A Thesis Entitled Relationship Between Hamstring Strength And
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Iliopsoas Tendonitis/Bursitis Exercises
ILIOPSOAS TENDONITIS / BURSITIS What is the Iliopsoas and Bursa? The iliopsoas is a muscle that runs from your lower back through the pelvis to attach to a small bump (the lesser trochanter) on the top portion of the thighbone near your groin. This muscle has the important job of helping to bend the hip—it helps you to lift your leg when going up and down stairs or to start getting out of a car. A fluid-filled sac (bursa) helps to protect and allow the tendon to glide during these movements. The iliopsoas tendon can become inflamed or overworked during repetitive activities. The tendon can also become irritated after hip replacement surgery. Signs and Symptoms Iliopsoas issues may feel like “a pulled groin muscle”. The main symptom is usually a catch during certain movements such as when trying to put on socks or rising from a seated position. You may find yourself leading with your other leg when going up the stairs to avoid lifting the painful leg. The pain may extend from the groin to the inside of the thigh area. Snapping or clicking within the front of the hip can also be experienced. Do not worry this is not your hip trying to pop out of socket but it is usually the iliopsoas tendon rubbing over the hip joint or pelvis. Treatment Conservative treatment in the form of stretching and strengthening usually helps with the majority of patients with iliopsoas bursitis. This issue is the result of soft tissue inflammation, therefore rest, ice, anti- inflammatory medications, physical therapy exercises, and/or injections are effective treatment options. -

Vastus Medialis: a Reappraisal of VMO and VML
J. Phys. Ther. Sci. 24: 475–479, 2012 Vastus Medialis: a Reappraisal of VMO and VML EMILY J SKINNER1), PHILIP J ADDS1) 1) Division of Biomedical Sciences (Anatomy), St. George’s, University of London: Cranmer Terrace, London SW17 0RE, U.K. TEL: +44 208-7255208, E-mail: [email protected] Abstract. [Purpose] The morphology and innervation pattern of the vastus medialis (VM) were investigated to determine if there was an anatomical distinction between the oblique (VMO) and longus (VML) parts. [Subjects and Methods] Forty lower limbs were dissected. The innervation pattern was observed in 39 specimens. Muscle length and fibre angles of 14 specimens were recorded. [Results] In 22 specimens there was a distinct separation between the VML and VMO (change in fibre angle, fibrofascial plane, vasculature or nerve branch). The mean fibre angle of VMO was 52°, and the mean VML fibre angle was 5° (relative to the shaft of the femur). Ten limbs (25.6%) had separate innervation to the VML. The separation between VMO and VML was found to be more proximal than expected, with the VMO on average accounting for 70% of the VM, in contrast to the much more distal point of separation reported elsewhere, leading us to raise the possibility that VML and vastus intermedius may have been misidentified in some previous studies. [Conclusions] In conclusion the VML/VMO division (when present) is much more proximal than has been previously reported, and there is no consistent pattern of innervation to the two parts of the muscle. Key words: Fibre angle, Vastus medialis, VML, VMO (This article was submitted Nov. -

The Effect of Stretching Hamstring, Gastrocnemius, Iliopsoas
Open Journal of Therapy and Rehabilitation, 2015, 3, 139-145 Published Online November 2015 in SciRes. http://www.scirp.org/journal/ojtr http://dx.doi.org/10.4236/ojtr.2015.34019 The Effect of Stretching Hamstring, Gastrocnemius, Iliopsoas and Back Muscles on Pain and Functional Activities in Patients with Chronic Low Back Pain: A Randomized Clinical Trial Hamada E. Seif1, Aqeel Alenazi2, Sahar Mahmoud Hassan1, Shaji John Kachanathu3, Ashraf R. Hafez1* 1Cairo UniversityHospital, Cairo University, Cairo, Egypt 2Physical Therapy and Rehabilitation Department, College of Applied Medical Sciences, Salman Bin Abdulaziz University, Alkharj, Saudi Arabia 3Collage of Applied Medical Sciences, King Saud University, Riyadh, Saudi Arabia Received 15 September 2015; accepted 6 November 2015; published 9 November 2015 Copyright © 2015 by authors and Scientific Research Publishing Inc. This work is licensed under the Creative Commons Attribution International License (CC BY). http://creativecommons.org/licenses/by/4.0/ Abstract A back pain lasting more than 12 weeks has been defined as a chronic low back pain (LBP) [1]. More than half of people suffer from LBP [1]. The purpose of this study was to examine the effect of gastrocnemius muscle stretching in the treatment of chronic low back pain. Methods: Forty pa- tients with chronic low back pain, ages ranging from 25 to 40 years, were recruited and divided randomly into two groups. The control group followed a physical therapy program that included stretching exercises for back, hamstring and iliopsoas muscles. Strengthening exercises for abdo- minal muscle and postural instructions for activities of daily living were also performed. The ex- perimental group followed the same control-group exercises with the addition of stretching exer- cises for gastrocnemius muscles. -

