Prediction of Chromatographic Retention Time in High Resolution Anti-Doping Screening Data Using Artificial Neural Networks
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-
![NCAA [R] Drug-Testing Program, 1999-2000](https://docslib.b-cdn.net/cover/2925/ncaa-r-drug-testing-program-1999-2000-42925.webp)
NCAA [R] Drug-Testing Program, 1999-2000
DOCUMENT RESUME ED 436 990 HE 032 608 AUTHOR Halpin, Ty, Ed. TITLE NCAA[R] Drug-Testing Program, 1999-2000. INSTITUTION National Collegiate Athletic Association, Indianapolis, IN. PUB DATE 1999-00-00 NOTE 15p. AVAILABLE FROM National Collegiate Athletic Association, P.O. Box 6222, Indianapolis, IN 46206-6222. Tel: 317-917-6222. PUB TYPE Legal/Legislative/Regulatory Materials (090) EDRS PRICE MF01/PC01 Plus Postage. DESCRIPTORS Athletes; *College Athletics; Drug Abuse; *Drug Use Testing; Higher Education; Illegal Drug Use IDENTIFIERS *National Collegiate Athletic Association ABSTRACT The drug testing program supports NCAA's goal to protect the health and safety of student-athletes competing for their institutions, while reaffirming the organization's commitment to fair and equitable competition. Proposal Nos. 30 and 52-54 provide a program for the NCAA's members to ensure that no one athlete has a chemically-induced advantage or is pressured to use chemical substances. The program involves urine collection on specific occasions and laboratory analyses for substances on a list of banned drugs including stimulants, anabolic agents such as steroids, diuretics, illegal drugs, or peptide hormones. Consent forms must be signed by student athletes if they wish to participate in NCAA programs. (JM) Reproductions supplied by EDRS are the best that can be made from the original document. 6' 3r; - RUG- ESTING ROGRAI4 1999-2000 , Pso (2 BEST COPY AVAILABLE U.S. DEPARTMENT OF EDUCATION Office of Educational Research and Improvement PERMISSION TO REPRODUCE AND EDUCATIONAL RESOURCES INFORMATION DISSEMINATE THIS MATERIAL HAS CENTER (ERIC) BEEN GRANTED BY his document has been reproduced as received from the person or organization originating it. -

Properties and Units in Clinical Pharmacology and Toxicology
Pure Appl. Chem., Vol. 72, No. 3, pp. 479–552, 2000. © 2000 IUPAC INTERNATIONAL FEDERATION OF CLINICAL CHEMISTRY AND LABORATORY MEDICINE SCIENTIFIC DIVISION COMMITTEE ON NOMENCLATURE, PROPERTIES, AND UNITS (C-NPU)# and INTERNATIONAL UNION OF PURE AND APPLIED CHEMISTRY CHEMISTRY AND HUMAN HEALTH DIVISION CLINICAL CHEMISTRY SECTION COMMISSION ON NOMENCLATURE, PROPERTIES, AND UNITS (C-NPU)§ PROPERTIES AND UNITS IN THE CLINICAL LABORATORY SCIENCES PART XII. PROPERTIES AND UNITS IN CLINICAL PHARMACOLOGY AND TOXICOLOGY (Technical Report) (IFCC–IUPAC 1999) Prepared for publication by HENRIK OLESEN1, DAVID COWAN2, RAFAEL DE LA TORRE3 , IVAN BRUUNSHUUS1, MORTEN ROHDE1, and DESMOND KENNY4 1Office of Laboratory Informatics, Copenhagen University Hospital (Rigshospitalet), Copenhagen, Denmark; 2Drug Control Centre, London University, King’s College, London, UK; 3IMIM, Dr. Aiguader 80, Barcelona, Spain; 4Dept. of Clinical Biochemistry, Our Lady’s Hospital for Sick Children, Crumlin, Dublin 12, Ireland #§The combined Memberships of the Committee and the Commission (C-NPU) during the preparation of this report (1994–1996) were as follows: Chairman: H. Olesen (Denmark, 1989–1995); D. Kenny (Ireland, 1996); Members: X. Fuentes-Arderiu (Spain, 1991–1997); J. G. Hill (Canada, 1987–1997); D. Kenny (Ireland, 1994–1997); H. Olesen (Denmark, 1985–1995); P. L. Storring (UK, 1989–1995); P. Soares de Araujo (Brazil, 1994–1997); R. Dybkær (Denmark, 1996–1997); C. McDonald (USA, 1996–1997). Please forward comments to: H. Olesen, Office of Laboratory Informatics 76-6-1, Copenhagen University Hospital (Rigshospitalet), 9 Blegdamsvej, DK-2100 Copenhagen, Denmark. E-mail: [email protected] Republication or reproduction of this report or its storage and/or dissemination by electronic means is permitted without the need for formal IUPAC permission on condition that an acknowledgment, with full reference to the source, along with use of the copyright symbol ©, the name IUPAC, and the year of publication, are prominently visible. -

