 17 groups ...................................................................................... 129 2.1.6. Gas detector tubes............................................................ 17 2.3.2. Identification of fatty oils by thin-layer 2.2. Physical and physico-chemical methods.......................... 21 chromatography...................................................................... 132 2.2.1. Clarity and degree of opalescence of -
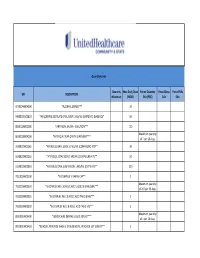
UHC PA Community and State Quantity Limit List
Quantity Limits Quantity Max Daily Dose Period Quantity Period Days Period Fills GPI DESCRIPTION Maximum (MDD) Edit (PQE) Edit Edit 97703040004300 *ALCOHOL SWABS*** 10 34000003101810 *AMLODIPINE BESYLATE ORAL SUSP 1 MG/ML (CMPD KIT) (BASE EQ)* 10 88501000002000 *ARTIFICIAL SALIVA - SOLUTION*** 120 Maximum quantity 86202000004200 *ARTIFICIAL TEAR OPHTH OINTMENT*** of 7 per 26 days. 33200020002065 *ATENOLOL ORAL SOLN 10 MG/ML (COMPOUND KIT)** 20 33200020002055 *ATENOLOL ORAL SOLN 2 MG/ML (COMPOUND KIT)** 50 33200020001810 *ATENOLOL ORAL SUSPENSION 1 MG/ML (CMPD KIT)** 200 78110000000100 *B-COMPLEX VITAMIN CAP** 1 Maximum quantity 78133000000920 *B-COMPLEX W/ C & FOLIC ACID LIQUID 0.9 MG/5ML*** of 237 per 26 days. 78133000000325 *B-COMPLEX W/ C & FOLIC ACID TAB 0.8 MG*** 1 78133000000330 *B-COMPLEX W/ C & FOLIC ACID TAB 1 MG*** 1 Maximum quantity 88350010006400 *BENZOCAINE DENTAL LIQUID 20% KIT*** of 1 per 26 days. 90050010006410 *BENZOYL PEROXIDE WASH 2.5% & BENZOYL PEROXIDE LOT 10% KIT** 1 47300005100100 *BIFIDOBACTERIUM BIFIDUM CAP** 2 97202007100900 *BLOOD GLUCOSE CALIBRATION - LIQUID*** 0.04 Maximum quantity 97202011006200 *BLOOD GLUCOSE METER DISPOSABLE DEVICE WITH TEST STRIPS*** of 1 per 999 days. Maximum quantity 97202010006200 *BLOOD GLUCOSE MONITORING DEVICES*** of 1 per 365 days. Maximum quantity 97202010006410 *BLOOD GLUCOSE MONITORING KIT W/ DEVICE*** of 1 per 365 days. Maximum quantity 97202010006400 *BLOOD GLUCOSE MONITORING KIT*** of 1 per 365 days. Maximum quantity 97750010006200 *BLOOD PRESSURE MONITORING - DEVICE*** of 1 per 999 days. Maximum quantity 9025990212B120 *CALCIPOTRIENE CR 0.005% & DIMETHICONE CR 5% THERAPY PACK*** of 1 per 26 days. 79109903450340 *CALCIUM CARB-VIT D W/ MINERALS TABS 600 MG-200 UNIT*** 5 79109903450355 *CALCIUM CARB-VIT D W/ MINERALS TABS 600 MG-800 UNIT*** 2 Maximum quantity 6610990420B120 *CELECOXIB CAP 200 MG & METH SAL-MEN-CAPSAICIN LIQD THER PK* of 1 per 26 days. -

PRAC Recommendations on Signals October 2018
29 October 20181 EMA/PRAC/689235/2018 Pharmacovigilance Risk Assessment Committee (PRAC) PRAC recommendations on signals Adopted at the 1-4 October 2018 PRAC meeting This document provides an overview of the recommendations adopted by the Pharmacovigilance Risk Assessment Committee (PRAC) on the signals discussed during the meeting of 1-4 October 2018 (including the signal European Pharmacovigilance Issues Tracking Tool [EPITT]2 reference numbers). PRAC recommendations to provide supplementary information are directly actionable by the concerned marketing authorisation holders (MAHs). PRAC recommendations for regulatory action (e.g. amendment of the product information) are submitted to the Committee for Medicinal Products for Human Use (CHMP) for endorsement when the signal concerns Centrally Authorised Products (CAPs), and to the Co-ordination Group for Mutual Recognition and Decentralised Procedures – Human (CMDh) for information in the case of Nationally Authorised Products (NAPs). Thereafter, MAHs are expected to take action according to the PRAC recommendations. When appropriate, the PRAC may also recommend the conduct of additional analyses by the Agency or Member States. MAHs are reminded that in line with Article 16(3) of Regulation No (EU) 726/2004 and Article 23(3) of Directive 2001/83/EC, they shall ensure that their product information is kept up to date with the current scientific knowledge including the conclusions of the assessment and recommendations published on the European Medicines Agency (EMA) website (currently acting as the EU medicines webportal). For CAPs, at the time of publication, PRAC recommendations for update of product information have been agreed by the CHMP at their plenary meeting (17-20 October 2018) and corresponding variations will be assessed by the CHMP. -