Changes in the Quadriceps-To-Hamstring Muscle
Original Article https://doi.org/10.14474/ptrs.2019.8.1.45 Phys Ther Rehabil Sci pISSN 2287-7576 2019, 8 (1), 45-51 eISSN 2287-7584 www.jptrs.org Changes in the quadriceps-to-hamstring muscle ratio http://crossmark.crossref.org/dialog/?doi=10.14474/ptrs.2019.8.1.45&domain=pdf&date_stamp=2019-3-25 during wall squatting according to the straight leg raise test angle Jaeeun Kima, HyeonA Kimb, JuYeong Leeb, HoYoung Leeb, Hyoseung Jungb, YunKi Chob, HyeMin Choib, Donghyun Yic, Daewon Kangc, Jongeun Yimb,c a Department of Physical Therapy, Barosun Hospital, Seoul, Republic of Korea b Department of Physical Therapy, College of Health and Welfare, Sahmyook University, Seoul, Republic of Korea c Department of Physical Therapy, The Graduate School, Sahmyook University, Seoul, Republic of Korea Objective: The purpose of this study was to investigate the muscle activity ratio of the lower limb according to changes in straight leg raise (SLR) test angles on hamstring muscle shortening during squat exercises. Design: Randomized controlled trial. Methods: The subjects were 14 healthy adults who were informed of and agreed to the method and purpose of the study. The par- ticipants were classified into SLR groups according to two angles (over 80° or under 80°) assessed using the SLR tests. After train- ing and practicing the wall squat posture to be applied to the experiment, electromyography (EMG) was used to measure changes in muscle activity during the performance of a wall squat. After stretching, a sequence of pre-stretch tests were performed again, and the active and passive SLR tests were also reconducted; thereafter, a wall squat was performed again by attaching EMG electrodes. -

Muscle Activation and Kinematic Analysis During the Inclined Leg Press Exercise in Young Females
International Journal of Environmental Research and Public Health Article Muscle Activation and Kinematic Analysis during the Inclined Leg Press Exercise in Young Females Isabel Martín-Fuentes 1 , José M. Oliva-Lozano 1 and José M. Muyor 1,2,* 1 Health Research Centre, University of Almería, 04120 Almería, Spain; [email protected] (I.M.-F.); [email protected] (J.M.O.-L.) 2 Laboratory of Kinesiology, Biomechanics and Ergonomics (KIBIOMER Lab.), Research Central Services, University of Almería, 04120 Almería, Spain * Correspondence: [email protected]; Tel.: +34-950214429 Received: 24 October 2020; Accepted: 20 November 2020; Published: 23 November 2020 Abstract: Knee joint muscle activation imbalances, especially weakness in the vastus medialis oblique, are related to patellofemoral pain within the female population. The available literature presents the leg press as an exercise which potentially targets vastus medialis oblique activation, thus reducing imbalances in the quadriceps muscles. The main aim of the present study was to compare thigh muscle activation and kinematic parameters under different conditions during the inclined leg press exercise in a young female population. A cross-sectional study was conducted on 10 young, trained females. Muscle activation of the vastus medialis oblique, vastus lateralis, rectus femoris and gluteus medialis was analyzed under five different inclined leg press conditions, modifying the feet rotation (0–45◦ external rotation) and the stance width (100–150% hip width) on the footplate. All the conditions were performed at two different movement velocities: controlled velocity (2” eccentric–2” concentric) and maximal intended velocity. Mean propulsive velocity, maximum velocity and maximum power were also assessed. The results show that both controlled velocity conditions and maximal intended velocity conditions elicited a similar muscle activation pattern with greater activation during the concentric phase (p < 0.001, ηp2 = 0.96). -

