Download.Html) to Perform Technologies, USA)
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-
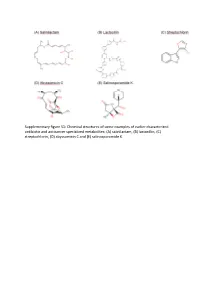
Chemical Structures of Some Examples of Earlier Characterized Antibiotic and Anticancer Specialized
Supplementary figure S1: Chemical structures of some examples of earlier characterized antibiotic and anticancer specialized metabolites: (A) salinilactam, (B) lactocillin, (C) streptochlorin, (D) abyssomicin C and (E) salinosporamide K. Figure S2. Heat map representing hierarchical classification of the SMGCs detected in all the metagenomes in the dataset. Table S1: The sampling locations of each of the sites in the dataset. Sample Sample Bio-project Site depth accession accession Samples Latitude Longitude Site description (m) number in SRA number in SRA AT0050m01B1-4C1 SRS598124 PRJNA193416 Atlantis II water column 50, 200, Water column AT0200m01C1-4D1 SRS598125 21°36'19.0" 38°12'09.0 700 and above the brine N "E (ATII 50, ATII 200, 1500 pool water layers AT0700m01C1-3D1 SRS598128 ATII 700, ATII 1500) AT1500m01B1-3C1 SRS598129 ATBRUCL SRS1029632 PRJNA193416 Atlantis II brine 21°36'19.0" 38°12'09.0 1996– Brine pool water ATBRLCL1-3 SRS1029579 (ATII UCL, ATII INF, N "E 2025 layers ATII LCL) ATBRINP SRS481323 PRJNA219363 ATIID-1a SRS1120041 PRJNA299097 ATIID-1b SRS1120130 ATIID-2 SRS1120133 2168 + Sea sediments Atlantis II - sediments 21°36'19.0" 38°12'09.0 ~3.5 core underlying ATII ATIID-3 SRS1120134 (ATII SDM) N "E length brine pool ATIID-4 SRS1120135 ATIID-5 SRS1120142 ATIID-6 SRS1120143 Discovery Deep brine DDBRINP SRS481325 PRJNA219363 21°17'11.0" 38°17'14.0 2026– Brine pool water N "E 2042 layers (DD INF, DD BR) DDBRINE DD-1 SRS1120158 PRJNA299097 DD-2 SRS1120203 DD-3 SRS1120205 Discovery Deep 2180 + Sea sediments sediments 21°17'11.0" -

Anoxygenic Photosynthesis in Photolithotrophic Sulfur Bacteria and Their Role in Detoxication of Hydrogen Sulfide
antioxidants Review Anoxygenic Photosynthesis in Photolithotrophic Sulfur Bacteria and Their Role in Detoxication of Hydrogen Sulfide Ivan Kushkevych 1,* , Veronika Bosáková 1,2 , Monika Vítˇezová 1 and Simon K.-M. R. Rittmann 3,* 1 Department of Experimental Biology, Faculty of Science, Masaryk University, 62500 Brno, Czech Republic; [email protected] (V.B.); [email protected] (M.V.) 2 Department of Biology, Faculty of Medicine, Masaryk University, 62500 Brno, Czech Republic 3 Archaea Physiology & Biotechnology Group, Department of Functional and Evolutionary Ecology, Universität Wien, 1090 Vienna, Austria * Correspondence: [email protected] (I.K.); [email protected] (S.K.-M.R.R.); Tel.: +420-549-495-315 (I.K.); +431-427-776-513 (S.K.-M.R.R.) Abstract: Hydrogen sulfide is a toxic compound that can affect various groups of water microorgan- isms. Photolithotrophic sulfur bacteria including Chromatiaceae and Chlorobiaceae are able to convert inorganic substrate (hydrogen sulfide and carbon dioxide) into organic matter deriving energy from photosynthesis. This process takes place in the absence of molecular oxygen and is referred to as anoxygenic photosynthesis, in which exogenous electron donors are needed. These donors may be reduced sulfur compounds such as hydrogen sulfide. This paper deals with the description of this metabolic process, representatives of the above-mentioned families, and discusses the possibility using anoxygenic phototrophic microorganisms for the detoxification of toxic hydrogen sulfide. Moreover, their general characteristics, morphology, metabolism, and taxonomy are described as Citation: Kushkevych, I.; Bosáková, well as the conditions for isolation and cultivation of these microorganisms will be presented. V.; Vítˇezová,M.; Rittmann, S.K.-M.R. -