Hormonal Contraceptives, Female Sexual Dysfunction, and Managing Strategies: a Review
Journal of Clinical Medicine Review Hormonal Contraceptives, Female Sexual Dysfunction, and Managing Strategies: A Review Nerea M. Casado-Espada 1, Rubén de Alarcón 1, Javier I. de la Iglesia-Larrad 1, Berta Bote-Bonaechea 1 and Ángel L. Montejo 1,2,* 1 Psychiatry Service, Institute of Biomedical Research of Salamanca (IBSAL), University Clinical Hospital of Salamanca, Paseo San Vicente, SN 37007 Salamanca, Spain; [email protected] (N.M.C.-E.); [email protected] (R.d.A.); [email protected] (J.I.d.l.I.-L.); [email protected] (B.B.-B.) 2 Nursing School E.U.E.F., University of Salamanca, Av. Donantes de Sangre SN 37007 Salamanca, Spain * Correspondence: [email protected]; Tel.:+34639754620 Received: 12 May 2019; Accepted: 24 June 2019; Published: 25 June 2019 Abstract: In recent decades, hormonal contraceptives (HC) has made a difference in the control of female fertility, taking an unequivocal role in improving contraceptive efficacy. Some side effects of hormonal treatments have been carefully studied. However, the influence of these drugs on female sexual functioning is not so clear, although variations in the plasma levels of sexual hormones could be associated with sexual dysfunction. Permanent hormonal modifications, during menopause or caused by some endocrine pathologies, could be directly related to sexual dysfunction in some cases but not in all of them. HC use seems to be responsible for a decrease of circulating androgen, estradiol, and progesterone levels, as well as for the inhibition of oxytocin functioning. Hormonal contraceptive use could alter women’s pair-bonding behavior, reduce neural response to the expectation of erotic stimuli, and increase sexual jealousy. -

Androgens in Women: Hormone-Modulating Therapies For
Androgens in women Hormone-modulating therapies for skin disease Sarah Azarchi, BS,a Amanda Bienenfeld, BA,a Kristen Lo Sicco, MD,b ShariMarchbein,MD,b Jerry Shapiro, MD,b and Arielle R. Nagler, MDb New York, New York Learning objectives After completing this learning activity, participants should be able to list, categorize, and explain the mechanisms of action, safety considerations, and contraindications of these androgen-modulating therapies; identify the hormone-modulating therapies used to treat acne, hirsutism, and androgenetic alopecia; and describe the evidence to support the efficacy of these therapies in the treatment of acne, hirsutism, and androgenetic alopecia. Disclosures Editors The editors involved with this CME activity and all content validation/peer reviewers of the journal-based CME activity have reported no relevant financial relationships with commercial interest(s). Authors The authors involved with this journal-based CME activity have reported no relevant financial relationships with commercial interest(s). Dr Shapiro is a stockholder in Replicel Life Sciences and has been a consultant for Aclaris, Samumed, Incyte, Bioniz, keeps.com, Applied Biology, and Pfizer. Planners The planners involved with this journal-based CME activity have reported no relevant financial relationships with commercial interest(s). The editorial and education staff involved with this journal-based CME activity have reported no relevant financial relationships with commercial interest(s). Androgen-mediated cutaneous disorders (AMCDs) in women, including acne, hirsutism, and female pattern hair loss, can be treated with hormone-modulating therapies. In the second article in this Continuing Medical Education series, we discuss the hormone-modulating therapies available to dermatologists for the treatment of AMCDs, including combined oral contraceptives, spironolactone, finasteride, dutasteride, and flutamide. -

Final Research Report
PATIENT-CENTERED OUTCOMES RESEARCH INSTITUTE FINAL RESEARCH REPORT Examining Health Outcomes for People Who Are Transgender Michael Goodman, MD, MPH; Rebecca Nash, MPH AFFILIATION: Emory University School of Public Health, Atlanta, Georgia Original Project Title: Comparative Risks and Benefits of Gender Reassignment Therapies PCORI ID: AD-12-11-4532 HSRProj ID: 20143115 _______________________________ To cite this document, please use: Goodman M, Nash R. (2019). Examining Health Outcomes for People Who Are Transgender. Patient-Centered Outcomes Research Institute (PCORI). https://doi.org/10.25302/2.2019.AD.12114532 TABLE OF CONTENTS ABSTRACT .............................................................................................................................. 6 BACKGROUND ........................................................................................................................ 8 Transgender Health Issues and Knowledge Gaps ...................................................................... 8 Methodological Challenges Facing Transgender Health Studies ............................................. 10 Introducing Study of Transition, Outcomes, and Gender ........................................................ 11 Table 1. STRONG Project Timeline .................................................................................... 12 STAKEHOLDER INVOLVEMENT ............................................................................................... 13 Preliminary Survey .................................................................................................................. -