The Role of and Relationship Between Hamstring and Quadriceps Muscle Myofascial Trigger Points in Patients with Patellofemoral Pain Syndrome
The role of and relationship between Hamstring and Quadriceps muscle myofascial trigger points in patients with patellofemoral pain syndrome. By Karen Louise Frandsen Smith A mini-dissertation submitted in partial compliance with the requirements for the Master’s Degree in Technology: Chiropractic at the Durban University of Technology I, Karen Louise Frandsen Smith, declare that this dissertation is representative of my own work in both conception and execution (except where acknowledgements indicate to the contrary). _______________________ ________________ Karen Louise Frandsen Smith Date Approved for Submission _______________________ ________________ Dr Brian Kruger (supervisor) Date M. Tech. Chiro., C.C.S.P. Dedication I dedicate this work to everyone who loves me and have supported me throughout these years of studying and all the difficult times. It is thanks to you that I have reached my dream. Dad, you would be so proud. i Acknowledgements Thank you to the DUT staff, patients, and supervisor, Dr Brian Kruger for making this happen. Thanks to my class mates for making the years fly by, and creating lifelong memories. This dissertation would not have been completed without supportive, generous and helpful people. Special thanks to you, Dr Danella Lubbe for motivating me and thank you so much Dr Charmaine Korporaal for “picking me up” and helping me finish. Endless gratitude goes to you Damon, Cherine, Mom and Viggo. You are / were all my anchors in the storm, and without your belief in me, I would be nowhere. ii Abstract Purpose: Patellofemoral Pain Syndrome is a common condition in all age groups, with a multi- factorial etiology. -

Muscle Length of the Hamstrings Using Ultrasonography Versus Musculoskeletal Modelling
Journal of Functional Morphology and Kinesiology Article Muscle Length of the Hamstrings Using Ultrasonography Versus Musculoskeletal Modelling Eleftherios Kellis * , Athina Konstantinidou and Athanasios Ellinoudis Laboratory of Neuromechanics, Department of Physical Education and Sport Sciences at Serres, Aristotle University of Thessaloniki, 62100 Serres, Greece; [email protected] (A.K.); [email protected] (A.E.) * Correspondence: [email protected] Abstract: Muscle morphology is an important contributor to hamstring muscle injury and malfunc- tion. The aim of this study was to examine if hamstring muscle-tendon lengths differ between various measurement methods as well as if passive length changes differ between individual hamstrings. The lengths of biceps femoris long head (BFlh), semimembranosus (SM), and semitendinosus (ST) of 12 healthy males were determined using three methods: Firstly, by identifying the muscle attach- ments using ultrasound (US) and then measuring the distance on the skin using a flexible ultrasound tape (TAPE-US). Secondly, by scanning each muscle using extended-field-of view US (EFOV-US) and, thirdly, by estimating length using modelling equations (MODEL). Measurements were performed with the participant relaxed at six combinations of hip (0◦, 90◦) and knee (0◦, 45◦, and 90◦) flexion angles. The MODEL method showed greater BFlh and SM lengths as well as changes in length than US methods. EFOV-US showed greater ST and SM lengths than TAPE-US (p < 0.05). SM length change across all joint positions was greater than BFlh and ST (p < 0.05). Hamstring length predicted using regression equations is greater compared with those measured using US-based methods. The EFOV-US method yielded greater ST and SM length than the TAPE-US method. -

