+ Complex Tissues
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Bark Anatomy and Cell Size Variation in Quercus Faginea
Turkish Journal of Botany Turk J Bot (2013) 37: 561-570 http://journals.tubitak.gov.tr/botany/ © TÜBİTAK Research Article doi:10.3906/bot-1201-54 Bark anatomy and cell size variation in Quercus faginea 1,2, 2 2 2 Teresa QUILHÓ *, Vicelina SOUSA , Fatima TAVARES , Helena PEREIRA 1 Centre of Forests and Forest Products, Tropical Research Institute, Tapada da Ajuda, 1347-017 Lisbon, Portugal 2 Centre of Forestry Research, School of Agronomy, Technical University of Lisbon, Tapada da Ajuda, 1349-017 Lisbon, Portugal Received: 30.01.2012 Accepted: 27.09.2012 Published Online: 15.05.2013 Printed: 30.05.2013 Abstract: The bark structure of Quercus faginea Lam. in trees 30–60 years old grown in Portugal is described. The rhytidome consists of 3–5 sequential periderms alternating with secondary phloem. The phellem is composed of 2–5 layers of cells with thin suberised walls and narrow (1–3 seriate) tangential band of lignified thick-walled cells. The phelloderm is thin (2–3 seriate). Secondary phloem is formed by a few tangential bands of fibres alternating with bands of sieve elements and axial parenchyma. Formation of conspicuous sclereids and the dilatation growth (proliferation and enlargement of parenchyma cells) affect the bark structure. Fused phloem rays give rise to broad rays. Crystals and druses were mostly seen in dilated axial parenchyma cells. Bark thickness, sieve tube element length, and secondary phloem fibre wall thickness decreased with tree height. The sieve tube width did not follow any regular trend. In general, the fibre length had a small increase toward breast height, followed by a decrease towards the top. -

Plant Histology and Anatomy Q
PLANT HISTOLOGY AND ANATOMY Q. Transcellular strands are seen in a) Xylem vessels b) ThidTracheids c) Parenchyma cells d) Sieve tubes Q. Epiphytes absorb water by a spongy tissue called a) Mesophy ll b) Velamen c) Conjuctive tissue d) Phloem Q. The presence of vessel s and companion cells are characters of a) Gymnosperms b) Angiosperms c) Bryophytes d) Pteridophytes Q. AhilAmphivasal vascul lbdlar bundle is found in a) Cycas and Dryopteris b) Dracaena and Yucca c) Helianthus and Cucurbita d) maize and Wheat Q. Bamboo and grasses elonggyate by the activity of a) Apical meristem b) Intercalary meristem c) SdSecondary meritistem d) Lateral ameristem Q. Fibres associated with phloem are called a) Intraxylary fibres b) Pericycle fibres c) Bast fibres d) Cortical fibres Q. Callose is found in a) Sieve Plates b) Cross walls of tracheids c) Phloem parenchyma d) Comapanion cell Q. Which are common in xylem and phloem tissues? a) PhParenchyma and CllCollench yma b) Collenchyma and Sclerenchyma c) Parenchyma and Sclerenchyma d) Aerenchyma and Sclerenchyma Q. Quiescent centre is found in a) Root tip b) Shoot tip c) Floral tip d) Leaf tip Q. The plastids in meristematic tissue are in a a) Fullyyp developed state b) Half developed state c) Proplastid state d) Plasmolysed state Q. In hydrophytes, aerenchyma helps in a) Attachment b) Photosynthesis c) Buoyancy d) Mechanical support Q. Cistoliths are composedfd of a) Calcium oxalate b) Calcium carbonate c) GGucosdeslucosides d) MgCO 3 Q. CllCollench yma differs f rom sclerenchyma a) Retaining protoplasm at maturity b) Having thick walls c) HHiaving w idlide lumen d) Being meristematic Q. -

