BMP Signaling Suppresses Gemc1 Expression and Ependymal
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Suppression of DNA Double-Strand Break Formation by DNA Polymerase B in Active DNA Demethylation Is Required for Development of Hippocampal Pyramidal Neurons
9012 • The Journal of Neuroscience, November 18, 2020 • 40(47):9012–9027 Development/Plasticity/Repair Suppression of DNA Double-Strand Break Formation by DNA Polymerase b in Active DNA Demethylation Is Required for Development of Hippocampal Pyramidal Neurons Akiko Uyeda,1 Kohei Onishi,1 Teruyoshi Hirayama,1,2,3 Satoko Hattori,4 Tsuyoshi Miyakawa,4 Takeshi Yagi,1,2 Nobuhiko Yamamoto,1 and Noriyuki Sugo1 1Graduate School of Frontier Biosciences, Osaka University, Suita, Osaka 565-0871, Japan, 2AMED-CREST, Japan Agency for Medical Research and Development, Suita, Osaka 565-0871, Japan, 3Department of Anatomy and Developmental Neurobiology, Tokushima University Graduate School of Medical Sciences, Kuramoto, Tokushima 770-8503, Japan, and 4Institute for Comprehensive Medical Science, Fujita Health University, Toyoake, Aichi 470-1192, Japan Genome stability is essential for brain development and function, as de novo mutations during neuronal development cause psychiatric disorders. However, the contribution of DNA repair to genome stability in neurons remains elusive. Here, we demonstrate that the base excision repair protein DNA polymerase b (Polb) is involved in hippocampal pyramidal neuron fl/fl differentiation via a TET-mediated active DNA demethylation during early postnatal stages using Nex-Cre/Polb mice of ei- ther sex, in which forebrain postmitotic excitatory neurons lack Polb expression. Polb deficiency induced extensive DNA dou- ble-strand breaks (DSBs) in hippocampal pyramidal neurons, but not dentate gyrus granule cells, and to a lesser extent in neocortical neurons, during a period in which decreased levels of 5-methylcytosine and 5-hydroxymethylcytosine were observed in genomic DNA. Inhibition of the hydroxylation of 5-methylcytosine by expression of microRNAs miR-29a/b-1 diminished DSB formation. -

Multifactorial Erβ and NOTCH1 Control of Squamous Differentiation and Cancer
Multifactorial ERβ and NOTCH1 control of squamous differentiation and cancer Yang Sui Brooks, … , Karine Lefort, G. Paolo Dotto J Clin Invest. 2014;124(5):2260-2276. https://doi.org/10.1172/JCI72718. Research Article Oncology Downmodulation or loss-of-function mutations of the gene encoding NOTCH1 are associated with dysfunctional squamous cell differentiation and development of squamous cell carcinoma (SCC) in skin and internal organs. While NOTCH1 receptor activation has been well characterized, little is known about how NOTCH1 gene transcription is regulated. Using bioinformatics and functional screening approaches, we identified several regulators of the NOTCH1 gene in keratinocytes, with the transcription factors DLX5 and EGR3 and estrogen receptor β (ERβ) directly controlling its expression in differentiation. DLX5 and ERG3 are required for RNA polymerase II (PolII) recruitment to the NOTCH1 locus, while ERβ controls NOTCH1 transcription through RNA PolII pause release. Expression of several identified NOTCH1 regulators, including ERβ, is frequently compromised in skin, head and neck, and lung SCCs and SCC-derived cell lines. Furthermore, a keratinocyte ERβ–dependent program of gene expression is subverted in SCCs from various body sites, and there are consistent differences in mutation and gene-expression signatures of head and neck and lung SCCs in female versus male patients. Experimentally increased ERβ expression or