Quadriceps and Hamstrings Coactivation in Exercises Used in Prevention And
bioRxiv preprint doi: https://doi.org/10.1101/574210; this version posted March 11, 2019. The copyright holder for this preprint (which was not certified by peer review) is the author/funder, who has granted bioRxiv a license to display the preprint in perpetuity. It is made available under aCC-BY 4.0 International license. 1 Quadriceps and hamstrings coactivation in exercises used in prevention and 2 rehabilitation of hamstring strain injury in young soccer players 3 Gonzalo Torres1¶; David Chorro1¶; Archit Navandar2¶; Javier Rueda1¶; Luís 4 Fernández3¶; Enrique Navarro1*¶ 5 6 1Faculty of Sport Sciences, Universidad Politécnica de Madrid, Madrid, Madrid, Spain. 7 2Faculty of Sport Sciences, Universidad Europea de Madrid, Madrid, Madrid, Spain. 8 3Medical Services of Atlético de Madrid, Club Atlético de Madrid, Madrid, Madrid, 9 Spain. 10 11 12 *Corresponding author 13 E-mail: [email protected] (EN) 14 15 ¶These authors contributed equally to this work. 16 17 18 19 20 21 22 23 24 bioRxiv preprint doi: https://doi.org/10.1101/574210; this version posted March 11, 2019. The copyright holder for this preprint (which was not certified by peer review) is the author/funder, who has granted bioRxiv a license to display the preprint in perpetuity. It is made available under aCC-BY 4.0 International license. 25 Abstract 26 This study aimed to study the co-activation of hamstring-quadriceps muscles 27 during submaximal strength exercises without the use of maximum voluntary isometric 28 contraction testing and compare (i) the inter-limb differences in muscle activation, (ii) the 29 intra-muscular group activation pattern, and (iii) the activation during different phases of 30 the exercise. -
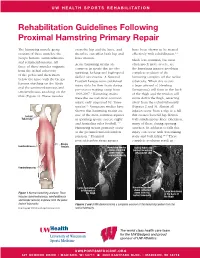
Rehabilitation Guidelines Following Proximal Hamstring Primary Repair
UW HEALTH SPORTS REHABILITATION Rehabilitation Guidelines Following Proximal Hamstring Primary Repair The hamstring muscle group cross the hip and the knee, and have been shown to be treated consists of three muscles: the therefore can affect both hip and effectively with rehabilitation.1, 8 biceps femoris, semitendinosus knee motion. Much less common, but most and semimembranosus. All Acute hamstring strains are often much more severe, are three of these muscles originate common in sports that involve the hamstring injuries involving from the ischial tuberosity sprinting, kicking and high-speed complete avulsion of the of the pelvis and then insert skilled movements. A National hamstring complex off the ischial below the knee with the biceps Football League team published tuberosity. When this occurs femoris attaching on the fibula injury data for their team during a large amount of bleeding and the semimembranosus and pre-season training camp from (hematoma) will form in the back semitendinosus attaching on the 1998-2007.1 Hamstring strains of the thigh and the tendon will tibia (Figure 1). These muscles were the second most common move down the thigh, retracting injury, only surpassed by “knee away from the ischial tuberosity sprains”.1 Numerous studies have (Figures 2 and 3). Almost all shown that hamstring strains are injuries occur from a slip or a fall Ischial one of the most common injuries that creates forceful hip flexion Tuberosity in sprinting sports, soccer, rugby with simultaneous knee extension, and Australian rules football.1-12 -

Hamstring Muscle Injuries
DISEASES & CONDITIONS Hamstring Muscle Injuries Hamstring muscle injuries — such as a "pulled hamstring" — occur frequently in athletes. They are especially common in athletes who participate in sports that require sprinting, such as track, soccer, and basketball. A pulled hamstring or strain is an injury to one or more of the muscles at the back of the thigh. Most hamstring injuries respond well to simple, nonsurgical treatments. Anatomy The hamstring muscles run down the back of the thigh. There are three hamstring muscles: Semitendinosus Semimembranosus Biceps femoris They start at the bottom of the pelvis at a place called the ischial tuberosity. They cross the knee joint and end at the lower leg. Hamstring muscle fibers join with the tough, connective tissue of the hamstring tendons near the points where the tendons attach to bones. The hamstring muscle group helps you extend your leg straight back and bend your knee. Normal hamstring anatomy. The three hamstring muscles start at the bottom of the pelvis and end near the top of the lower leg. Description A hamstring strain can be a pull, a partial tear, or a complete tear. Muscle strains are graded according to their severity. A grade 1 strain is mild and usually heals readily; a grade 3 strain is a complete tear of the muscle that may take months to heal. Most hamstring injuries occur in the thick, central part of the muscle or where the muscle fibers join tendon fibers. In the most severe hamstring injuries, the tendon tears completely away from the bone. It may even pull a piece of bone away with it. -

