Download PDF (Inglês)
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-
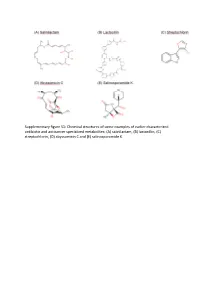
Chemical Structures of Some Examples of Earlier Characterized Antibiotic and Anticancer Specialized
Supplementary figure S1: Chemical structures of some examples of earlier characterized antibiotic and anticancer specialized metabolites: (A) salinilactam, (B) lactocillin, (C) streptochlorin, (D) abyssomicin C and (E) salinosporamide K. Figure S2. Heat map representing hierarchical classification of the SMGCs detected in all the metagenomes in the dataset. Table S1: The sampling locations of each of the sites in the dataset. Sample Sample Bio-project Site depth accession accession Samples Latitude Longitude Site description (m) number in SRA number in SRA AT0050m01B1-4C1 SRS598124 PRJNA193416 Atlantis II water column 50, 200, Water column AT0200m01C1-4D1 SRS598125 21°36'19.0" 38°12'09.0 700 and above the brine N "E (ATII 50, ATII 200, 1500 pool water layers AT0700m01C1-3D1 SRS598128 ATII 700, ATII 1500) AT1500m01B1-3C1 SRS598129 ATBRUCL SRS1029632 PRJNA193416 Atlantis II brine 21°36'19.0" 38°12'09.0 1996– Brine pool water ATBRLCL1-3 SRS1029579 (ATII UCL, ATII INF, N "E 2025 layers ATII LCL) ATBRINP SRS481323 PRJNA219363 ATIID-1a SRS1120041 PRJNA299097 ATIID-1b SRS1120130 ATIID-2 SRS1120133 2168 + Sea sediments Atlantis II - sediments 21°36'19.0" 38°12'09.0 ~3.5 core underlying ATII ATIID-3 SRS1120134 (ATII SDM) N "E length brine pool ATIID-4 SRS1120135 ATIID-5 SRS1120142 ATIID-6 SRS1120143 Discovery Deep brine DDBRINP SRS481325 PRJNA219363 21°17'11.0" 38°17'14.0 2026– Brine pool water N "E 2042 layers (DD INF, DD BR) DDBRINE DD-1 SRS1120158 PRJNA299097 DD-2 SRS1120203 DD-3 SRS1120205 Discovery Deep 2180 + Sea sediments sediments 21°17'11.0" -

The 2014 Golden Gate National Parks Bioblitz - Data Management and the Event Species List Achieving a Quality Dataset from a Large Scale Event
National Park Service U.S. Department of the Interior Natural Resource Stewardship and Science The 2014 Golden Gate National Parks BioBlitz - Data Management and the Event Species List Achieving a Quality Dataset from a Large Scale Event Natural Resource Report NPS/GOGA/NRR—2016/1147 ON THIS PAGE Photograph of BioBlitz participants conducting data entry into iNaturalist. Photograph courtesy of the National Park Service. ON THE COVER Photograph of BioBlitz participants collecting aquatic species data in the Presidio of San Francisco. Photograph courtesy of National Park Service. The 2014 Golden Gate National Parks BioBlitz - Data Management and the Event Species List Achieving a Quality Dataset from a Large Scale Event Natural Resource Report NPS/GOGA/NRR—2016/1147 Elizabeth Edson1, Michelle O’Herron1, Alison Forrestel2, Daniel George3 1Golden Gate Parks Conservancy Building 201 Fort Mason San Francisco, CA 94129 2National Park Service. Golden Gate National Recreation Area Fort Cronkhite, Bldg. 1061 Sausalito, CA 94965 3National Park Service. San Francisco Bay Area Network Inventory & Monitoring Program Manager Fort Cronkhite, Bldg. 1063 Sausalito, CA 94965 March 2016 U.S. Department of the Interior National Park Service Natural Resource Stewardship and Science Fort Collins, Colorado The National Park Service, Natural Resource Stewardship and Science office in Fort Collins, Colorado, publishes a range of reports that address natural resource topics. These reports are of interest and applicability to a broad audience in the National Park Service and others in natural resource management, including scientists, conservation and environmental constituencies, and the public. The Natural Resource Report Series is used to disseminate comprehensive information and analysis about natural resources and related topics concerning lands managed by the National Park Service. -

