Aortic Arches
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

THE DORSAL AORTAE, Which Run with the Long Axis of the Embryo and Form the Continuation of the Endocardial Heart Tubes
AORTIC ARCH SYSTEM The major arteries in an early embryo are represented by a pair of vessels THE DORSAL AORTAE, which run with the long axis of the embryo and form the continuation of the endocardial heart tubes. The cranial portion of each dorsal aorta forms an arc on both sides of the foregut, thus establishing the first pair of aortic arch arteries, termed aortic arches Dr. Amjad Shatarat, School of Medicine, 1 The University of Jordan Arterial System • Aortic Arches • they run within branchial (pharyngeal) arches • These arteries, the aortic arches, arise from the aortic sac, the most distal part of the truncus arteriosus . • The aortic sac, giving rise to a total of five pairs of arteries. • The pharyngeal arches and their vessels appear in a cranial-to-caudal sequence, so that they are not all present simultaneously. • Consequently, the five arches are numbered I, II, III, IV, and VI . • During further development, this arterial pattern becomes modified, and some vessels regress completely. Dr. Amjad Shatarat, School of Medicine, 2 The University of Jordan • Division of the truncus arteriosus by the aorticopulmonary septum divides the outflow channel of the heart into the ventral aorta and the pulmonary trunk. The aortic sac then forms right and left horns, which subsequently give rise to the brachiocephalic artery and the proximal segment of the aortic arch, respectively . Dr. Amjad Shatarat, School of Medicine, 3 The University of Jordan The first pair of arteries largely disappears but remnants of them form part of the maxillary arteries, which supply the ears, teeth, and muscles of the eyes and face Derivatives of Second Pair of Pharyngeal Arch Arteries Dorsal parts of these arteries persist and form the stems of the small stapedial arteries; these small vessels run through the ring of the stapes, a small bone in the middle ear Dr. -

Fetal Blood Flow and Genetic Mutations in Conotruncal Congenital Heart Disease
Journal of Cardiovascular Development and Disease Review Fetal Blood Flow and Genetic Mutations in Conotruncal Congenital Heart Disease Laura A. Dyer 1 and Sandra Rugonyi 2,* 1 Department of Biology, University of Portland, Portland, OR 97203, USA; [email protected] 2 Department of Biomedical Engineering, Oregon Health & Science University, Portland, OR 97239, USA * Correspondence: [email protected] Abstract: In congenital heart disease, the presence of structural defects affects blood flow in the heart and circulation. However, because the fetal circulation bypasses the lungs, fetuses with cyanotic heart defects can survive in utero but need prompt intervention to survive after birth. Tetralogy of Fallot and persistent truncus arteriosus are two of the most significant conotruncal heart defects. In both defects, blood access to the lungs is restricted or non-existent, and babies with these critical conditions need intervention right after birth. While there are known genetic mutations that lead to these critical heart defects, early perturbations in blood flow can independently lead to critical heart defects. In this paper, we start by comparing the fetal circulation with the neonatal and adult circulation, and reviewing how altered fetal blood flow can be used as a diagnostic tool to plan interventions. We then look at known factors that lead to tetralogy of Fallot and persistent truncus arteriosus: namely early perturbations in blood flow and mutations within VEGF-related pathways. The interplay between physical and genetic factors means that any one alteration can cause significant disruptions during development and underscore our need to better understand the effects of both blood flow and flow-responsive genes. -

Download PDF Version
FIG. 4–1 Dorsal aspect of the 10-somite embryo. 24 IV the fourth week of life somite and neural tube period I. EMBRYO PROPER caudal openings of the tube are called neuropores. The rostral neuropore closes between 18 and 20 somites. The caudal neuro- A. EXTERNAL APPEARANCE pore closes at 25 somites. Figs. 4–1, 4–2 1. The specimens measure approximately 1 to 3.5 mm in length Brain and have 1 to 29 pairs of somites. Three brain subdivisions are present in the cranial portion of the 2. The head and tail folds move the attachment of the amnion tube and are named, from cranial to caudal, the prosencephalon, to the ventral side of the head and tail regions, respectively. mesencephalon and rhombencephalon. The boundary between the The lateral body folds move the amnion attachment to the pros- and mesencephalon is demarcated by a ventral bend, called ventrolateral surface in the midportion of the embryo. the cephalic flexure. An external groove and a prominent swelling 3. The head region is elevated above the yolk sac by the large on the medial surface of the neural plate may also demarcate the pericardial sac, the midportion lies upon the yolk sac and the boundary. The boundary between the mes- and rhombencephalon caudal region is curved toward the yolk sac. is distinguished by a groove on the medial and lateral surfaces of 4. The embryo possesses somites, which are apparent through the neural plate or tube. the ectoderm. 5. The neural tube develops from the neural plate and remains Prosencephalon open at each end for 2 to 4 days. -