Prohibited Substances List
Prohibited Substances List This is the Equine Prohibited Substances List that was voted in at the FEI General Assembly in November 2009 alongside the new Equine Anti-Doping and Controlled Medication Regulations(EADCMR). Neither the List nor the EADCM Regulations are in current usage. Both come into effect on 1 January 2010. The current list of FEI prohibited substances remains in effect until 31 December 2009 and can be found at Annex II Vet Regs (11th edition) Changes in this List : Shaded row means that either removed or allowed at certain limits only SUBSTANCE ACTIVITY Banned Substances 1 Acebutolol Beta blocker 2 Acefylline Bronchodilator 3 Acemetacin NSAID 4 Acenocoumarol Anticoagulant 5 Acetanilid Analgesic/anti-pyretic 6 Acetohexamide Pancreatic stimulant 7 Acetominophen (Paracetamol) Analgesic/anti-pyretic 8 Acetophenazine Antipsychotic 9 Acetylmorphine Narcotic 10 Adinazolam Anxiolytic 11 Adiphenine Anti-spasmodic 12 Adrafinil Stimulant 13 Adrenaline Stimulant 14 Adrenochrome Haemostatic 15 Alclofenac NSAID 16 Alcuronium Muscle relaxant 17 Aldosterone Hormone 18 Alfentanil Narcotic 19 Allopurinol Xanthine oxidase inhibitor (anti-hyperuricaemia) 20 Almotriptan 5 HT agonist (anti-migraine) 21 Alphadolone acetate Neurosteriod 22 Alphaprodine Opiod analgesic 23 Alpidem Anxiolytic 24 Alprazolam Anxiolytic 25 Alprenolol Beta blocker 26 Althesin IV anaesthetic 27 Althiazide Diuretic 28 Altrenogest (in males and gelidngs) Oestrus suppression 29 Alverine Antispasmodic 30 Amantadine Dopaminergic 31 Ambenonium Cholinesterase inhibition 32 Ambucetamide Antispasmodic 33 Amethocaine Local anaesthetic 34 Amfepramone Stimulant 35 Amfetaminil Stimulant 36 Amidephrine Vasoconstrictor 37 Amiloride Diuretic 1 Prohibited Substances List This is the Equine Prohibited Substances List that was voted in at the FEI General Assembly in November 2009 alongside the new Equine Anti-Doping and Controlled Medication Regulations(EADCMR). -

Correspondence
SEPT. 26, 1959 TO-DAY'S DRUGS MEDICAL JOURNAL 577 convulsions. The electroencephalogram changes can be reversed and the convulsions stopped by the intravenous administration of sodium thiopentone. Correspondence Although at irst it was thought that bemegride had a specific action in antagonizing the depressant effects of Because of heavy pressure on our space, correspondents are barbiturates on the central nervous system, this view is now asked to keep their letters short. no longer held. Bemegride is looked upon as a non- specific stimulant of the central nervous system and beneficial in treating cases of poisoning by various other Isaac Wolfson Foundation's Fund narcotics (other than barbiturates), such as primidone and glutethimide, as well as barbiturates. `SIR,-I would like to thank you for your leading Therapeutic uses of bemegride are as follows: article on the subject of the recent magnificent grant to (1) In the treatment of acute barbiturate poisoning. Here, in the Royal College of Physicians (Journal, September 12, addition to the usual general method of dealing with a comatose p. 416) and for giving space to a description of the patient, a 5% glucose saline intravenous infusion is given slowly, important activities of the Isaac Wolfson Foundation and 10 ml. of 0.5% solution of bemegride (50 mg.) is injected (p. 427). This grant is indeed a historic gift which my into the tubing leading to the intravenous cannula every three and Medicine are not to to five minutes. Some clinicians'precede each injection with 1 ml. College British likely forget. of a 1.5% solution of amiphenazole (15 mg.). -

