Utilization of L-Methionine and S-Adenosyl-L-Methionine for Methylation of Soluble RNA by Mouse Liver and Hepatoma Extracts1
Total Page:16
File Type:pdf, Size:1020Kb

Load more
Recommended publications
-

A Disease Spectrum for ITPA Variation: Advances in Biochemical and Clinical Research Nicholas E
Burgis Journal of Biomedical Science (2016) 23:73 DOI 10.1186/s12929-016-0291-y REVIEW Open Access A disease spectrum for ITPA variation: advances in biochemical and clinical research Nicholas E. Burgis Abstract Human ITPase (encoded by the ITPA gene) is a protective enzyme which acts to exclude noncanonical (deoxy) nucleoside triphosphates ((d)NTPs) such as (deoxy)inosine 5′-triphosphate ((d)ITP), from (d)NTP pools. Until the last few years, the importance of ITPase in human health and disease has been enigmatic. In 2009, an article was published demonstrating that ITPase deficiency in mice is lethal. All homozygous null offspring died before weaning as a result of cardiomyopathy due to a defect in the maintenance of quality ATP pools. More recently, a whole exome sequencing project revealed that very rare, severe human ITPA mutation results in early infantile encephalopathy and death. It has been estimated that nearly one third of the human population has an ITPA status which is associated with decreased ITPase activity. ITPA status has been linked to altered outcomes for patients undergoing thiopurine or ribavirin therapy. Thiopurine therapy can be toxic for patients with ITPA polymorphism, however, ITPA polymorphism is associated with improved outcomes for patients undergoing ribavirin treatment. ITPA polymorphism has also been linked to early-onset tuberculosis susceptibility. These data suggest a spectrum of ITPA-related disease exists in human populations. Potentially, ITPA status may affect a large number of patient outcomes, suggesting that modulation of ITPase activity is an important emerging avenue for reducing the number of negative outcomes for ITPA-related disease. -

Nucleotide Metabolism 22
Nucleotide Metabolism 22 For additional ancillary materials related to this chapter, please visit thePoint. I. OVERVIEW Ribonucleoside and deoxyribonucleoside phosphates (nucleotides) are essential for all cells. Without them, neither ribonucleic acid (RNA) nor deoxyribonucleic acid (DNA) can be produced, and, therefore, proteins cannot be synthesized or cells proliferate. Nucleotides also serve as carriers of activated intermediates in the synthesis of some carbohydrates, lipids, and conjugated proteins (for example, uridine diphosphate [UDP]-glucose and cytidine diphosphate [CDP]- choline) and are structural components of several essential coenzymes, such as coenzyme A, flavin adenine dinucleotide (FAD[H2]), nicotinamide adenine dinucleotide (NAD[H]), and nicotinamide adenine dinucleotide phosphate (NADP[H]). Nucleotides, such as cyclic adenosine monophosphate (cAMP) and cyclic guanosine monophosphate (cGMP), serve as second messengers in signal transduction pathways. In addition, nucleotides play an important role as energy sources in the cell. Finally, nucleotides are important regulatory compounds for many of the pathways of intermediary metabolism, inhibiting or activating key enzymes. The purine and pyrimidine bases found in nucleotides can be synthesized de novo or can be obtained through salvage pathways that allow the reuse of the preformed bases resulting from normal cell turnover. [Note: Little of the purines and pyrimidines supplied by diet is utilized and is degraded instead.] II. STRUCTURE Nucleotides are composed of a nitrogenous base; a pentose monosaccharide; and one, two, or three phosphate groups. The nitrogen-containing bases belong to two families of compounds: the purines and the pyrimidines. A. Purine and pyrimidine bases Both DNA and RNA contain the same purine bases: adenine (A) and guanine (G). -