Methanogenic Activity in Río Tinto, a Terrestrial Mars Analogue R
Methanogenic activity in Río Tinto, a terrestrial Mars analogue R. Amils Centro de Biología Molecular Severo Ochoa (UAM-CSIC) y Centro de Astrobiología (INTA- CSIC) Frascati, noviembre 2009 new insides in the Mars exploration H2O on Mars methane (PFS) it can be concluded that on Mars there are sedimentary rocks that were formed in acidic conditions (acidic lakes or oceans) • possible terrestrial analogs: - submarine hydrothermalism - acidic environments to explore the deep sea requires expensive equipment (Alvin) natural acidic waters natural acidic environments: - areas with volcanic activity 0 SO2 + H2S ——> S + H2O - metal mining activities 3+ 2- + FeS2 + H2O —> Fe + SO4 + H in this case the extreme acidic conditions are promoted by biological activity geomicrobiology of metallic sulfides pyrite, molibdenite, tungstenite (thiosulfate mec.) 3+ 2- 2+ + FeS2+6Fe +3H2O → S2O3 +7Fe +6H 2- 3+ 2- 2+ + S2O3 +8Fe +5H2O → 2SO4 +8Fe +10H Rest of sulfides (polisulfide mec.) 3+ + 2+ 2+ 8MS+8Fe +8H → 8M +4H2Sn+8Fe (n≥2) 3+ o 2+ + 4H2Sn+8Fe → S8 +8Fe +8H o 2- + S8 +4H2O (S oxidizers) → SO4 +8H Bacterias come-meteoritos role of the microbial activity in the leaching of pyrite chemical 3+ reaction Fe Fe2+ microbial activity 2- + SO4 + H Rio Tinto rise at the heart of the Iberian Pyritic Belt Río Tinto is an acidic river, pH 2.3, 100 km long and with a high concentration of soluble metals the iron concentration at the origin is between 15-20 g/l and the sulfate is constant and around 15 g/l geoMICROBIOLOGY combination of conventional microbial ecology techniques and molecular ecology tools A B Phylogeny of acidophilic microorganisms detected in Rio Tinto Actinobacteria Cyanobacteria . -

Microbial Community Structure Dynamics in Ohio River Sediments During Reductive Dechlorination of Pcbs
University of Kentucky UKnowledge University of Kentucky Doctoral Dissertations Graduate School 2008 MICROBIAL COMMUNITY STRUCTURE DYNAMICS IN OHIO RIVER SEDIMENTS DURING REDUCTIVE DECHLORINATION OF PCBS Andres Enrique Nunez University of Kentucky Right click to open a feedback form in a new tab to let us know how this document benefits ou.y Recommended Citation Nunez, Andres Enrique, "MICROBIAL COMMUNITY STRUCTURE DYNAMICS IN OHIO RIVER SEDIMENTS DURING REDUCTIVE DECHLORINATION OF PCBS" (2008). University of Kentucky Doctoral Dissertations. 679. https://uknowledge.uky.edu/gradschool_diss/679 This Dissertation is brought to you for free and open access by the Graduate School at UKnowledge. It has been accepted for inclusion in University of Kentucky Doctoral Dissertations by an authorized administrator of UKnowledge. For more information, please contact [email protected]. ABSTRACT OF DISSERTATION Andres Enrique Nunez The Graduate School University of Kentucky 2008 MICROBIAL COMMUNITY STRUCTURE DYNAMICS IN OHIO RIVER SEDIMENTS DURING REDUCTIVE DECHLORINATION OF PCBS ABSTRACT OF DISSERTATION A dissertation submitted in partial fulfillment of the requirements for the degree of Doctor of Philosophy in the College of Agriculture at the University of Kentucky By Andres Enrique Nunez Director: Dr. Elisa M. D’Angelo Lexington, KY 2008 Copyright © Andres Enrique Nunez 2008 ABSTRACT OF DISSERTATION MICROBIAL COMMUNITY STRUCTURE DYNAMICS IN OHIO RIVER SEDIMENTS DURING REDUCTIVE DECHLORINATION OF PCBS The entire stretch of the Ohio River is under fish consumption advisories due to contamination with polychlorinated biphenyls (PCBs). In this study, natural attenuation and biostimulation of PCBs and microbial communities responsible for PCB transformations were investigated in Ohio River sediments. Natural attenuation of PCBs was negligible in sediments, which was likely attributed to low temperature conditions during most of the year, as well as low amounts of available nitrogen, phosphorus, and organic carbon. -