EVIDENCE for AUXIN REGULATION of BORDERED-PIT POSITIONING DURING TRACHEID DIFFERENTIATION in LARIX LARICINA By
IAWA Journal, Vol. 16 (3),1995: 289-297 EVIDENCE FOR AUXIN REGULATION OF BORDERED-PIT POSITIONING DURING TRACHEID DIFFERENTIATION IN LARIX LARICINA by Mathew Adam Leitch & Rodney Arthur Savidge 1 Faculty of Forestry and Environmental Management, University of New Brunswick, Fredericton, New Brunswick, E3B 6C2, Canada SUMMARY Chips containing cambium intact between xylem and phloem were cut from 8-year-old stem regions of 20-30-year-old dormant Larix laricina in late spring and cultured under controlled conditions for six weeks on a defined medium containing varied concentrations of I-naphthalene acetic acid (NAA, a synthetic auxin). Microscopy revealed that auxin was essential for cambial growth and tracheid differentiation. Low (0.1 mg/l) and high (10.0 mg/l) auxin concentrations were conducive to bordered pits forming in tangential walls, whereas an intermediate concentration (1.0 mg/l) of NAA favoured positioning of pits in radial walls. INTRODUCTION Two decades ago, Barnett and Harris (1975) pointed out that the processes involved in bordered-pit formation were incompletely understood, in particular the way in which the pit site is determined and how adjoining cells form a perfectly symmetrical bor dered-pit pair. Observing that radial walls of enlarging cambial derivatives were vari ably thick and thin, Barnett and Harris (1975) advanced the hypothesis that bordered pit development occurs at sites where the primary wall is thinned through the process of centrifugal displacement of microfibrils. Others, however, considered that a thick ening rather than a thinning of the primary wall was indicative of the site where pit development commenced (FengeI1972; Parham & Baird 1973). -

YELLOW-POPLAR (LIRIODENDRON TULIPIFERA L.)' George Lowerts E. A. Wheeler Robert C. Kellison
CHARACTERISTICS OF WOUND-ASSOCIATED WOOD OF YELLOW-POPLAR (LIRIODENDRON TULIPIFERA L.)' George Lowerts Forest Geneticist Woodlands Research, Union Camp Corp. Rincon, GA 31426 E. A. Wheeler Associate F'rofessor Department of Wood and Paper Science, North Carolina State University Raleigh, NC 27695-8005 and Robert C. Kellison Professor, Department of Forestry and Director, Hardwood Research Cooperative School of Forest Resoumes Raleigh, NC 27695-8002 (Received May 1985) ABSTRACT Selectedanatomical characteristicsand specificgravity ofydlow-poplar wood formed after wounding and adjacent to the wound were compared to similar characteristics of yellow-poplar wood formed before and after wounding and away from the wound. The wood formed immediately after wounding was similar anatomically to the bamer zones described for other species. Vessel volume, vessel diameter, percentage of vessel multiples, and vessel elcment length were significantly lower in wound- associated wood, while ray volume, ray density, and specific gravity were significantly greater. Such changes in the vessel system would result in a decrease in conductivity in the wounded area, while the increase in parenchyma would increase the potential for manufacture of fungitoxic compounds. With increasing radial distance from the wound area, the anatomical features of the wound-associated wood -~radualh . a~~roached .. those of normal wood, although. by. four years after wounding, the wood still had not returned to normal. The specific gravity stayed significantly greater. Keywords: Liriodendron lulipirpra L., yellow-poplar, barrier zones, wood anatomy, wounding, dis- coloration and decay. INTRODUCTION Wounds extending into the wood of branches, stems, or roots of a tree create an opportunity for the initiation of discoloration and decay. -

Microbiology and Plant Pathology
ALAGAPPA UNIVERSITY (Accredited with ‘A+’ Grade by NAAC (with CGPA: 3.64) in the Third Cycle and Graded As category - I University by MHRD-UGC) (A State University Established by the Government of TamilNadu) KARAIKUDI – 630 003 DIRECTORATE OF DISTANCE EDUCATION M. Sc. BOTONY Second Year – Third Semester 34631- MICROBIOLOGY AND PLANT PATHOLOGY Copy Right Reserved For Private Use only Authors: Dr. A. Arun, Associate Professor & Head (i/c), Department of Microbiology, Alagappa University, Karaikudi- 630 003. (Units 1-7) Dr. M. Jothibasu Assistant Professor Department of Botany Alagappa University Karaikudi-03 (Units 8-14) “The Copyright shall be vested with Alagappa University” All rights reserved. No part of this publication which is material protected by this copyright notice may be reproduced or transmitted or utilized or stored in any form or by any means now known or hereinafter invented, electronic, digital or mechanical, including photocopying, scanning, recording or by any information storage or retrieval system, without prior written permission from the Alagappa University, Karaikudi, Tamil Nadu. SYLLABI – BOOK MAPPING TABLE 34631- MICROBIOLOGY AND PLANT PATHOLOGY SYLLABI MAPPING IN BOOK BLOCK-1: SCOPE OF MICROBIOLOGY UNIT I Introduction to Microbiology- Scope of Microbiology- 1-8 Evolution into Science- Characterization of microorganisms. UNIT II Classification of Microorganisms- Archea and Bacteria- 9-15 Characteristics of Bacteria- Morphology -Ultra Structure. UNIT III Nutrition – Growth – Reproduction- Bacterial Cultural and 16-27 Cultural characteristics- Economic importance of bacteria BLOCK 2: VIRUS AND MYCOPLASMAS UNIT IV Virology – General features- Classification of virus- 28-34 Characteristics – Ultra structure. UNIT V Virus- Isolation – Purification –Chemical nature- Replication- 35-46 Transmission- Virions. -