Inosine in Biology and Disease
G C A T T A C G G C A T genes Review Inosine in Biology and Disease Sundaramoorthy Srinivasan 1, Adrian Gabriel Torres 1 and Lluís Ribas de Pouplana 1,2,* 1 Institute for Research in Biomedicine, Barcelona Institute of Science and Technology, 08028 Barcelona, Catalonia, Spain; [email protected] (S.S.); [email protected] (A.G.T.) 2 Catalan Institution for Research and Advanced Studies, 08010 Barcelona, Catalonia, Spain * Correspondence: [email protected]; Tel.: +34-934034868; Fax: +34-934034870 Abstract: The nucleoside inosine plays an important role in purine biosynthesis, gene translation, and modulation of the fate of RNAs. The editing of adenosine to inosine is a widespread post- transcriptional modification in transfer RNAs (tRNAs) and messenger RNAs (mRNAs). At the wobble position of tRNA anticodons, inosine profoundly modifies codon recognition, while in mRNA, inosines can modify the sequence of the translated polypeptide or modulate the stability, localization, and splicing of transcripts. Inosine is also found in non-coding and exogenous RNAs, where it plays key structural and functional roles. In addition, molecular inosine is an important secondary metabolite in purine metabolism that also acts as a molecular messenger in cell signaling pathways. Here, we review the functional roles of inosine in biology and their connections to human health. Keywords: inosine; deamination; adenosine deaminase acting on RNAs; RNA modification; translation Citation: Srinivasan, S.; Torres, A.G.; Ribas de Pouplana, L. Inosine in 1. Introduction Biology and Disease. Genes 2021, 12, 600. https://doi.org/10.3390/ Inosine was one of the first nucleobase modifications discovered in nucleic acids, genes12040600 having been identified in 1965 as a component of the first sequenced transfer RNA (tRNA), tRNAAla [1]. -

Central Nervous System Dysfunction and Erythrocyte Guanosine Triphosphate Depletion in Purine Nucleoside Phosphorylase Deficiency
Arch Dis Child: first published as 10.1136/adc.62.4.385 on 1 April 1987. Downloaded from Archives of Disease in Childhood, 1987, 62, 385-391 Central nervous system dysfunction and erythrocyte guanosine triphosphate depletion in purine nucleoside phosphorylase deficiency H A SIMMONDS, L D FAIRBANKS, G S MORRIS, G MORGAN, A R WATSON, P TIMMS, AND B SINGH Purine Laboratory, Guy's Hospital, London, Department of Immunology, Institute of Child Health, London, Department of Paediatrics, City Hospital, Nottingham, Department of Paediatrics and Chemical Pathology, National Guard King Khalid Hospital, Jeddah, Saudi Arabia SUMMARY Developmental retardation was a prominent clinical feature in six infants from three kindreds deficient in the enzyme purine nucleoside phosphorylase (PNP) and was present before development of T cell immunodeficiency. Guanosine triphosphate (GTP) depletion was noted in the erythrocytes of all surviving homozygotes and was of equivalent magnitude to that found in the Lesch-Nyhan syndrome (complete hypoxanthine-guanine phosphoribosyltransferase (HGPRT) deficiency). The similarity between the neurological complications in both disorders that the two major clinical consequences of complete PNP deficiency have differing indicates copyright. aetiologies: (1) neurological effects resulting from deficiency of the PNP enzyme products, which are the substrates for HGPRT, leading to functional deficiency of this enzyme. (2) immunodeficiency caused by accumulation of the PNP enzyme substrates, one of which, deoxyguanosine, is toxic to T cells. These studies show the need to consider PNP deficiency (suggested by the finding of hypouricaemia) in patients with neurological dysfunction, as well as in T cell immunodeficiency. http://adc.bmj.com/ They suggest an important role for GTP in normal central nervous system function. -