Geomicrobiological Processes in Extreme Environments: a Review
202 Articles by Hailiang Dong1, 2 and Bingsong Yu1,3 Geomicrobiological processes in extreme environments: A review 1 Geomicrobiology Laboratory, China University of Geosciences, Beijing, 100083, China. 2 Department of Geology, Miami University, Oxford, OH, 45056, USA. Email: [email protected] 3 School of Earth Sciences, China University of Geosciences, Beijing, 100083, China. The last decade has seen an extraordinary growth of and Mancinelli, 2001). These unique conditions have selected Geomicrobiology. Microorganisms have been studied in unique microorganisms and novel metabolic functions. Readers are directed to recent review papers (Kieft and Phelps, 1997; Pedersen, numerous extreme environments on Earth, ranging from 1997; Krumholz, 2000; Pedersen, 2000; Rothschild and crystalline rocks from the deep subsurface, ancient Mancinelli, 2001; Amend and Teske, 2005; Fredrickson and Balk- sedimentary rocks and hypersaline lakes, to dry deserts will, 2006). A recent study suggests the importance of pressure in the origination of life and biomolecules (Sharma et al., 2002). In and deep-ocean hydrothermal vent systems. In light of this short review and in light of some most recent developments, this recent progress, we review several currently active we focus on two specific aspects: novel metabolic functions and research frontiers: deep continental subsurface micro- energy sources. biology, microbial ecology in saline lakes, microbial Some metabolic functions of continental subsurface formation of dolomite, geomicrobiology in dry deserts, microorganisms fossil DNA and its use in recovery of paleoenviron- Because of the unique geochemical, hydrological, and geological mental conditions, and geomicrobiology of oceans. conditions of the deep subsurface, microorganisms from these envi- Throughout this article we emphasize geomicrobiological ronments are different from surface organisms in their metabolic processes in these extreme environments. -

Composition of Free-Living, Aggregate-Associated and Sediment Surface-Associated Bacterial Communities in the German Wadden Sea
AQUATIC MICROBIAL ECOLOGY Vol. 38: 15–30, 2005 Published January 21 Aquat Microb Ecol Composition of free-living, aggregate-associated and sediment surface-associated bacterial communities in the German Wadden Sea Heike Stevens, Thorsten Brinkhoff, Meinhard Simon* Institute for Chemistry and Biology of the Marine Environment (ICBM), University of Oldenburg, PO Box 2503, 26111 Oldenburg, Germany ABSTRACT: The Wadden Sea is a tidal flat ecosystem at the land-sea interface of the southern North Sea with a high load of suspended matter and nutrients. Despite the general importance of microbial processes, the composition of bacterial communities in this ecosystem has been little studied. There- fore, we investigated the composition of the bacterial communities freely suspended (FL) in the bulk water, associated with suspended aggregates (AG) and with the oxic sediment surface (SE) over an annual cycle (April 1999 to June 2000) in the East Frisian Wadden Sea, by applying denaturing gra- dient gel electrophoresis (DGGE) of PCR-amplified 16S rRNA gene fragments. Wind strength, sus- pended particulate matter (SPM), particulate organic carbon (POC), chlorophyll, phaeopigments, and the composition of the phytoplankton community were also studied. The dynamics of these parame- ters covaried to various extents, but not systematically, with that of bacterial numbers and community composition. A cluster analysis revealed that the DGGE banding patterns of the 3 communities grouped separately even though band overlaps among the communities occurred. Some identical or highly similar phylotypes were found in the FL- and AG-associated communities, and in the AG- and SE-associated communities, as shown by sequence analysis of excised bands. -