INTERXYLARY PHLOEM (Included Phloem) by Marcelo R
INTERXYLARY PHLOEM (included phloem) By Marcelo R. Pace Interxylary phloem is the presence of phloem strands embedded within the secondary xylem (wood), and produced by the activity of a single cambium (Carlquist 2013). Stems with this cambial variant are also referred to as foraminate, due to the conspicuous interxylary phloem strands in the shape of dots scattered within the wood. (Fig. 1A). However, the presence of interxylary phloem is sometimes less evident and can only be confirmed by microscopy. Fig. 1. Stem cross-section of Strychnos (Loganiaceae). A. S. guianensis, macroscopic view. B. S. millepunctata, microscopic view. Interxylary phloem can have four different ontogenetic origins. The first one is where the cambium produces phloem in both directions (inside and outside), followed by the formation of xylem only towards the inside, and as a result, enclosing the phloem in the wood. Examples of this origin are present in Thunbergia (Acanthaceae; Fig. 2A) and Dicella (Malpighiaceae; Fig 2B). However, in Thunbergia the interxylary phloem is derived from the interfascicular cambium resulting in radial patches that alternate with regions of the xylem that originate from the fascicular cambium (Fig. 2A). A second origin of the interxylary phloem is through the formation of small phloem arcs. These later become embedded in the wood through the production of xylem by the cambium on their flanks. The resulting phloem islands will contain a fragment of cambium at the bottom. This type is present in Strychnos (Loganiaceae; Fig. 1), the African species of Combretum (Combretaceae; Van Vliet, 1979), and in at least one neotropical species of Combretum (Acevedo-Rodríguez, pers. -

Tissues and Other Levels of Organization MODULE - 1 Diversity and Evolution of Life
Tissues and Other Levels of Organization MODULE - 1 Diversity and Evolution of Life 5 Notes TISSUES AND OTHER LEVELS OF ORGANIZATION You have just learnt that cell is the fundamental structural and functional unit of organisms and that bodies of organisms are made up of cells of various shapes and sizes. Groups of similar cells aggregate to collectively perform a particular function. Such groups of cells are termed “tissues”. This lesson deals with the various kinds of tissues of plants and animals. OBJECTIVES After completing this lesson, you will be able to : z define tissues; z classify plant tissues; z name the various kinds of plant tissues; z enunciate the tunica corpus theory and histogen theory; z classify animal tissues; z describe the structure and function of various kinds of epithelial tissues; z describe the structure and function of various kinds of connective tissues; z describe the structure and function of muscular tissue; z describe the structure and function of nervous tissue. 5.1 WHAT IS A TISSUE Organs such as stem, and roots in plants, and stomach, heart and lungs in animals are made up of different kinds of tissues. A tissue is a group of cells with a common origin, structure and function. Their common origin means they are derived from the same layer (details in lesson No. 20) of cells in the embryo. Being of a common origin, there are similar in structure and hence perform the same function. Several types of tissues organise to form an organ. Example : Blood, bone, and cartilage are some examples of animal tissues whereas parenchyma, collenchyma, xylem and phloem are different tissues present in the plants. -