Federal Register / Vol. 60, No. 80 / Wednesday, April 26, 1995 / Notices DIX to the HTSUS—Continued
20558 Federal Register / Vol. 60, No. 80 / Wednesday, April 26, 1995 / Notices DEPARMENT OF THE TREASURY Services, U.S. Customs Service, 1301 TABLE 1.ÐPHARMACEUTICAL APPEN- Constitution Avenue NW, Washington, DIX TO THE HTSUSÐContinued Customs Service D.C. 20229 at (202) 927±1060. CAS No. Pharmaceutical [T.D. 95±33] Dated: April 14, 1995. 52±78±8 ..................... NORETHANDROLONE. A. W. Tennant, 52±86±8 ..................... HALOPERIDOL. Pharmaceutical Tables 1 and 3 of the Director, Office of Laboratories and Scientific 52±88±0 ..................... ATROPINE METHONITRATE. HTSUS 52±90±4 ..................... CYSTEINE. Services. 53±03±2 ..................... PREDNISONE. 53±06±5 ..................... CORTISONE. AGENCY: Customs Service, Department TABLE 1.ÐPHARMACEUTICAL 53±10±1 ..................... HYDROXYDIONE SODIUM SUCCI- of the Treasury. NATE. APPENDIX TO THE HTSUS 53±16±7 ..................... ESTRONE. ACTION: Listing of the products found in 53±18±9 ..................... BIETASERPINE. Table 1 and Table 3 of the CAS No. Pharmaceutical 53±19±0 ..................... MITOTANE. 53±31±6 ..................... MEDIBAZINE. Pharmaceutical Appendix to the N/A ............................. ACTAGARDIN. 53±33±8 ..................... PARAMETHASONE. Harmonized Tariff Schedule of the N/A ............................. ARDACIN. 53±34±9 ..................... FLUPREDNISOLONE. N/A ............................. BICIROMAB. 53±39±4 ..................... OXANDROLONE. United States of America in Chemical N/A ............................. CELUCLORAL. 53±43±0 -

Appendix B Banned Drugs
APPENDIX B BANNED DRUGS (This list complies with 2004-2005 NCAA Bylaw 31.2.3.1 Banned Drugs) A. Stimulants: amiphenazole amphetamine bemigride benzphetamine bromantan caffeine1 (guarana) chlorphentermine cocaine cropropamide crothetamide diethylpropion dimethylamphetamine doxapram ephedrine (ephedra, ma huang) ethamivan ethylamphetamine fencamfamine meclofenoxate methamphetamine methylene-dioxymethamphetamine [MDMA (ecstasy)] methylphenidate nikethamide pemoline pentetrazol phendimetrazine phenmetrazine phentermine phenylephrine phenylpropanolamine (ppa) picrotoxine pipradol prolintane strychnine synephrine (citrus aurantium, zhi shi, bitter orange) and related compounds Approved by Cabinet:1/31/05 Adopted by Board of Trustees:3/18/05 Page 12 of 15 B. Anabolic Agents: polythiazide anabolic steroids quinethazone androstenediol spironolactone androstenedione triamterene boldenone trichlormethiazide clostebol and related compounds dehydrochlormethyltestosterone dehydroepiandrosterone (DHEA) dihydrotestosterone (DHT) D. Street Drugs: dromostanolone heroin fluoxymesterone marijuana3 gestrinone THC mesterolone (tetrahydrocannabinol3 methandienone methenolone methyltestosterone nandrolone norandrostenediol norandrostenedione norethandrolone oxandrolone oxymesterone oxymetholone stanozolol testosterone2 tetrahydrogestrinone (THG) trenbolone and related compounds other anabolic agents clenbuterol C. Diuretics: acetazolamide bendroflumethiazide benzthiazide bumetanide chlorothiazide chlorthalidone ethacrynic acid flumethiazide furosemide hydrochlorothiazide -