Full Paper Ilumatobacter Fluminis Gen. Nov., Sp. Nov., a Novel Actinobacterium Isolated from the Sediment of an Estuary
J. Gen. Appl. Microbiol., 55, 201‒205 (2009) Full Paper Ilumatobacter fl uminis gen. nov., sp. nov., a novel actinobacterium isolated from the sediment of an estuary Atsuko Matsumoto,1 Hiroki Kasai,2 Yoshihide Matsuo,2 Satoshi Ōmura,1 Yoshikazu Shizuri,2 and Yōko Takahashi1,* 1 Kitasato Institute for Life Sciences, Kitasato University, Minato-ku, Tokyo 108‒8641, Japan 2 Marine Biotechnology Institute, Kitasato University, Kamaishi, Iwate 026‒0001, Japan (Received December 1, 2008; Accepted February 2, 2009) Bacterial strain YM22-133T was isolated from the sediment of an estuary and grew in media with an artifi cial seawater base. Strain YM22-133T was Gram-positive, aerobic, non-motile and rod shaped. The cell-wall peptidoglycan contained LL-DAP, glycine, alanine and hydroxyglutamate. The predominant menaquinone was MK-9 (H8), with MK-9 (H0), MK-9 (H2), MK-9 (H4) and MK-9 (H6) present as minor menaquinones. The G+C content of the genomic DNA from the strain was 68 mol%. Phylogenetic analysis of the 16S rRNA gene sequence showed that the strain is near- est to Acidimicrobium ferrooxidans DSM 10331T. However, the similarity is relatively low (87.1%) and the physiological characteristics are also different: Acidimicrobium ferrooxidans is thermo- tolerant and acidophilic. Therefore, strain YM22-133T can be classifi ed as a novel genus and species, Ilumatobacter fl uminis gen. nov., sp. nov. (type strain YM22-133T =DSM 18936T=MBIC 08263T). Key Words—Acidimicrobium; Actinobacteria; artifi cial sea water; Ilumatobacter fl uminis gen. nov., sp. nov. Introduction isolated as part of this study. Phylogenetic analysis on the basis of 16S rRNA gene sequence analysis showed Recently, bacteria isolated from marine environ- that the strain is most closely related to the genus Aci- ments have attracted attention due to the recognition dimicrobium (Clark and Norris, 1996). -

Development of Bacterial Communities in Biological Soil Crusts Along
1 Development of bacterial communities in biological soil crusts along 2 a revegetation chronosequence in the Tengger Desert, northwest 3 China 4 5 Author names and affiliations: 6 Lichao Liu1, Yubing Liu1, 2 *, Peng Zhang1, Guang Song1, Rong Hui1, Zengru Wang1, Jin Wang1, 2 7 1Shapotou Desert Research & Experiment Station, Northwest Institute of Eco-Environment and Resources, Chinese 8 Academy of Sciences, Lanzhou, 730000, China 9 2Key Laboratory of Stress Physiology and Ecology in Cold and Arid Regions of Gansu Province, Northwest Institute 10 of Eco–Environment and Resources, Chinese Academy of Sciences, Lanzhou 730000, China 11 12 * Corresponding author: Yubing Liu 13 Address: Donggang West Road 320, Lanzhou 730000, P. R. China. 14 Tel: +86 0931 4967202. 15 E-mail address: [email protected] 16 17 Abstract. Knowledge of structure and function of microbial communities in different 18 successional stages of biological soil crusts (BSCs) is still scarce for desert areas. In this study, 19 Illumina MiSeq sequencing was used to assess the composition changes of bacterial communities 20 in different ages of BSCs in the revegetation of Shapotou in the Tengger Desert. The most dominant 21 phyla of bacterial communities shifted with the changed types of BSCs in the successional stages, 22 from Firmicutes in mobile sand and physical crusts to Actinobacteria and Proteobacteria in BSCs, 23 and the most dominant genera shifted from Bacillus, Enterococcus and Lactococcus to 24 RB41_norank and JG34-KF-361_norank. Alpha diversity and quantitative real-time PCR analysis 25 indicated that bacteria richness and abundance reached their highest levels after 15 years of BSC 26 development. -