Chapter 5: the Shoot System I: the Stem
Chapter 5 The Shoot System I: The Stem THE FUNCTIONS AND ORGANIZATION OF THE SHOOT SYSTEM PRIMARY GROWTH AND STEM ANATOMY Primary Tissues of Dicot Stems Develop from the Primary Meristems The Distribution of the Primary Vascular Bundles Depends on the Position of Leaves Primary Growth Differs in Monocot and Dicot Stems SECONDARY GROWTH AND THE ANATOMY OF WOOD Secondary Xylem and Phloem Develop from Vascular Cambium Wood Is Composed of Secondary Xylem Gymnosperm Wood Differs from Angiosperm Wood Bark Is Composed of Secondary Phloem and Periderm Buds Are Compressed Branches Waiting to Elongate Some Monocot Stems Have Secondary Growth STEM MODIFICATIONS FOR SPECIAL FUNCTIONS THE ECONOMIC VALUE OF WOODY STEMS SUMMARY ECONOMIC BOTANY: How Do You Make A Barrel? 1 KEY CONCEPTS 1. The shoot system is composed of the stem and its lateral appendages: leaves, buds, and flowers. Leaves are arranged in different patterns (phyllotaxis): alternate, opposite, whorled, and spiral. 2. Stems provide support to the leaves, buds, and flowers. They conduct water and nutrients and produce new cells in meristems (shoot apical meristem, primary and secondary meristems). 3. Dicot stems and monocot stems are usually different. Dicot stems tend to have vascular bundles distributed in a ring, whereas in monocot stems they tend to be scattered. 4. Stems are composed of the following: epidermis, cortex and pith, xylem and phloem, and periderm. 5. Secondary xylem is formed by the division of cells in the vascular cambium and is called wood. The bark is composed of all of the tissues outside the vascular cambium, including the periderm (formed from cork cambium) and the secondary phloem. -
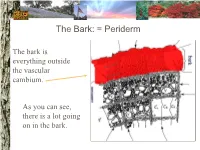
The Bark: = Periderm
The Bark: = Periderm The bark is everything outside the vascular cambium. As you can see, there is a lot going on in the bark. The Bark: periderm: phellogen (cork cambium): The phellogen is the region of cell division that forms the periderm tissues. Phellogen development influences bark appearance. The Bark: periderm: phellem (cork): Phellem replaces the epidermis as the tree increases in girth. Photosynthesis can take place in some trees both through the phellem and in fissures. The Bark: periderm: phelloderm: Phelloderm is active parenchyma tissue. Parenchyma cells can be used for storage, photosynthesis, defense, and even cell division! The Bark: phloem: Phloem tissue makes up the inner bark. However, it is vascular tissue formed from the vascular cambium. The Bark: phloem: sieve tube elements: Sieve tube elements actively transport photosynthates down the stem. Conifers have sieve cells instead. The cambium: The cambium is the primary meristem producing radial growth. It forms the phloem & xylem. The Xylem (wood): The xylem includes everything inside the vascular cambium. The Xylem: a growth increment (ring): The rings seen in many trees represent one growth increment. Growth rings provide the texture seen in wood. The Xylem: vessel elements: Hardwood species have vessel elements in addition to trachieds. Notice their location in the growth rings of this tree The Xylem: fibers: Fibers are cells with heavily lignified walls making them stiff. Many fibers in sapwood are alive at maturity and can be used for storage. The Xylem: axial parenchyma: Axial parenchyma is living tissue! Remember that parenchyma cells can be used for storage and cell division. -

Secretory Tissues (Gesneriaceae)
Acta Bot. Neerl. 46(4), December 1997, p.413-420 Secretory tissues of the flower of Sanango racemosum (Gesneriaceae). I. Light microscopy Sara Maldonadoi* and Marisa Oteguiif * Institute de Recursos Bioldgicos, INTA 1712, Villa Udaondo, Castelar, Argentina; fFacultad de Ciencias La 1900 La Naturales y Museo, Universidad Nacional de Plata, Plata, Argentina SUMMARY Sanango racemosum (Ruiz & Pav.) Barringer has a dry stigma without a free-flowing secretion fluid but with a hydrated proteinaceous pellicle. The stigmatic surface is covered with unicellular, bottle-shaped papillae. At maturity, a viscous emulsion is accumulated between the cuticle and the pecto-cellulosic wall of the papillae, causing it to become detached from the surface of the papilla cell walls. The style has a central solid core of transmitting tissue. The cells of the transmitting tissue are rich in starch and exhibit thick lateral walls rich in pectic substance. The nectary disk is a ring elongated into a cup, with five lobes at the top. One of the most conspicuous histological features of the disk is the abundance of starch in the secretory cells. The disk is supplied only by phloem; the stomata are found in the top of the lobes. A fluid substance is produced just before anthesis and secreted through the stomata with no visible decline in starch level. During anthesis and after fertilization, a rapid decline in starch is observed. The hypothesis that the disk has other functions besides that of a nectary is discussed. Key-words: disk, nectary, osmophore, Sanango, stigma, transmitting tissue. INTRODUCTION The monotypic genus Sanango G. S. Bunting and J. -