treatment with ERβ agonists inhibited proliferation of SCC cells and promoted NOTCH1 expression and squamous differentiation both in vitro and in mouse xenotransplants. Our data identify a link between transcriptional control of NOTCH1 expression and the estrogen response in keratinocytes, with implications for differentiation therapy of squamous cancer. Find the latest version: https://jci.me/72718/pdf Research article Multifactorial ERβ and NOTCH1 control of squamous differentiation and cancer Yang Sui Brooks,1,2 Paola Ostano,3 Seung-Hee Jo,1,2 Jun Dai,1,2 Spiro Getsios,4 Piotr Dziunycz,5 Günther F.L. -

Prospective Isolation of NKX2-1–Expressing Human Lung Progenitors Derived from Pluripotent Stem Cells
The Journal of Clinical Investigation RESEARCH ARTICLE Prospective isolation of NKX2-1–expressing human lung progenitors derived from pluripotent stem cells Finn Hawkins,1,2 Philipp Kramer,3 Anjali Jacob,1,2 Ian Driver,4 Dylan C. Thomas,1 Katherine B. McCauley,1,2 Nicholas Skvir,1 Ana M. Crane,3 Anita A. Kurmann,1,5 Anthony N. Hollenberg,5 Sinead Nguyen,1 Brandon G. Wong,6 Ahmad S. Khalil,6,7 Sarah X.L. Huang,3,8 Susan Guttentag,9 Jason R. Rock,4 John M. Shannon,10 Brian R. Davis,3 and Darrell N. Kotton1,2 2 1Center for Regenerative Medicine, and The Pulmonary Center and Department of Medicine, Boston University School of Medicine, Boston, Massachusetts, USA. 3Center for Stem Cell and Regenerative Medicine, Brown Foundation Institute of Molecular Medicine, University of Texas Health Science Center, Houston, Texas, USA. 4Department of Anatomy, UCSF, San Francisco, California, USA. 5Division of Endocrinology, Diabetes and Metabolism, Beth Israel Deaconess Medical Center and Harvard Medical School, Boston, Massachusetts, USA. 6Department of Biomedical Engineering and Biological Design Center, Boston University, Boston, Massachusetts, USA. 7Wyss Institute for Biologically Inspired Engineering, Harvard University, Boston, Massachusetts, USA. 8Columbia Center for Translational Immunology & Columbia Center for Human Development, Columbia University Medical Center, New York, New York, USA. 9Department of Pediatrics, Monroe Carell Jr. Children’s Hospital, Vanderbilt University, Nashville, Tennessee, USA. 10Division of Pulmonary Biology, Cincinnati Children’s Hospital, Cincinnati, Ohio, USA. It has been postulated that during human fetal development, all cells of the lung epithelium derive from embryonic, endodermal, NK2 homeobox 1–expressing (NKX2-1+) precursor cells. -

A Computational Approach for Defining a Signature of Β-Cell Golgi Stress in Diabetes Mellitus
Page 1 of 781 Diabetes A Computational Approach for Defining a Signature of β-Cell Golgi Stress in Diabetes Mellitus Robert N. Bone1,6,7, Olufunmilola Oyebamiji2, Sayali Talware2, Sharmila Selvaraj2, Preethi Krishnan3,6, Farooq Syed1,6,7, Huanmei Wu2, Carmella Evans-Molina 1,3,4,5,6,7,8* Departments of 1Pediatrics, 3Medicine, 4Anatomy, Cell Biology & Physiology, 5Biochemistry & Molecular Biology, the 6Center for Diabetes & Metabolic Diseases, and the 7Herman B. Wells Center for Pediatric Research, Indiana University School of Medicine, Indianapolis, IN 46202; 2Department of BioHealth Informatics, Indiana University-Purdue University Indianapolis, Indianapolis, IN, 46202; 8Roudebush VA Medical Center, Indianapolis, IN 46202. *Corresponding Author(s): Carmella Evans-Molina, MD, PhD ([email protected]) Indiana University School of Medicine, 635 Barnhill Drive, MS 2031A, Indianapolis, IN 46202, Telephone: (317) 274-4145, Fax (317) 274-4107 Running Title: Golgi Stress Response in Diabetes Word Count: 4358 Number of Figures: 