Standard Abbreviations
Journal of CancerJCP Prevention Standard Abbreviations Journal of Cancer Prevention provides a list of standard abbreviations. Standard Abbreviations are defined as those that may be used without explanation (e.g., DNA). Abbreviations not on the Standard Abbreviations list should be spelled out at first mention in both the abstract and the text. Abbreviations should not be used in titles; however, running titles may carry abbreviations for brevity. ▌Abbreviations monophosphate ADP, dADP adenosine diphosphate, deoxyadenosine IR infrared diphosphate ITP, dITP inosine triphosphate, deoxyinosine AMP, dAMP adenosine monophosphate, deoxyadenosine triphosphate monophosphate LOH loss of heterozygosity ANOVA analysis of variance MDR multiple drug resistance AP-1 activator protein-1 MHC major histocompatibility complex ATP, dATP adenosine triphosphate, deoxyadenosine MRI magnetic resonance imaging trip hosphate mRNA messenger RNA bp base pair(s) MTS 3-(4,5-dimethylthiazol-2-yl)-5-(3- CDP, dCDP cytidine diphosphate, deoxycytidine diphosphate carboxymethoxyphenyl)-2-(4-sulfophenyl)- CMP, dCMP cytidine monophosphate, deoxycytidine mono- 2H-tetrazolium phosphate mTOR mammalian target of rapamycin CNBr cyanogen bromide MTT 3-(4,5-Dimethylthiazol-2-yl)-2,5- cDNA complementary DNA diphenyltetrazolium bromide CoA coenzyme A NAD, NADH nicotinamide adenine dinucleotide, reduced COOH a functional group consisting of a carbonyl and nicotinamide adenine dinucleotide a hydroxyl, which has the formula –C(=O)OH, NADP, NADPH nicotinamide adnine dinucleotide -

And Triphosphate from Royal Jelly Using Liquid Chromatography - Tandem Mass Spectrometry," Journal of Food and Drug Analysis: Vol
Volume 28 Issue 3 Article 2 2020 Quantification of Adenosine Mono-, Di- and riphosphateT from Royal Jelly using Liquid Chromatography - Tandem Mass Spectrometry Follow this and additional works at: https://www.jfda-online.com/journal Part of the Food Science Commons, Medicinal Chemistry and Pharmaceutics Commons, Pharmacology Commons, and the Toxicology Commons This work is licensed under a Creative Commons Attribution-Noncommercial-No Derivative Works 4.0 License. Recommended Citation Liao, Wan-Rou; Huang, Jen-Pang; and Chen, Sung-Fang (2020) "Quantification of Adenosine Mono-, Di- and Triphosphate from Royal Jelly using Liquid Chromatography - Tandem Mass Spectrometry," Journal of Food and Drug Analysis: Vol. 28 : Iss. 3 , Article 2. Available at: https://doi.org/10.38212/2224-6614.1007 This Original Article is brought to you for free and open access by Journal of Food and Drug Analysis. It has been accepted for inclusion in Journal of Food and Drug Analysis by an authorized editor of Journal of Food and Drug Analysis. Quantification of adenosine Mono-, Di- and triphosphate from royal jelly using liquid chromatography - Tandem mass spectrometry ORIGINAL ARTICLE Wan-Rou Liao a, Jen-Pang Huang b, Sung-Fang Chen a,* a Department of Chemistry, National Taiwan Normal University, Taipei, Taiwan b MSonline Scientific Co., Ltd., Taipei, Taiwan Abstract Nucleotides are composed of nitrogen bases, ribose units and phosphate groups. Adenine (Ade), adenosine mono- phosphate (AMP), adenosine diphosphate (ADP) and adenosine triphosphate (ATP) all play important roles in physio- logical metabolism. Royal jelly, a secretion produced by worker bees, contains a variety of natural ingredients and several studies have shown that royal jelly can serve as a source of nutrition for humans. -