How Trees Modify Wood Development to Cope with Drought
DOI: 10.1002/ppp3.29 REVIEW Wood and water: How trees modify wood development to cope with drought F. Daniela Rodriguez‐Zaccaro | Andrew Groover US Forest Service, Pacific Southwest Research Station; Department of Plant Societal Impact Statement Biology, University of California Davis, Drought plays a conspicuous role in forest mortality, and is expected to become more Davis, California, USA severe in future climate scenarios. Recent surges in drought-associated forest tree Correspondence mortality have been documented worldwide. For example, recent droughts in Andrew Groover, US Forest Service, Pacific Southwest Research Station; Department of California and Texas killed approximately 129 million and 300 million trees, respec- Plant Biology, University of California Davis, tively. Drought has also induced acute pine tree mortality across east-central China, Davis, CA, USA. Email: [email protected], agroover@ and across extensive areas in southwest China. Understanding the biological pro- ucdavis.edu cesses that enable trees to modify wood development to mitigate the adverse effects Funding information of drought will be crucial for the development of successful strategies for future for- USDA AFRI, Grant/Award Number: 2015- est management and conservation. 67013-22891; National Science Foundation, Grant/Award Number: 1650042; DOE Summary Office of Science, Office of Biological and Drought is a recurrent stress to forests, causing periodic forest mortality with enor- Environmental Research, Grant/Award Number: DE-SC0007183 mous economic and environmental costs. Wood is the water-conducting tissue of tree stems, and trees modify wood development to create anatomical features and hydraulic properties that can mitigate drought stress. This modification of wood de- velopment can be seen in tree rings where not only the amount of wood but also the morphology of the water-conducting cells are modified in response to environmental conditions. -

Intra-Organismal Variation in the Structure of Plant Vascular Transport Tissues in Poplar Trees
Trees (2018) 32:1335–1346 https://doi.org/10.1007/s00468-018-1714-z ORIGINAL ARTICLE Intra-organismal variation in the structure of plant vascular transport tissues in poplar trees Anna L. Jacobsen1 · Jessica Valdovinos‑Ayala1 · F. Daniela Rodriguez‑Zaccaro1 · M. Angela Hill‑Crim1 · Marta I. Percolla1 · Martin D. Venturas2 Received: 8 December 2017 / Accepted: 17 May 2018 / Published online: 24 May 2018 © Springer-Verlag GmbH Germany, part of Springer Nature 2018 Abstract Key message Phloem and xylem conduit structure vary greatly throughout the body of Populus trichocarpa trees, par- ticularly between roots and shoots. This has implications for understanding organ and whole plant vascular function. Abstract Woody plant vascular transport occurs predominantly within secondary xylem and phloem, which are both produced by the vascular cambium during secondary growth. We examined how vessel and sieve tube structure varied throughout the plant body of P. trichocarpa trees and whether xylem and phloem conduit structure was correlated across different positions within the plant. We excavated entire juvenile P. trichocarpa trees and measured vessel and sieve tube structural traits of current-year growth in 1 m increments along the main root:shoot axis. Trees were > 4 m tall and had roots that extended 4–5 m at their longest length. We found that both sieve tube and vessel diameters greatly varied throughout the plant body and with organ diameter. Roots had wider diameter conduits than shoots. Sieve tube diameter was strongly correlated with vessel diameter, which may be related to their common developmental origin. Other structural traits, such as pit membrane area and pit density for xylem, and sieve plate area and number of sieve areas per plate for phloem, also varied and were correlated with changes in conduit diameter.