Endothelium in the Pharyngeal Arches 3, 4 and 6 Is Derived from the Second Heart Field
Thomas Jefferson University Jefferson Digital Commons Center for Translational Medicine Faculty Papers Center for Translational Medicine 1-15-2017 Endothelium in the pharyngeal arches 3, 4 and 6 is derived from the second heart field. Xia Wang Thomas Jefferson University Dongying Chen Thomas Jefferson University Kelley Chen Thomas Jefferson University Ali Jubran Thomas Jefferson University AnnJosette Ramirez Thomas Jefferson University Follow this and additional works at: https://jdc.jefferson.edu/transmedfp Part of the Translational Medical Research Commons LetSee next us page know for additional how authors access to this document benefits ouy Recommended Citation Wang, Xia; Chen, Dongying; Chen, Kelley; Jubran, Ali; Ramirez, AnnJosette; and Astrof, Sophie, "Endothelium in the pharyngeal arches 3, 4 and 6 is derived from the second heart field." (2017). Center for Translational Medicine Faculty Papers. Paper 45. https://jdc.jefferson.edu/transmedfp/45 This Article is brought to you for free and open access by the Jefferson Digital Commons. The Jefferson Digital Commons is a service of Thomas Jefferson University's Center for Teaching and Learning (CTL). The Commons is a showcase for Jefferson books and journals, peer-reviewed scholarly publications, unique historical collections from the University archives, and teaching tools. The Jefferson Digital Commons allows researchers and interested readers anywhere in the world to learn about and keep up to date with Jefferson scholarship. This article has been accepted for inclusion in Center for Translational Medicine Faculty Papers by an authorized administrator of the Jefferson Digital Commons. For more information, please contact: [email protected]. Authors Xia Wang, Dongying Chen, Kelley Chen, Ali Jubran, AnnJosette Ramirez, and Sophie Astrof This article is available at Jefferson Digital Commons: https://jdc.jefferson.edu/transmedfp/45 HHS Public Access Author manuscript Author ManuscriptAuthor Manuscript Author Dev Biol Manuscript Author . -