Epistasis-Driven Identification of SLC25A51 As a Regulator of Human
ARTICLE https://doi.org/10.1038/s41467-020-19871-x OPEN Epistasis-driven identification of SLC25A51 as a regulator of human mitochondrial NAD import Enrico Girardi 1, Gennaro Agrimi 2, Ulrich Goldmann 1, Giuseppe Fiume1, Sabrina Lindinger1, Vitaly Sedlyarov1, Ismet Srndic1, Bettina Gürtl1, Benedikt Agerer 1, Felix Kartnig1, Pasquale Scarcia 2, Maria Antonietta Di Noia2, Eva Liñeiro1, Manuele Rebsamen1, Tabea Wiedmer 1, Andreas Bergthaler1, ✉ Luigi Palmieri2,3 & Giulio Superti-Furga 1,4 1234567890():,; About a thousand genes in the human genome encode for membrane transporters. Among these, several solute carrier proteins (SLCs), representing the largest group of transporters, are still orphan and lack functional characterization. We reasoned that assessing genetic interactions among SLCs may be an efficient way to obtain functional information allowing their deorphanization. Here we describe a network of strong genetic interactions indicating a contribution to mitochondrial respiration and redox metabolism for SLC25A51/MCART1, an uncharacterized member of the SLC25 family of transporters. Through a combination of metabolomics, genomics and genetics approaches, we demonstrate a role for SLC25A51 as enabler of mitochondrial import of NAD, showcasing the potential of genetic interaction- driven functional gene deorphanization. 1 CeMM Research Center for Molecular Medicine of the Austrian Academy of Sciences, Vienna, Austria. 2 Laboratory of Biochemistry and Molecular Biology, Department of Biosciences, Biotechnologies and Biopharmaceutics, -

High-Purity Rebaudioside D and Applications Hochreines Rebaudiosid-D Und Anwendungen Rébaudioside D De Grande Pureté Et Applications
(19) TZZ Z_T (11) EP 2 708 548 B1 (12) EUROPEAN PATENT SPECIFICATION (45) Date of publication and mention (51) Int Cl.: of the grant of the patent: C07H 1/08 (2006.01) A21D 2/36 (2006.01) 06.12.2017 Bulletin 2017/49 A23G 1/42 (2006.01) (21) Application number: 13196410.8 (22) Date of filing: 13.10.2010 (54) High-Purity Rebaudioside D and Applications Hochreines Rebaudiosid-D und Anwendungen Rébaudioside D de grande pureté et applications (84) Designated Contracting States: (72) Inventors: AL AT BE BG CH CY CZ DE DK EE ES FI FR GB • Abelyan, Varuzhan GR HR HU IE IS IT LI LT LU LV MC MK MT NL NO 50480 Kuala Lumpur (MY) PL PT RO RS SE SI SK SM TR • Markosyan, Avetik 59200 Kuala Lumpur (MY) (30) Priority: 15.10.2009 US 580233 • Abelyan, Lidia 24.05.2010 US 785501 50480 Kuala Lumpur (MY) 24.05.2010 US 785504 24.05.2010 US 785506 (74) Representative: Hocking, Adrian Niall et al 24.05.2010 US 785507 Albright IP Limited 24.05.2010 US 785508 County House 24.05.2010 US 786392 Bayshill Road 24.05.2010 US 786402 Cheltenham, Glos. GL50 3BA (GB) 24.05.2010 US 786413 24.05.2010 US 786416 (56) References cited: 24.05.2010 US 786427 WO-A1-2009/071277 24.05.2010 US 786430 24.05.2010 US 786419 • I. SAKAMOTO ET AL: "Application of 13C NMR spectroscopy to chemistry of natural glycosices: (43) Date of publication of application: rebaudioside-C,a newsweet diterpeneglycoside 19.03.2014 Bulletin 2014/12 of Stevia rebaudiana", CHEM. -