6 Keywords: Golgi apparatus stress, Islets, β cell, Type 1 diabetes, Type 2 diabetes 1 Diabetes Publish Ahead of Print, published online August 20, 2020 Diabetes Page 2 of 781 ABSTRACT The Golgi apparatus (GA) is an important site of insulin processing and granule maturation, but whether GA organelle dysfunction and GA stress are present in the diabetic β-cell has not been tested. We utilized an informatics-based approach to develop a transcriptional signature of β-cell GA stress using existing RNA sequencing and microarray datasets generated using human islets from donors with diabetes and islets where type 1(T1D) and type 2 diabetes (T2D) had been modeled ex vivo. To narrow our results to GA-specific genes, we applied a filter set of 1,030 genes accepted as GA associated. -

The Capacity of Long-Term in Vitro Proliferation of Acute Myeloid
The Capacity of Long-Term in Vitro Proliferation of Acute Myeloid Leukemia Cells Supported Only by Exogenous Cytokines Is Associated with a Patient Subset with Adverse Outcome Annette K. Brenner, Elise Aasebø, Maria Hernandez-Valladares, Frode Selheim, Frode Berven, Ida-Sofie Grønningsæter, Sushma Bartaula-Brevik and Øystein Bruserud Supplementary Material S2 of S31 Table S1. Detailed information about the 68 AML patients included in the study. # of blasts Viability Proliferation Cytokine Viable cells Change in ID Gender Age Etiology FAB Cytogenetics Mutations CD34 Colonies (109/L) (%) 48 h (cpm) secretion (106) 5 weeks phenotype 1 M 42 de novo 241 M2 normal Flt3 pos 31.0 3848 low 0.24 7 yes 2 M 82 MF 12.4 M2 t(9;22) wt pos 81.6 74,686 low 1.43 969 yes 3 F 49 CML/relapse 149 M2 complex n.d. pos 26.2 3472 low 0.08 n.d. no 4 M 33 de novo 62.0 M2 normal wt pos 67.5 6206 low 0.08 6.5 no 5 M 71 relapse 91.0 M4 normal NPM1 pos 63.5 21,331 low 0.17 n.d. yes 6 M 83 de novo 109 M1 n.d. wt pos 19.1 8764 low 1.65 693 no 7 F 77 MDS 26.4 M1 normal wt pos 89.4 53,799 high 3.43 2746 no 8 M 46 de novo 26.9 M1 normal NPM1 n.d. n.d. 3472 low 1.56 n.d. no 9 M 68 MF 50.8 M4 normal D835 pos 69.4 1640 low 0.08 n.d. -

A Flexible Microfluidic System for Single-Cell Transcriptome Profiling
www.nature.com/scientificreports OPEN A fexible microfuidic system for single‑cell transcriptome profling elucidates phased transcriptional regulators of cell cycle Karen Davey1,7, Daniel Wong2,7, Filip Konopacki2, Eugene Kwa1, Tony Ly3, Heike Fiegler2 & Christopher R. Sibley 1,4,5,6* Single cell transcriptome profling has emerged as a breakthrough technology for the high‑resolution understanding of complex cellular systems. Here we report a fexible, cost‑efective and user‑ friendly droplet‑based microfuidics system, called the Nadia Instrument, that can allow 3′ mRNA capture of ~ 50,000 single cells or individual nuclei in a single run. The precise pressure‑based system demonstrates highly reproducible droplet size, low doublet rates and high mRNA capture efciencies that compare favorably in the feld. Moreover, when combined with the Nadia Innovate, the system can be transformed into an adaptable setup that enables use of diferent bufers and barcoded bead confgurations to facilitate diverse applications. Finally, by 3′ mRNA profling asynchronous human and mouse cells at diferent phases of the cell cycle, we demonstrate the system’s ability to readily distinguish distinct cell populations and infer underlying transcriptional regulatory networks. Notably this provided supportive evidence for multiple transcription factors that had little or no known link to the cell cycle (e.g. DRAP1, ZKSCAN1 and CEBPZ). In summary, the Nadia platform represents a promising and fexible technology for future transcriptomic studies, and other related applications, at cell resolution. Single cell transcriptome profling has recently emerged as a breakthrough technology for understanding how cellular heterogeneity contributes to complex biological systems. Indeed, cultured cells, microorganisms, biopsies, blood and other tissues can be rapidly profled for quantifcation of gene expression at cell resolution. -