Patent Ductus Arteriosus About This Factsheet the Normal Heart
Understanding your child’s heart Patent ductus arteriosus About this factsheet The normal heart This factsheet is for parents of babies and children who The heart is a muscular pump which pumps blood through the have patent ductus arteriosus (PDA), which is also known as body and lungs. There are four chambers in the heart. The two persistent arterial duct. upper ones are called the right atrium and left atrium. These are separated by a wall called the atrial septum. The two lower It explains: chambers are called the right and left ventricles, and are separated • what patent ductus arteriosus is and how it is diagnosed by a wall called the ventricular septum. • how patent ductus arteriosus is treated • the benefits and risks of treatments. On each side of the heart, blood passes from the atrium, through a heart valve – the tricuspid valve on the right, and the mitral valve This factsheet does not replace the advice that doctors or on the left – into the ventricle. The ventricles are the main pumping nurses may give you, but it should help you to understand chambers of the heart. Each ventricle pumps blood out into an artery. what they tell you. The right ventricle pumps blood – blue in the illustration – into the pulmonary artery (the blood vessel that takes blood to the lungs). The left ventricle pumps blood – red in the illustration – into the aorta (the blood vessel that takes blood to the rest of the body). Blood flows from the right side of the heart, through the pulmonary valve into the pulmonary artery, and then to the lungs where it picks up oxygen. -
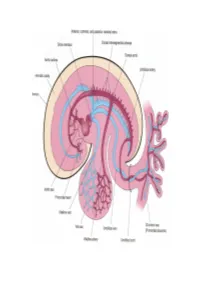
Development of HEART 4-VEINS
Development of brachiocephalic veins 1. Right brachiocephalic vein is formed by cranial part of right anterior cardinal vein and 2. Left brachiocephalic is formed by cranial part of left anterior cardinal vein and the interant.cardinal anastomosis. Development of superior vena cava 1. The part up to the opening of vena azygos develops from caudal part of right ant.cardinal vein and 2. The part below the opening (intrapericardial part) is formed by the right common cardinal vein. Development of azygos and hemiazygos veins A. 1. Vena azygos develops from right azygos line vein and 2. The arch of vena azygos is formed by the cranial end of right postcardinal vein. B. Hemiazygos veins are formed by the left azygos line vein. Development of Inferior vena cava Inferior vena cava is formed, from below upwards by: 1. Begins by the union of the two common iliac veins (postcardinal veins), 2. Right supracardinal, 3. Right supra-subcardinal anastomosis, 4. Right subcardinal, 5. New formation (hepatic segment) and 6. Hepatocardiac channel (terminal part of right vitelline vein). Congenital anomalies • Double inferior vena cava • Absence • Left SVC • Double SVC DEVELOPMENT OF PORTAL VEIN 1. The portal vein is formed behind the neck of pancreas by the union of superior mesentric and splenic vein to the left vitelline vein. 2. The part of the portal vein which is behind the Ist part of duodenum is formed by middle dorsal transverse anastomosis. 3. Part of portal vein which is in the free margin of lesser omentum is formed by cranial or distal part of right vitelline vein. -

Fate of the Mammalian Cardiac Neural Crest
Development 127, 1607-1616 (2000) 1607 Printed in Great Britain © The Company of Biologists Limited 2000 DEV4300 Fate of the mammalian cardiac neural crest Xiaobing Jiang1,3, David H. Rowitch4,*, Philippe Soriano5, Andrew P. McMahon4 and Henry M. Sucov2,3,‡ Departments of 1Biological Sciences and 2Cell & Neurobiology, 3Institute for Genetic Medicine, Keck School of Medicine, University of Southern California, 2250 Alcazar St., IGM 240, Los Angeles, CA 90033, USA 4Department of Molecular and Cell Biology, Harvard University, 16 Divinity Ave., Cambridge, MA 02138, USA 5Program in Developmental Biology, Division of Basic Sciences, A2-025, Fred Hutchinson Cancer Research Center, 1100 Fairview Avenue North, PO Box 19024, Seattle, WA 98109, USA *Present address: Department of Pediatric Oncology, Dana Farber Cancer Institute, 44 Binney St., Boston, MA 02115, USA ‡Author for correspondence (e-mail: [email protected]) Accepted 26 January; published on WWW 21 March 2000 SUMMARY A subpopulation of neural crest termed the cardiac neural of these vessels. Labeled cells populate the crest is required in avian embryos to initiate reorganization aorticopulmonary septum and conotruncal cushions prior of the outflow tract of the developing cardiovascular to and during overt septation of the outflow tract, and system. In mammalian embryos, it has not been previously surround the thymus and thyroid as these organs form. experimentally possible to study the long-term fate of this Neural-crest-derived mesenchymal cells are abundantly population, although there is strong inference that a similar distributed in midgestation (E9.5-12.5), and adult population exists and is perturbed in a number of genetic derivatives of the third, fourth and sixth pharyngeal arch and teratogenic contexts. -