Adenosine Diphosphate Glucose Pyrophosphatase: a Plastidial Phosphodiesterase That Prevents Starch Biosynthesis
Adenosine diphosphate glucose pyrophosphatase: A plastidial phosphodiesterase that prevents starch biosynthesis Milagros Rodrı´guez-Lo´ pez, Edurne Baroja-Ferna´ ndez, Aitor Zandueta-Criado, and Javier Pozueta-Romero* Instituto de Agrobiotecnologı´ay Recursos Naturales, Universidad Pu´blica de Navarra ͞Consejo Superior de Investigaciones Cientı´ficas,Carretera de Mutilva s͞n, Mutilva Baja, 31192 Navarra, Spain Communicated by Andre´T. Jagendorf, Cornell University, Ithaca, NY, April 13, 2000 (received for review November 28, 1999) A distinct phosphodiesterasic activity (EC 3.1.4) was found in both their possible occurrence in several plant species. As a result, we mono- and dicotyledonous plants that catalyzes the hydrolytic have now found a phosphodiesterasic activity that catalyzes the breakdown of ADPglucose (ADPG) to produce equimolar amounts hydrolytic breakdown of ADPG. In this paper, we report of glucose-1-phosphate and AMP. The enzyme responsible for this the subcellular localization and biochemical characterization of activity, referred to as ADPG pyrophosphatase (AGPPase), was the enzyme responsible for this activity, referred to as ADPG purified over 1,100-fold from barley leaves and subjected to pyrophosphatase (AGPPase).† Based on the results presented in biochemical characterization. The calculated Keq (modified equi- this work using different plant sources, we discuss that AGPPase librium constant) value for the ADPG hydrolytic reaction at pH 7.0 may be involved in controlling the intracellular levels of ADPG and 25°C is 110, and its standard-state free-energy change value linked to starch biosynthesis. kJ). Kinetic analyses showed 4.18 ؍ G)is؊2.9 kcal͞mol (1 kcal⌬) that, although AGPPase can hydrolyze several low-molecular Materials and Methods weight phosphodiester bond-containing compounds, ADPG Plant Material. -

Role of Inosine Triphosphate Pyrophosphatase Gene Variant on Fever Incidence During Zidovudine Antiretroviral Therapy
Role of inosine triphosphate pyrophosphatase gene variant on fever incidence during zidovudine antiretroviral therapy A.V.C. Coelho1, S.P.S. Silva2, L. Zandonà3, G. Stocco3, G. Decorti3 and S. Crovella1,4 1Departamento de Genética, Universidade Federal de Pernambuco Federal de Pernambuco, Recife, PE, Brasil 2Programa de Pós-Graduação em Inovação Terapêutica, Universidade Federal de Pernambuco Federal de Pernambuco, Recife, PE, Brasil 3Department of Life Sciences, University of Trieste, Trieste, Italy 4Institute for Maternal and Child Health, Scientific Institute for Research, Hospitalization and Care Burlo Garofolo, Trieste, Italy Corresponding author: A.V.C. Coelho E-mail: [email protected] / [email protected] Genet. Mol. Res. 16 (1): gmr16019373 Received September 22, 2016 Accepted November 18, 2016 Published January 23, 2017 DOI http://dx.doi.org/10.4238/gmr16019373 Copyright © 2017 The Authors. This is an open-access article distributed under the terms of the Creative Commons Attribution ShareAlike (CC BY-SA) 4.0 License. ABSTRACT. Zidovudine, the antiretroviral drug used to treat HIV infection, commonly causes adverse effects, such as systemic fever and gastrointestinal alterations. In the present study, the potential role of inosine triphosphate pyrophosphatase (ITPA) gene variant on the incidence of adverse events during antiretroviral therapy (ART) of HIV with zidovudine was discussed. Individuals from Northeastern Brazil (N = 204) receiving treatment for HIV-1 infection were recruited. Zidovudine-related adverse effects developed during the treatment were registered. The rs1127354 polymorphism in the ITPA gene was Genetics and Molecular Research 16 (1): gmr16019373 A.V.C. Coelho et al. 2 genotyped using real-time PCR to assess whether this single nucleotide polymorphism was associated with the occurrence of zidovudine- related adverse effects. -
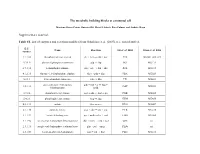
The Metabolic Building Blocks of a Minimal Cell Supplementary
The metabolic building blocks of a minimal cell Mariana Reyes-Prieto, Rosario Gil, Mercè Llabrés, Pere Palmer and Andrés Moya Supplementary material. Table S1. List of enzymes and reactions modified from Gabaldon et. al. (2007). n.i.: non identified. E.C. Name Reaction Gil et. al. 2004 Glass et. al. 2006 number 2.7.1.69 phosphotransferase system glc + pep → g6p + pyr PTS MG041, 069, 429 5.3.1.9 glucose-6-phosphate isomerase g6p ↔ f6p PGI MG111 2.7.1.11 6-phosphofructokinase f6p + atp → fbp + adp PFK MG215 4.1.2.13 fructose-1,6-bisphosphate aldolase fbp ↔ gdp + dhp FBA MG023 5.3.1.1 triose-phosphate isomerase gdp ↔ dhp TPI MG431 glyceraldehyde-3-phosphate gdp + nad + p ↔ bpg + 1.2.1.12 GAP MG301 dehydrogenase nadh 2.7.2.3 phosphoglycerate kinase bpg + adp ↔ 3pg + atp PGK MG300 5.4.2.1 phosphoglycerate mutase 3pg ↔ 2pg GPM MG430 4.2.1.11 enolase 2pg ↔ pep ENO MG407 2.7.1.40 pyruvate kinase pep + adp → pyr + atp PYK MG216 1.1.1.27 lactate dehydrogenase pyr + nadh ↔ lac + nad LDH MG460 1.1.1.94 sn-glycerol-3-phosphate dehydrogenase dhp + nadh → g3p + nad GPS n.i. 2.3.1.15 sn-glycerol-3-phosphate acyltransferase g3p + pal → mag PLSb n.i. 2.3.1.51 1-acyl-sn-glycerol-3-phosphate mag + pal → dag PLSc MG212 acyltransferase 2.7.7.41 phosphatidate cytidyltransferase dag + ctp → cdp-dag + pp CDS MG437 cdp-dag + ser → pser + 2.7.8.8 phosphatidylserine synthase PSS n.i. cmp 4.1.1.65 phosphatidylserine decarboxylase pser → peta PSD n.i. -