Exploring Antibiotic Susceptibility, Resistome and Mobilome Structure of Planctomycetes from Gemmataceae Family
sustainability Article Exploring Antibiotic Susceptibility, Resistome and Mobilome Structure of Planctomycetes from Gemmataceae Family Anastasia A. Ivanova *, Kirill K. Miroshnikov and Igor Y. Oshkin Research Center of Biotechnology of the Russian Academy of Sciences, Winogradsky Institute of Microbiology, 119071 Moscow, Russia; [email protected] (K.K.M.); [email protected] (I.Y.O.) * Correspondence: [email protected] Abstract: The family Gemmataceae accomodates aerobic, chemoorganotrophic planctomycetes with large genome sizes, is mostly distributed in freshwater and terrestrial environments. However, these bacteria have recently also been found in locations relevant to human health. Since the antimi- crobial resistance genes (AMR) from environmental resistome have the potential to be transferred to pathogens, it is essential to explore the resistant capabilities of environmental bacteria. In this study, the reconstruction of in silico resistome was performed for all nine available gemmata genomes. Furthermore, the genome of the newly isolated yet-undescribed strain G18 was sequenced and added to all analyses steps. Selected genomes were screened for the presence of mobile genetic elements. The flanking location of mobilizable genomic milieu around the AMR genes was of particular in- terest since such colocalization may appear to promote the horizontal gene transfer (HGT) events. Moreover the antibiotic susceptibility profile of six phylogenetically distinct strains of Gemmataceae planctomycetes was determined. Citation: Ivanova, A.A.; Keywords: planctomycetes; Gemmataceae; antibiotic resistance profile; resistome; mobilome Miroshnikov, K.K.; Oshkin, I.Y. Exploring Antibiotic Susceptibility, Resistome and Mobilome Structure of Planctomycetes from Gemmataceae 1. Introduction Family. Sustainability 2021, 13, 5031. Several decades ago humanity faced the global issue of growing antibiotic resistance https://doi.org/10.3390/su13095031 of bacterial pathogens in clinic [1–5]. -

Limits of Life on Earth Some Archaea and Bacteria
Limits of life on Earth Thermophiles Temperatures up to ~55C are common, but T > 55C is Some archaea and bacteria (extremophiles) can live in associated usually with geothermal features (hot springs, environments that we would consider inhospitable to volcanic activity etc) life (heat, cold, acidity, high pressure etc) Thermophiles are organisms that can successfully live Distinguish between growth and survival: many organisms can survive intervals of harsh conditions but could not at high temperatures live permanently in such conditions (e.g. seeds, spores) Best studied extremophiles: may be relevant to the Interest: origin of life. Very hot environments tolerable for life do not seem to exist elsewhere in the Solar System • analogs for extraterrestrial environments • `extreme’ conditions may have been more common on the early Earth - origin of life? • some unusual environments (e.g. subterranean) are very widespread Extraterrestrial Life: Spring 2008 Extraterrestrial Life: Spring 2008 Grand Prismatic Spring, Yellowstone National Park Hydrothermal vents: high pressure in the deep ocean allows liquid water Colors on the edge of the at T >> 100C spring are caused by different colonies of thermophilic Vents emit superheated water (300C or cyanobacteria and algae more) that is rich in minerals Hottest water is lifeless, but `cooler’ ~50 species of such thermophiles - mostly archae with some margins support array of thermophiles: cyanobacteria and anaerobic photosynthetic bacteria oxidize sulphur, manganese, grow on methane + carbon monoxide etc… Sulfolobus: optimum T ~ 80C, minimum 60C, maximum 90C, also prefer a moderately acidic pH. Live by oxidizing sulfur Known examples can grow (i.e. multiply) at temperatures which is abundant near hot springs. -