Chapter 10 the Knee Joint
The Knee Joint • Knee joint – largest joint in body Chapter 10 – very complex The Knee Joint – primarily a hinge joint Manual of Structural Kinesiology Modified for Prentice WE: Arnheim’s principles of athletic training , ed 12, New R.T. Floyd, EdD, ATC, CSCS York, 2006, McGraw-Hill; from Saladin, KS: Anatomy &physiology: the unity of forms and function , ed 2, New York, 2001, McGraw- Hill. © 2007 McGraw-Hill Higher Education. All rights reserved. 10-1 © 2007 McGraw-Hill Higher Education. All rights reserved. 10-2 Bones Bones • Enlarged femoral condyles articulate on • Fibula - lateral enlarged tibial condyles – serves as the attachment for • Medial & lateral tibial condyles (medial & knee joint lateral tibial plateaus) - receptacles for structures femoral condyles – does not articulate • Tibia – medial with femur or patella – bears most of weight – not part of knee joint Modified from Anthony CP, Kolthoff NJ: Textbook of anatomy and physiology , ed 9, St. Louis, 1975, Mosby. © 2007 McGraw-Hill Higher Education. All rights reserved. 10-3 © 2007 McGraw-Hill Higher Education. All rights reserved. 10-4 Bones Bones • Patella • Key bony landmarks – sesamoid (floating) bone – Superior & inferior patellar poles – imbedded in quadriceps – Tibial tuberosity & patellar tendon – Gerdy’s tubercle – serves similar to a pulley – Medial & lateral femoral in improving angle of condyles pull, resulting in greater – Upper anterior medial tibial mechanical advantage in surface – Head of fibula knee extension Modified from Anthony CP, Kolthoff NJ: Textbook of anatomy and physiology , ed 9, St. Louis, 1975, Mosby. © 2007 McGraw-Hill Higher Education. All rights reserved. 10-5 © 2007 McGraw-Hill Higher Education. All rights reserved. -

Back of Thigh
Back of thigh Dr Garima Sehgal Associate Professor King George’s Medical University UP, Lucknow DISCLAIMER: • The presentation includes images which have been taken from google images or books. • The author of the presentation claims no personal ownership over these images taken from books or google images. • They are being used in the presentation only for educational purpose. Learning Objectives By the end of this teaching session on back of thigh – I all the MBBS 1st year students must be able to: • Enumerate the contents of posterior compartment of thigh • Describe the cutaneous innervation of skin of back of thigh • Enumerate the hamstring muscles • List the criteria for inclusion of muscles as hamstring muscles • Describe the origin, insertion, nerve supply & actions of hamstring muscles • Describe origin, course and branches of sciatic nerve • Write a short note on posterior cutaneous nerve of thigh • Write a note on arteries and arterial anastomosis at the back of thigh • Discuss applied anatomy of back of thigh Compartments of the thigh Cutaneous innervation of back of thigh 1 4 2 3 Contents of Back of thigh Muscles: Hamstring muscles & short head of biceps femoris Nerves: Sciatic nerve & posterior cutaneous nerve of thigh Arterial Anastomosis: The Posterior Femoral Cutaneous Nerve (n. cutaneus femoralis posterior) Dorsal divisions of – S1, S2 & Ventral divisions of S2, S3 Exits from pelvis through the greater sciatic foramen below the Piriformis. Descends beneath the Gluteus maximus with the inferior gluteal artery Runs down the back of the thigh beneath the fascia lata, to the back of the knee and leg Posterior Femoral Cutaneous Nerve contd….