Specialized Microbiome of the Cave-Dwelling Sponge
Specialized microbiome of the cave-dwelling sponge Plakina kanaky (Porifera, Homoscleromorpha) Cesar Ruiz, Marcela Villegas-Plazas, Olivier Thomas, Howard Junca, Thierry Perez To cite this version: Cesar Ruiz, Marcela Villegas-Plazas, Olivier Thomas, Howard Junca, Thierry Perez. Specialized mi- crobiome of the cave-dwelling sponge Plakina kanaky (Porifera, Homoscleromorpha). FEMS Micro- biology Ecology, Wiley-Blackwell, 2020, 96 (4), pp.fiaa043. 10.1093/femsec/fiaa043. hal-02545390 HAL Id: hal-02545390 https://hal-amu.archives-ouvertes.fr/hal-02545390 Submitted on 20 Apr 2021 HAL is a multi-disciplinary open access L’archive ouverte pluridisciplinaire HAL, est archive for the deposit and dissemination of sci- destinée au dépôt et à la diffusion de documents entific research documents, whether they are pub- scientifiques de niveau recherche, publiés ou non, lished or not. The documents may come from émanant des établissements d’enseignement et de teaching and research institutions in France or recherche français ou étrangers, des laboratoires abroad, or from public or private research centers. publics ou privés. Specialized microbiome of the cave-dwelling sponge Plakina kanaky (Porifera, Homoscleromorpha) César Ruiz1#, Marcela Villegas-Plazas2#, Olivier P. Thomas3, Howard Junca2, Thierry 1* Pérez 1 Institut Méditerranéen de Biodiversité et d’Ecologie Marine et Continentale, UMR 7263 CNRS, Aix Marseille Université, IRD, Avignon Université, Station Marine d’Endoume, Rue Batterie des Lions, 13007, Marseille, France. 2 RG Microbial Ecology: -

Characterization of the Uncommon Enzymes from (2004)
OPEN Mannosylglucosylglycerate biosynthesis SUBJECT AREAS: in the deep-branching phylum WATER MICROBIOLOGY MARINE MICROBIOLOGY Planctomycetes: characterization of the HOMEOSTASIS MULTIENZYME COMPLEXES uncommon enzymes from Rhodopirellula Received baltica 13 March 2013 Sofia Cunha1, Ana Filipa d’Avo´1, Ana Mingote2, Pedro Lamosa3, Milton S. da Costa1,4 & Joana Costa1,4 Accepted 23 July 2013 1Center for Neuroscience and Cell Biology, University of Coimbra, 3004-517 Coimbra, Portugal, 2Instituto de Tecnologia Quı´mica Published e Biolo´gica, Universidade Nova de Lisboa, 2780-157 Oeiras, Portugal, 3Centro de Ressonaˆncia Magne´tica Anto´nio Xavier, 7 August 2013 Instituto de Tecnologia Quı´mica e Biolo´gica, Universidade Nova de Lisboa, 2781-901 Oeiras, Portugal, 4Department of Life Sciences, University of Coimbra, Apartado 3046, 3001-401 Coimbra, Portugal. Correspondence and The biosynthetic pathway for the rare compatible solute mannosylglucosylglycerate (MGG) accumulated by requests for materials Rhodopirellula baltica, a marine member of the phylum Planctomycetes, has been elucidated. Like one of the should be addressed to pathways used in the thermophilic bacterium Petrotoga mobilis, it has genes coding for J.C. ([email protected].) glucosyl-3-phosphoglycerate synthase (GpgS) and mannosylglucosyl-3-phosphoglycerate (MGPG) synthase (MggA). However, unlike Ptg. mobilis, the mesophilic R. baltica uses a novel and very specific MGPG phosphatase (MggB). It also lacks a key enzyme of the alternative pathway in Ptg. mobilis – the mannosylglucosylglycerate synthase (MggS) that catalyses the condensation of glucosylglycerate with GDP-mannose to produce MGG. The R. baltica enzymes GpgS, MggA, and MggB were expressed in E. coli and characterized in terms of kinetic parameters, substrate specificity, temperature and pH dependence. -