Cardiovascular System Heart Development Cardiovascular System Heart Development
Cardiovascular System Heart Development Cardiovascular System Heart Development In human embryos, the heart begins to beat at approximately 22-23 days, with blood flow beginning in the 4th week. The heart is one of the earliest differentiating and functioning organs. • This emphasizes the critical nature of the heart in distributing blood through the vessels and the vital exchange of nutrients, oxygen, and wastes between the developing baby and the mother. • Therefore, the first system that completes its development in the embryo is called cardiovascular system. https://www.slideshare.net/DrSherifFahmy/intraembryonic-mesoderm-general-embryology Mesoderm is one of the three • Connective tissue primary germ layers that • Smooth and striated muscle • Cardiovascular System differentiates early in • Kidneys development that collectively • Spleen • Genital organs, ducts gives rise to all subsequent • Adrenal gland cortex tissues and organs. The cardiovascular system begins to develop in the third week of gestation. Blood islands develop in the newly formed mesoderm, and consist of (a) a central group of haemoblasts, the embryonic precursors of blood cells; (b) endothelial cells. Development of the heart and vascular system is often described together as the cardiovascular system. Development begins very early in mesoderm both within (embryonic) and outside (extra embryonic, vitelline, umblical and placental) the embryo. Vascular development occurs in many places. • Blood islands coalesce to form a vascular plexus. Preferential channels form arteries and veins. • Day 17 - Blood islands form first in the extra-embryonic mesoderm • Day 18 - Blood islands form next in the intra-embryonic mesoderm • Day 19 - Blood islands form in the cardiogenic mesoderm and coalesce to form a pair of endothelial heart tubes Development of a circulation • A circulation is established during the 4th week after the myocardium is differentiated. -

Congenital Abnormalities of the Aortic Arch: Revisiting the 1964 Stewart
Cardiovascular Pathology 39 (2019) 38–50 Contents lists available at ScienceDirect Cardiovascular Pathology Review Article Congenital abnormalities of the aortic arch: revisiting the 1964 ☆ Stewart classification Shengli Li a,⁎,HuaxuanWena,MeilingLianga,DandanLuoa, Yue Qin a,YimeiLiaoa, Shuyuan Ouyang b, Jingru Bi a, Xiaoxian Tian c, Errol R. Norwitz d,GuoyangLuoe,⁎⁎ a Department of Ultrasound, Shenzhen Maternity & Child Healthcare Hospital, Affiliated to Southern Medical University, Shenzhen, 518028, China b Department of Laboratory Medicine, Shenzhen Maternity & Child Healthcare Hospital, Affiliated to Southern Medical University, Shenzhen, 518028, China c Department of Ultrasound, Maternity & Child Healthcare Hospital of Guangxi Zhuang Autonomous Region, Nanning, Guangxi, 538001, China d Department of Obstetrics & Gynecology, Tufts University School of Medicine, Boston, MA 02111 e Department of Obstetrics & Gynecology, Howard University, College of Medicine, Washington, DC 20060, USA article info abstract Article history: The traditional classification of congenital aortic arch abnormalities was described by James Stewart and col- Received 12 June 2018 leagues in 1964. Since that time, advances in diagnostic imaging technology have led to better delineation of Received in revised form 27 November 2018 the vasculature anatomy and the identification of previously unrecognized and unclassified anomalies. In this Accepted 28 November 2018 manuscript, we review the existing literature and propose a series of modifications to the original Stewart -

Fetal Circulation
The Fetal Circulation Dr. S. Mathieu, Specialist Registrar in Anaesthesia Dr. D. J. Dalgleish, Consultant Anaesthetist Royal Bournemouth and Christchurch Hospitals Trust, UK Questions 1. In the fetal circulation: a) There are two umbilical arteries and one umbilical vein? b) Over 90% of blood passes the liver via the ductus venosus c) The foramen ovale divides the left and right ventricle d) The umbilical artery carries oxygenated blood from the placenta to the fetus e) The foramen ovale allows oxygenated blood to bypass the pulmonary circulation 2. In the fetal circulation: a) The oxygen dissociation curve of fetal haemoglobin is shifted to the left compared with adult haemoglobin ensuring oxygen delivery to the fetus despite low oxygen partial pressures b) It is the presence of the ductus arteriosus and large pulmonary vascular resistance which ensures most of the right ventricular output passes into the aorta c) The patency of the ductus arteriosus is maintained by high oxygen tensions d) The patency of the ductus arteriosus is maintained by the vasodilating effects of prostaglandin G2 e) 2,3-DPG levels are higher in fetal haemoglobin compared with adult haemaglobin 3. Changes at birth include: a) a fall in pulmonary vascular resistance b) a rise in systemic vascular resistance with clamping of the cord c) an increase in hypoxic pulmonary vasoconstriction d) a rise in left atrial pressure e) closure of the ductus arteriosus within 24 hours 4. The following congenital heart lesions are cyanotic: a) Ventricular septal defect b) Atrial septal defect c) Patent ductus arteriosus d) Tetralogy of Fallot e) Transposition of the great arteries MCQ answers at end Key points • The fetal circulation supplies the fetal tissues with oxygen and nutrients from the placenta. -