Pharmacology of Stimulants Prohibited by the World Anti-Doping Agency (WADA)
British Journal of Pharmacology (2008) 154, 606–622 & 2008 Nature Publishing Group All rights reserved 0007– 1188/08 $30.00 www.brjpharmacol.org REVIEW Pharmacology of stimulants prohibited by the World Anti-Doping Agency (WADA) JR Docherty Department of Physiology, Royal College of Surgeons in Ireland, Dublin, Ireland This review examines the pharmacology of stimulants prohibited by the World Anti-Doping Agency (WADA). Stimulants that increase alertness/reduce fatigue or activate the cardiovascular system can include drugs like ephedrine available in many over- the-counter medicines. Others such as amphetamines, cocaine and hallucinogenic drugs, available on prescription or illegally, can modify mood. A total of 62 stimulants (61 chemical entities) are listed in the WADA List, prohibited in competition. Athletes may have stimulants in their body for one of three main reasons: inadvertent consumption in a propriety medicine; deliberate consumption for misuse as a recreational drug and deliberate consumption to enhance performance. The majority of stimulants on the list act on the monoaminergic systems: adrenergic (sympathetic, transmitter noradrenaline), dopaminergic (transmitter dopamine) and serotonergic (transmitter serotonin, 5-HT). Sympathomimetic describes agents, which mimic sympathetic responses, and dopaminomimetic and serotoninomimetic can be used to describe actions on the dopamine and serotonin systems. However, many agents act to mimic more than one of these monoamines, so that a collective term of monoaminomimetic may be useful. Monoaminomimietic actions of stimulants can include blockade of re-uptake of neurotransmitter, indirect release of neurotransmitter, direct activation of monoaminergic receptors. Many of the stimulants are amphetamines or amphetamine derivatives, including agents with abuse potential as recreational drugs. -

A Abacavir Abacavirum Abakaviiri Abagovomab Abagovomabum
A abacavir abacavirum abakaviiri abagovomab abagovomabum abagovomabi abamectin abamectinum abamektiini abametapir abametapirum abametapiiri abanoquil abanoquilum abanokiili abaperidone abaperidonum abaperidoni abarelix abarelixum abareliksi abatacept abataceptum abatasepti abciximab abciximabum absiksimabi abecarnil abecarnilum abekarniili abediterol abediterolum abediteroli abetimus abetimusum abetimuusi abexinostat abexinostatum abeksinostaatti abicipar pegol abiciparum pegolum abisipaaripegoli abiraterone abirateronum abirateroni abitesartan abitesartanum abitesartaani ablukast ablukastum ablukasti abrilumab abrilumabum abrilumabi abrineurin abrineurinum abrineuriini abunidazol abunidazolum abunidatsoli acadesine acadesinum akadesiini acamprosate acamprosatum akamprosaatti acarbose acarbosum akarboosi acebrochol acebrocholum asebrokoli aceburic acid acidum aceburicum asebuurihappo acebutolol acebutololum asebutololi acecainide acecainidum asekainidi acecarbromal acecarbromalum asekarbromaali aceclidine aceclidinum aseklidiini aceclofenac aceclofenacum aseklofenaakki acedapsone acedapsonum asedapsoni acediasulfone sodium acediasulfonum natricum asediasulfoninatrium acefluranol acefluranolum asefluranoli acefurtiamine acefurtiaminum asefurtiamiini acefylline clofibrol acefyllinum clofibrolum asefylliiniklofibroli acefylline piperazine acefyllinum piperazinum asefylliinipiperatsiini aceglatone aceglatonum aseglatoni aceglutamide aceglutamidum aseglutamidi acemannan acemannanum asemannaani acemetacin acemetacinum asemetasiini aceneuramic -