J. Gen. Appl. Microbiol., 48, 109–115 (2002)
J. Gen. Appl. Microbiol., 48, 109–115 (2002) Full Paper Haliangium ochraceum gen. nov., sp. nov. and Haliangium tepidum sp. nov.: Novel moderately halophilic myxobacteria isolated from coastal saline environments Ryosuke Fudou,* Yasuko Jojima, Takashi Iizuka, and Shigeru Yamanaka† Central Research Laboratories, Ajinomoto Co., Inc., Kawasaki 210–8681, Japan (Received November 21, 2001; Accepted January 31, 2002) Phenotypic and phylogenetic studies were performed on two myxobacterial strains, SMP-2 and SMP-10, isolated from coastal regions. The two strains are morphologically similar, in that both produce yellow fruiting bodies, comprising several sessile sporangioles in dense packs. They are differentiated from known terrestrial myxobacteria on the basis of salt requirements (2–3% NaCl) and the presence of anteiso-branched fatty acids. Comparative 16S rRNA gene sequencing studies revealed that SMP-2 and SMP-10 are genetically related, and constitute a new cluster within the myxobacteria group, together with the Polyangium vitellinum Pl vt1 strain as the clos- est neighbor. The sequence similarity between the two strains is 95.6%. Based on phenotypic and phylogenetic evidence, it is proposed that these two strains be assigned to a new genus, JCM؍) .Haliangium gen. nov., with SMP-2 designated as Haliangium ochraceum sp. nov .(DSM 14436T؍JCM 11304T؍) .DSM 14365T), and SMP-10 as Haliangium tepidum sp. nov؍11303T Key Words——Haliangium gen. nov.; Haliangium ochraceum sp. nov.; Haliangium tepidum sp. nov.; marine myxobacteria Introduction terized as Gram-negative, rod-shaped gliding bacteria with high GϩC content. Analyses of 16S rRNA gene Myxobacteria are unique in their complex life cycle. sequences reveal that they form a relatively homoge- Under starvation conditions, bacterial cells gather to- neous cluster within the d-subclass of Proteobacteria gether to form aggregates that subsequently differenti- (Shimkets and Woese, 1992). -

Genomic and Transcriptomic Resolution of Organic Matter Utilization Among Deep-Sea Bacteria in Guaymas Basin Hydrothermal Plumes
fmicb-07-01125 July 27, 2016 Time: 11:18 # 1 ORIGINAL RESEARCH published: 27 July 2016 doi: 10.3389/fmicb.2016.01125 Genomic and Transcriptomic Resolution of Organic Matter Utilization Among Deep-Sea Bacteria in Guaymas Basin Hydrothermal Plumes Meng Li1*, Sunit Jain2 and Gregory J. Dick2,3,4 1 Institute for Advanced Study, Shenzhen University, Shenzhen, China, 2 Department of Earth and Environmental Sciences, University of Michigan, Ann Arbor, MI, USA, 3 Department of Ecology and Evolutionary Biology, University of Michigan, Ann Arbor, MI, USA, 4 Center of Computational Medicine and Bioinformatics, University of Michigan, Ann Arbor, MI, USA Microbial chemosynthesis within deep-sea hydrothermal vent plumes is a regionally important source of organic carbon to the deep ocean. Although chemolithoautotrophs within hydrothermal plumes have attracted much attention, a gap remains in Edited by: understanding the fate of organic carbon produced via chemosynthesis. In the present Marc Strous, University of Calgary, Canada study, we conducted shotgun metagenomic and metatranscriptomic sequencing on Reviewed by: samples from deep-sea hydrothermal vent plumes and surrounding background A. Murat Eren, seawaters at Guaymas Basin (GB) in the Gulf of California. De novo assembly University of Chicago, USA of metagenomic reads and binning by tetranucleotide signatures using emergent Carolin Regina Löscher, GEOMAR Helmholtz Centre for Ocean self-organizing maps (ESOM) revealed 66 partial and nearly complete bacterial Research Kiel, Germany genomes. These