The Pattern and Mechanisms of Response to Oxygen by the Ductus Arteriosus and Umbilical Artery
Pediat. Res. 6: 693-700 (1972) Acetylcholine neonate atropine prematurity bradykinin sympathetic nervous system ductus arteriosus The Pattern and Mechanisms of Response to Oxygen by the Ductus Arteriosus and Umbilical Artery INGRID OBERHANSLI-WEISS, MICHAEL A. HEYMANN1391, ABRAHAM M. RUDOLPH, AND KENNETH L. MELMON Cardiovascular Research Institute and Departments of Pediatrics, Physiology and Pharmacology, University of California San Francisco, San Francisco, California, USA Extract Response of the ductus arteriosus and umbilical artery to changes in oxygen tension, to acetylcholine, and to sympathetic and parasympathetic blocking agents was studied in vitro in isolated rings obtained from 22 fetal lambs of 98- to 147-day gestation. After stabilization of tension at a baseline level (0.3-0.7 g) in a PO2 environment of 35-45 mm Hg, both increase of the PO2 to 550 mm Hg and decrease of the PO2 to 8 mm Hg of the bathing solution produced constriction. The mean maximal tension developed by the ductus arteriosus.was 3.91 g at high PO2 and 3.87 g at low POr The increase in maximal tension developed with advancing gestation was also similar at both high and low POj. At P02 levels of 8-550 mm Hg, acetylcholine produced a further increase in tension, whereas bradykinin only produced an increase in tension at high PO2- Alpha and beta sympathetic blockade had no effect on the constrictor response to oxygen. Atropine relaxed the ductus arteriosus and umbilical artery at both high and low Po2 levels; the degree of relaxation was related to drug concentration. Acetylcholin- esterase also relaxed the ductus arteriosus constricted by oxygen. -

Echocardiographic Follow-Up of Patent Foramen Ovale and the Factors Affecting Spontaneous Closure
Acta Cardiol Sin 2016;32:731-737 Brief Report doi: 10.6515/ACS20160205A Echocardiographic Follow-Up of Patent Foramen Ovale and the Factors Affecting Spontaneous Closure Ali Yildirim,1 Alperen Aydin,2 Tevfik Demir,1 Fatma Aydin,2 Birsen Ucar1 and Zubeyir Kilic1 Background: The aim of the present study was to evaluate the echocardiographic follow-up of patent foramen ovale, which is considered a potential etiological factor in various diseases, and to determine the factors affecting spontaneous closure. Methods: Between January 2000 and June 2012, records of 918 patients with patent foramen ovale were retrospectively reviewed. Patency of less than 3 mm around the fossa ovalis is called patent foramen ovale. Patients with cyanotic congenital heart diseases, severe heart valve disorders and severe hemodynamic left to right shunts were excluded from the study. The patients were divided into three groups based on age; 1 day-1 monthingroup1,1month-12monthsingroup2,andmorethan12monthsingroup3. Results: Of the 918 patients, 564 (61.4%) had spontaneous closure, 328 (35.8%) had patent foramen ovale continued, 15 (1.6%) patients had patent foramen ovale enlarged to 3-5 mm, 6 patients were enlarged to 5-8 mm, and in one patient patent foramen ovale reached to more than 8 mm size. Defect was spontaneously closed in 65.9% of the patients in group 1, 66.7% of the patients in group 2, and 52.3% of the patients in group 3. There was a negative correlation between the age of diagnosis and spontaneous closure (p < 0.05). Gender, prematurity and coexisting malformations such as patent ductus arteriosus and atrial septal aneurysm did not have any effect on spontaneous closure of patent foramen ovale (p > 0.05).