Deciphering the Nature of Trp73 Isoforms in Mouse
cancers Article Deciphering the Nature of Trp73 Isoforms in Mouse Embryonic Stem Cell Models: Generation of Isoform-Specific Deficient Cell Lines Using the CRISPR/Cas9 Gene Editing System Lorena López-Ferreras 1,2,†, Nicole Martínez-García 1,3,†, Laura Maeso-Alonso 1,2,‡, Marta Martín-López 1,4,‡, Ángela Díez-Matilla 1,‡ , Javier Villoch-Fernandez 1,2, Hugo Alonso-Olivares 1,2, Margarita M. Marques 3,5,* and Maria C. Marin 1,2,* 1 Instituto de Biomedicina (IBIOMED), Universidad de León, 24071 León, Spain; [email protected] (L.L.-F.); [email protected] (N.M.-G.); [email protected] (L.M.-A.); [email protected] (M.M.-L.); [email protected] (Á.D.-M.); [email protected] (J.V.-F.); [email protected] (H.A.-O.) 2 Departamento de Biología Molecular, Universidad de León, 24071 León, Spain 3 Departamento de Producción Animal, Universidad de León, 24071 León, Spain 4 Biomar Microbial Technologies, Parque Tecnológico de León, Armunia, 24009 León, Spain 5 Instituto de Desarrollo Ganadero y Sanidad Animal (INDEGSAL), Universidad de León, 24071 León, Spain * Correspondence: [email protected] (M.M.M.); [email protected] (M.C.M.); Tel.: +34-987-291757 Citation: López-Ferreras, L.; (M.M.M.); +34-987-291490 (M.C.M.) Martínez-García, N.; Maeso-Alonso, † Equal contribution. ‡ Equal contribution. L.; Martín-López, M.; Díez-Matilla, Á.; Villoch-Fernandez, J.; Simple Summary: The Trp73 gene is involved in the regulation of multiple biological processes Alonso-Olivares, H.; Marques, M.M.; Marin, M.C. Deciphering the Nature such as response to stress, differentiation and tissue architecture. -

Ten Commandments for a Good Scientist
Unravelling the mechanism of differential biological responses induced by food-borne xeno- and phyto-estrogenic compounds Ana María Sotoca Covaleda Wageningen 2010 Thesis committee Thesis supervisors Prof. dr. ir. Ivonne M.C.M. Rietjens Professor of Toxicology Wageningen University Prof. dr. Albertinka J. Murk Personal chair at the sub-department of Toxicology Wageningen University Thesis co-supervisor Dr. ir. Jacques J.M. Vervoort Associate professor at the Laboratory of Biochemistry Wageningen University Other members Prof. dr. Michael R. Muller, Wageningen University Prof. dr. ir. Huub F.J. Savelkoul, Wageningen University Prof. dr. Everardus J. van Zoelen, Radboud University Nijmegen Dr. ir. Toine F.H. Bovee, RIKILT, Wageningen This research was conducted under the auspices of the Graduate School VLAG Unravelling the mechanism of differential biological responses induced by food-borne xeno- and phyto-estrogenic compounds Ana María Sotoca Covaleda Thesis submitted in fulfillment of the requirements for the degree of doctor at Wageningen University by the authority of the Rector Magnificus Prof. dr. M.J. Kropff, in the presence of the Thesis Committee appointed by the Academic Board to be defended in public on Tuesday 14 September 2010 at 4 p.m. in the Aula Unravelling the mechanism of differential biological responses induced by food-borne xeno- and phyto-estrogenic compounds. Ana María Sotoca Covaleda Thesis Wageningen University, Wageningen, The Netherlands, 2010, With references, and with summary in Dutch. ISBN: 978-90-8585-707-5 “Caminante no hay camino, se hace camino al andar. Al andar se hace camino, y al volver la vista atrás se ve la senda que nunca se ha de volver a pisar” - Antonio Machado – A mi madre. -