Coupled Nucleoside Phosphorylase Reactions in Escherichia Coli John Lewis Ott Iowa State College
Iowa State University Capstones, Theses and Retrospective Theses and Dissertations Dissertations 1956 Coupled nucleoside phosphorylase reactions in Escherichia coli John Lewis Ott Iowa State College Follow this and additional works at: https://lib.dr.iastate.edu/rtd Part of the Biochemistry Commons, and the Microbiology Commons Recommended Citation Ott, John Lewis, "Coupled nucleoside phosphorylase reactions in Escherichia coli " (1956). Retrospective Theses and Dissertations. 13758. https://lib.dr.iastate.edu/rtd/13758 This Dissertation is brought to you for free and open access by the Iowa State University Capstones, Theses and Dissertations at Iowa State University Digital Repository. It has been accepted for inclusion in Retrospective Theses and Dissertations by an authorized administrator of Iowa State University Digital Repository. For more information, please contact [email protected]. NOTE TO USERS This reproduction is the best copy available. UMI COUPLED NUCLEOSIDE PHOSPHORYLASE REACTIONS IN ESCHERICHIA COLI / by John Lewis Ott A Dissertation Submitted to the Graduate Faculty in Partial Fulfillment of The Requirements for the Degree of DOCTOR OF PHILOSOPHY Major Subject: Physlolgglcal Bacteriology Approved: Signature was redacted for privacy. In Charge of Major Work Signature was redacted for privacy. Head of Major Department Signature was redacted for privacy. Dean of Graduate College Iowa State College 1956 UMI Number: DP12892 INFORMATION TO USERS The quality of this reproduction is dependent upon the quality of the copy submitted. Broken or indistinct print, colored or poor quality illustrations and photographs, print bleed-through, substandard margins, and improper alignment can adversely affect reproduction. In the unlikely event that the author did not send a complete manuscript and there are missing pages, these will be noted.