Medicines Amendment Regulations 2018
2018/179 Medicines Amendment Regulations 2018 Patsy Reddy, Governor-General Order in Council At Wellington this 24th day of September 2018 Present: Her Excellency the Governor-General in Council These regulations are made under section 105(1)(j) of the Medicines Act 1981— (a) on the advice and with the consent of the Executive Council; and (b) on the advice of the Minister of Health tendered after consultation with the organisations or bodies appearing to the Minister to be representative of per- sons likely to be substantially affected. Contents Page 1 Title 1 2 Commencement 2 3 Principal regulations 2 4 Schedule 1 replaced 2 5 Regulations revoked 2 Schedule 2 Schedule 1 replaced Regulations 1 Title These regulations are the Medicines Amendment Regulations 2018. 1 r 2 Medicines Amendment Regulations 2018 2018/179 2 Commencement These regulations come into force on the 28th day after the date of their notifi- cation in the Gazette. 3 Principal regulations These regulations amend the Medicines Regulations 1984 (the principal regu- lations). 4 Schedule 1 replaced Replace Schedule 1 with the Schedule 1 set out in the Schedule of these regula- tions. 5 Regulations revoked The Medicines Amendment Regulations (No 2) 2015 (LI 2015/180) are revoked. Schedule Schedule 1 replaced r 4 Schedule 1 Prescription, restricted, and pharmacy-only medicines r 3 Every reference to a medicine in this schedule applies whether the medicine is syn- thetic in origin or is from biological or mineral sources. Unless specific reference is made otherwise, every reference applies also to medicines that are— • preparations and admixtures containing any proportion of any substance listed in this schedule: • salts and esters of any substance listed in this schedule: • preparations or extracts of biological materials listed in this schedule: • salts or oxides of elements listed in this schedule. -
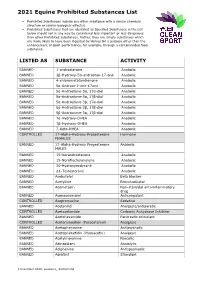
2021 Equine Prohibited Substances List
2021 Equine Prohibited Substances List . Prohibited Substances include any other substance with a similar chemical structure or similar biological effect(s). Prohibited Substances that are identified as Specified Substances in the List below should not in any way be considered less important or less dangerous than other Prohibited Substances. Rather, they are simply substances which are more likely to have been ingested by Horses for a purpose other than the enhancement of sport performance, for example, through a contaminated food substance. LISTED AS SUBSTANCE ACTIVITY BANNED 1-androsterone Anabolic BANNED 3β-Hydroxy-5α-androstan-17-one Anabolic BANNED 4-chlorometatandienone Anabolic BANNED 5α-Androst-2-ene-17one Anabolic BANNED 5α-Androstane-3α, 17α-diol Anabolic BANNED 5α-Androstane-3α, 17β-diol Anabolic BANNED 5α-Androstane-3β, 17α-diol Anabolic BANNED 5α-Androstane-3β, 17β-diol Anabolic BANNED 5β-Androstane-3α, 17β-diol Anabolic BANNED 7α-Hydroxy-DHEA Anabolic BANNED 7β-Hydroxy-DHEA Anabolic BANNED 7-Keto-DHEA Anabolic CONTROLLED 17-Alpha-Hydroxy Progesterone Hormone FEMALES BANNED 17-Alpha-Hydroxy Progesterone Anabolic MALES BANNED 19-Norandrosterone Anabolic BANNED 19-Noretiocholanolone Anabolic BANNED 20-Hydroxyecdysone Anabolic BANNED Δ1-Testosterone Anabolic BANNED Acebutolol Beta blocker BANNED Acefylline Bronchodilator BANNED Acemetacin Non-steroidal anti-inflammatory drug BANNED Acenocoumarol Anticoagulant CONTROLLED Acepromazine Sedative BANNED Acetanilid Analgesic/antipyretic CONTROLLED Acetazolamide Carbonic Anhydrase Inhibitor BANNED Acetohexamide Pancreatic stimulant CONTROLLED Acetominophen (Paracetamol) Analgesic BANNED Acetophenazine Antipsychotic BANNED Acetophenetidin (Phenacetin) Analgesic BANNED Acetylmorphine Narcotic BANNED Adinazolam Anxiolytic BANNED Adiphenine Antispasmodic BANNED Adrafinil Stimulant 1 December 2020, Lausanne, Switzerland 2021 Equine Prohibited Substances List . Prohibited Substances include any other substance with a similar chemical structure or similar biological effect(s). -