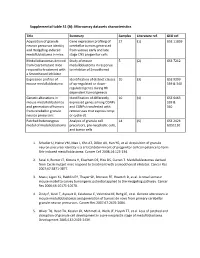
Supplemental Table S1 (A): Microarray Datasets Characteristics
Supplemental table S1 (A): Microarray datasets characteristics Title Summary Samples Literature ref. GEO ref. Acquisition of granule Gene expression profiling of 27 (1) GSE 11859 neuron precursor identity cerebellar tumors generated and Hedgehog‐induced from various early and late medulloblastoma in mice. stage CNS progenitor cells Medulloblastomas derived Study of mouse 5 (2) GSE 7212 from Cxcr6 mutant mice medulloblastoma in response respond to treatment with to inhibitor of Smoothened a Smoothened inhibitor Expression profiles of Identification of distinct classes 10 (3) GSE 9299 mouse medulloblastoma of up‐regulated or down‐ 339 & 340 regulated genes during Hh dependent tumorigenesis Genetic alterations in Identification of differently 10 (4) GSE 6463 mouse medulloblastomas expressed genes among CGNPs 339 & and generation of tumors and CGNPs transfected with 340 from cerebellar granule retroviruses that express nmyc neuron precursors or cyclin‐d1 Patched heterozygous Analysis of granule cell 14 (5) GSE 2426 model of medulloblastoma precursors, pre‐neoplastic cells, GDS1110 and tumor cells 1. Schuller U, Heine VM, Mao J, Kho AT, Dillon AK, Han YG, et al. Acquisition of granule neuron precursor identity is a critical determinant of progenitor cell competence to form Shh‐induced medulloblastoma. Cancer Cell 2008;14:123‐134. 2. Sasai K, Romer JT, Kimura H, Eberhart DE, Rice DS, Curran T. Medulloblastomas derived from Cxcr6 mutant mice respond to treatment with a smoothened inhibitor. Cancer Res 2007;67:3871‐3877. 3. Mao J, Ligon KL, Rakhlin EY, Thayer SP, Bronson RT, Rowitch D, et al. A novel somatic mouse model to survey tumorigenic potential applied to the Hedgehog pathway. Cancer Res 2006;66:10171‐10178. -

Mir-205: a Potential Biomedicine for Cancer Therapy
cells Review miR-205: A Potential Biomedicine for Cancer Therapy Neeraj Chauhan 1,2 , Anupam Dhasmana 1,2, Meena Jaggi 1,2, Subhash C. Chauhan 1,2 and Murali M. Yallapu 1,2,* 1 Department of Immunology and Microbiology, School of Medicine, University of Texas Rio Grande Valley, McAllen, TX 78504, USA; [email protected] (N.C.); [email protected] (A.D.); [email protected] (M.J.); [email protected] (S.C.C.) 2 South Texas Center of Excellence in Cancer Research, School of Medicine, University of Texas Rio Grande Valley, McAllen, TX 78504, USA * Correspondence: [email protected]; Tel.: +1-(956)-296-1734 Received: 3 June 2020; Accepted: 21 August 2020; Published: 25 August 2020 Abstract: microRNAs (miRNAs) are a class of small non-coding RNAs that regulate the expression of their target mRNAs post transcriptionally. miRNAs are known to regulate not just a gene but the whole gene network (signaling pathways). Accumulating evidence(s) suggests that miRNAs can work either as oncogenes or tumor suppressors, but some miRNAs have a dual nature since they can act as both. miRNA 205 (miR-205) is one such highly conserved miRNA that can act as both, oncomiRNA and tumor suppressor. However, most reports confirm its emerging role as a tumor suppressor in many cancers. This review focuses on the downregulated expression of miR-205 and discusses its dysregulation in breast, prostate, skin, liver, gliomas, pancreatic, colorectal and renal cancers. This review also confers its role in tumor initiation, progression, cell proliferation, epithelial to mesenchymal transition, and tumor metastasis. -