List of Formulary Drug Removals
July 2021 Formulary Drug Removals Below is a list of medicines by drug class that have been removed from your plan’s formulary. If you continue using one of the drugs listed below and identified as a Formulary Drug Removal, you may be required to pay the full cost. If you are currently using one of the formulary drug removals, ask your doctor to choose one of the generic or brand formulary options listed below. Category Formulary Drug Formulary Options Drug Class Removals Acromegaly SANDOSTATIN LAR SOMATULINE DEPOT SIGNIFOR LAR SOMAVERT Allergies dexchlorpheniramine levocetirizine Antihistamines Diphen Elixir RyClora CARBINOXAMINE TABLET 6 MG Allergies BECONASE AQ flunisolide spray, fluticasone spray, mometasone spray, DYMISTA Nasal Steroids / Combinations OMNARIS QNASL ZETONNA Anticonvulsants topiramate ext-rel capsule carbamazepine, carbamazepine ext-rel, clobazam, divalproex sodium, (generics for QUDEXY XR only) divalproex sodium ext-rel, gabapentin, lamotrigine, lamotrigine ext-rel, levetiracetam, levetiracetam ext-rel, oxcarbazepine, phenobarbital, phenytoin, phenytoin sodium extended, primidone, rufinamide, tiagabine, topiramate, valproic acid, zonisamide, FYCOMPA, OXTELLAR XR, TROKENDI XR, VIMPAT, XCOPRI BANZEL SUSPENSION clobazam, lamotrigine, rufinamide, topiramate, TROKENDI XR ONFI SABRIL vigabatrin ZONEGRAN carbamazepine, carbamazepine ext-rel, divalproex sodium, divalproex sodium ext-rel, gabapentin, lamotrigine, lamotrigine ext-rel, levetiracetam, levetiracetam ext-rel, oxcarbazepine, phenobarbital, phenytoin, phenytoin sodium -

Oral Contraceptive Identification Chart
Oral Contraceptive Identification Chart View the full Prescribing Information by clicking on the Teva Generic product name. Brand Teva Generic NDC Strength(s) Size Alesse®* Aviane® (levonorgestrel and ethinyl estradiol tablets, USP) 00555-9045-58 0.1 mg/0.02 mg 6 x 28 Blister Packs 0.1 mg/0.025 mg; 0.125 Velivet® (desogestrel and ethinyl estradiol tablets, USP - triphasic Cyclessa® 00555-9051-67 mg/0.025 mg; 0.15 3 x 28 Blister Packs regimen) mg/0.025 mg Kelnor® 1/35 (ethynodiol diacetate and ethinyl estradiol tablets, 00555-9064-58 1 mg/35 mcg 6 x 28 Blister Packs USP) Demulen®* Kelnor® 1/50 (ethynodiol diacetate and ethinyl estradiol tablets 00093-8073-16 1 mg/50 mcg 6 x 28 Blister Packs USP) 1 mg/20 mcg; Tri-Legest® Fe 28 (norethindrone acetate and ethinyl estradiol Estrostep® Fe 00555-9032-70 1 mg/30 mcg; 5 x 28 Blister Packs tablets, USP and ferrous fumarate tablets†) 1 mg/35 mcg Layolis® Fe (norethindrone and ethinyl estradiol chewable tablets Generess® Fe 52544-0064-31 0.8 mg/25 mcg 3 x 28 Blister Packs and ferrous fumarate chewable tablets†)‡ Levlite®* Lessina® (levonorgestrel and ethinyl estradiol tablets, USP) 00555-9014-67 0.1 mg/0.02 mg 3 x 28 Blister Packs Lo/Ovral®* Cryselle® (norgestrel and ethinyl estradiol tablets, USP) 00555-9049-58 0.3 mg/0.03 mg 6 x 28 Blister Packs Junel® 1/20 (norethindrone acetate and ethinyl estradiol tablets, 00555-9025-42 1 mg/20 mcg 3 x 21 Blister Packs USP) Loestrin® 21 Junel® 1.5/30 (norethindrone acetate and ethinyl estradiol tablets, 00555-9027-42 1.5 mg/30 mcg 3 x 21 Blister Packs USP)