ATHLETICS STANDARD OPERATING PROCEDURES (SOP) Core Values Community, Integrity, Diversity, Scholarship and Outreach
ATHLETICS STANDARD OPERATING PROCEDURES (SOP) Core Values Community, Integrity, Diversity, Scholarship and Outreach DELAWARE STATE UNIVERSITY 1 Table of Contents Administration Athletics Directory………………………………………………………………………8 Delaware State University (DSU) Mission and Vision Statement….……………….…11 Athletics Mission and Vision Statement ………………………..…………………….12 Organizational Chart……………………………………………………….………….13 Onboarding……….…………………………………………………...........................14 Out Processing Employees………………………………………………………….....14 Salaries (Athletics)………………………………………………………………….....15 Email Signature………………………………………………………………………..16 Leave/Vacation (Athletics)-DSU……………………………………………………...17 Benefits-Paystubs-W-2’s………………………………………………………………18 Outside Employment…………………………………………………………………..19 Game/Competition Contracts …………………………………………………………19 Budget and Finance Policies and Procedures Responsibility and Control…………………………………………………………….20 Budget…………………………………………………………………………………20 Annual Audit….…………………………………………………………………….....20 Team Travel …………………………………………………………………………...20 Individual Travel……………………………………………………………………….21 Petty Cash for Student Host during On-Campus Recruiting…………………………..22 Rights Holder Multiyear Agreement…………………………………………………...22 Scope of Services Rendered by Peak Sports…………………………………...22 Money received by Athletics…………………………………………………………..23 DSU Athletic Camps………………………………………………….………………..25 Receipt book procedures………….…………………………………………...25 Cash Handling Prior to deposit………………………………………………..25 Deposits………………………………………………………………………..25 Other Miscellaneous -

Prohibited List 2021
WORLD ANTI-DOPING CODE INTERNATIONAL STANDARD PROHIBITED LIST 2021 This List shall come into effect on 1 January 2021. TABLE OF CONTENTS Please note that the list of examples of medical conditions below is not inclusive. SUBSTANCES & METHODS PROHIBITED AT ALL TIMES S0 Non-approved substances ..................................................................................4 S1 Anabolic agents ...................................................................................................5 Some of these substance(s) may be found, without limitation, in medications used for the treatment of e.g. male hypogonadism. S2 Peptide hormones, growth factors, related substances, and mimetics ...........7 Some of these substance(s) may be found, without limitation, in medications used for the treatment of e.g. anaemia, male hypogonadism, growth hormone deficiency. S3 Beta-2 agonists ...................................................................................................9 Some of these substance(s) may be found, without limitation, in medications used for the treatment of e.g. asthma and other respiratory disorders. S4 Hormone and metabolic modulators ...............................................................10 Some of these substance(s) may be found, without limitation, in medications used for the treatment of e.g. breast cancer, diabetes, infertility (female), polycystic ovarian syndrome. S5 Diuretics and masking agents ..........................................................................12 Some of these substance(s) may