NERVOUS SYSTEM هذا الملف لالستزادة واثراء المعلومات Neuropsychiatry Block
NERVOUS SYSTEM هذا الملف لﻻستزادة واثراء المعلومات Neuropsychiatry block. قال تعالى: ) َو َل َق د َخ َل قنَا ا ِْلن َسا َن ِمن ُس ََل َل ة ِ من ِطي ن }12{ ثُ م َجعَ لنَاه ُ نُ ط َفة فِي َق َرا ر م ِكي ن }13{ ثُ م َخ َل قنَا ال ُّن ط َفة َ َع َل َقة َف َخ َل قنَا ا لعَ َل َقة َ ُم ضغَة َف َخ َل قنَا ا ل ُم ضغَة َ ِع َظا ما َف َك َس ونَا ا ل ِع َظا َم َل ح ما ثُ م أَن َشأنَاه ُ َخ ل قا آ َخ َر َفتَبَا َر َك ّللا ُ أَ ح َس ُن ا ل َخا ِل ِقي َن }14{( Resources BRS Embryology Book. Pathoma Book ( IN DEVELOPMENTAL ANOMALIES PART ). [email protected] 1 OVERVIEW A- Central nervous system (CNS) is formed in week 3 of development, during which time the neural plate develops. The neural plate, consisting of neuroectoderm, becomes the neural tube, which gives rise to the brain and spinal cord. B- Peripheral nervous system (PNS) is derived from three sources: 1. Neural crest cells 2. Neural tube, which gives rise to all preganglionic autonomic nerves (sympathetic and parasympathetic) and all nerves (-motoneurons and -motoneurons) that innervate skeletal muscles 3. Mesoderm, which gives rise to the dura mater and to connective tissue investments of peripheral nerve fibers (endoneurium, perineurium, and epineurium) DEVELOPMENT OF THE NEURAL TUBE Neurulation refers to the formation and closure of the neural tube. BMP-4 (bone morphogenetic protein), noggin (an inductor protein), chordin (an inductor protein), FGF-8 (fibroblast growth factor), and N-CAM (neural cell adhesion molecule) appear to play a role in neurulation. -

Molecular Markers and Potential Therapeutic Targets in Non-WNT/Non
Menyhárt et al. Journal of Hematology & Oncology (2019) 12:29 https://doi.org/10.1186/s13045-019-0712-y REVIEW Open Access Molecular markers and potential therapeutic targets in non-WNT/non-SHH (group 3 and group 4) medulloblastomas Otília Menyhárt1,2, Felice Giangaspero3,4 and Balázs Győrffy1,2* Abstract Childhood medulloblastomas (MB) are heterogeneous and are divided into four molecular subgroups. The provisional non-wingless-activated (WNT)/non-sonic hedgehog-activated (SHH) category combining group 3 and group 4 represents over two thirds of all MBs, coupled with the highest rates of metastases and least understood pathology. The molecular era expanded our knowledge about molecular aberrations involved in MB tumorigenesis, and here, we review processes leading to non-WNT/non-SHH MB formations. The heterogeneous group 3 and group 4 MBs frequently harbor rare individual genetic alterations, yet the emerging profiles suggest that infrequent events converge on common, potentially targetable signaling pathways. A mutual theme is the altered epigenetic regulation, and in vitro approaches targeting epigenetic machinery are promising. Growing evidence indicates the presence of an intermediate, mixed signature group along group 3 and group 4, and future clarifications are imperative for concordant classification, as misidentifying patient samples has serious implications for therapy and clinical trials. To subdue the high MB mortality, we need to discern mechanisms of disease spread and recurrence. Current preclinical models do not represent the full scale of group 3 and group 4 heterogeneity: all of existing group 3 cell lines are MYC-amplified and most mouse models resemble MYC-activated MBs. Clinical samples provide a wealth of information about the genetic divergence between primary tumors and metastatic clones, but recurrent MBs are rarely resected.