Updating the Taxonomic Toolbox: Classification of Alteromonas Spp
1 Updating the taxonomic toolbox: classification of Alteromonas spp. 2 using Multilocus Phylogenetic Analysis and MALDI-TOF Mass 3 Spectrometry a a a 4 Hooi Jun Ng , Hayden K. Webb , Russell J. Crawford , François a b b c 5 Malherbe , Henry Butt , Rachel Knight , Valery V. Mikhailov and a, 6 Elena P. Ivanova * 7 aFaculty of Life and Social Sciences, Swinburne University of Technology, 8 PO Box 218, Hawthorn, Vic 3122, Australia 9 bBioscreen, Bio21 Institute, The University of Melbourne, Vic 3010, Australia 10 cG.B. Elyakov Pacific Institute of Bioorganic Chemistry, Far Eastern Branch, Russian 11 Academy of Sciences, Vladivostok 690022, Russian Federation 12 13 *Corresponding author: Tel: +61-3-9214-5137. Fax: +61-3-9214-5050. 14 E-mail: [email protected] 15 16 Abstract 17 Bacteria of the genus Alteromonas are Gram-negative, strictly aerobic, motile, 18 heterotrophic marine bacteria, known for their versatile metabolic activities. 19 Identification and classification of novel species belonging to the genus Alteromonas 20 generally involves DNA-DNA hybridization (DDH) as distinct species often fail to be 1 21 resolved at the 97% threshold value of the 16S rRNA gene sequence similarity. In this 22 study, the applicability of Multilocus Phylogenetic Analysis (MLPA) and Matrix- 23 Assisted Laser Desorption Ionization Time-of-Flight Mass Spectrometry (MALDI-TOF 24 MS) for the differentiation of Alteromonas species has been evaluated. Phylogenetic 25 analysis incorporating five house-keeping genes (dnaK, sucC, rpoB, gyrB, and rpoD) 26 revealed a threshold value of 98.9% that could be considered as the species cut-off 27 value for the delineation of Alteromonas spp. -

UNIVERSITY of CALIFORNIA, SAN DIEGO Indicators of Iron
UNIVERSITY OF CALIFORNIA, SAN DIEGO Indicators of Iron Metabolism in Marine Microbial Genomes and Ecosystems A dissertation submitted in partial satisfaction of the requirements for the degree Doctor of Philosophy in Oceanography by Shane Lahman Hogle Committee in charge: Katherine Barbeau, Chair Eric Allen Bianca Brahamsha Christopher Dupont Brian Palenik Kit Pogliano 2016 Copyright Shane Lahman Hogle, 2016 All rights reserved . The Dissertation of Shane Lahman Hogle is approved, and it is acceptable in quality and form for publication on microfilm and electronically: Chair University of California, San Diego 2016 iii DEDICATION Mom, Dad, Joel, and Marie thank you for everything iv TABLE OF CONTENTS Signature Page ................................................................................................................... iii Dedication .......................................................................................................................... iv Table of Contents .................................................................................................................v List of Figures ................................................................................................................... vii List of Tables ..................................................................................................................... ix Acknowledgements ..............................................................................................................x Vita .................................................................................................................................. -

Motiliproteus Sediminis Gen. Nov., Sp. Nov., Isolated from Coastal Sediment
Antonie van Leeuwenhoek (2014) 106:615–621 DOI 10.1007/s10482-014-0232-2 ORIGINAL PAPER Motiliproteus sediminis gen. nov., sp. nov., isolated from coastal sediment Zong-Jie Wang • Zhi-Hong Xie • Chao Wang • Zong-Jun Du • Guan-Jun Chen Received: 3 April 2014 / Accepted: 4 July 2014 / Published online: 20 July 2014 Ó Springer International Publishing Switzerland 2014 Abstract A novel Gram-stain-negative, rod-to- demonstrated that the novel isolate was 93.3 % similar spiral-shaped, oxidase- and catalase- positive and to the type strain of Neptunomonas antarctica, 93.2 % facultatively aerobic bacterium, designated HS6T, was to Neptunomonas japonicum and 93.1 % to Marino- isolated from marine sediment of Yellow Sea, China. bacterium rhizophilum, the closest cultivated rela- It can reduce nitrate to nitrite and grow well in marine tives. The polar lipid profile of the novel strain broth 2216 (MB, Hope Biol-Technology Co., Ltd) consisted of phosphatidylethanolamine, phosphatidyl- with an optimal temperature for growth of 30–33 °C glycerol and some other unknown lipids. Major (range 12–45 °C) and in the presence of 2–3 % (w/v) cellular fatty acids were summed feature 3 (C16:1 NaCl (range 0.5–7 %, w/v). The pH range for growth x7c/iso-C15:0 2-OH), C18:1 x7c and C16:0 and the main was pH 6.2–9.0, with an optimum at 6.5–7.0. Phylo- respiratory quinone was Q-8. The DNA G?C content genetic analysis based on 16S rRNA gene sequences of strain HS6T was 61.2 mol %. Based on the phylogenetic, physiological and biochemical charac- teristics, strain HS6T represents a novel genus and The GenBank accession number for the 16S rRNA gene T species and the name Motiliproteus sediminis gen. -

PROTISTS Shore and the Waves Are Large, Often the Largest of a Storm Event, and with a Long Period
(seas), and these waves can mobilize boulders. During this phase of the storm the rapid changes in current direction caused by these large, short-period waves generate high accelerative forces, and it is these forces that ultimately can move even large boulders. Traditionally, most rocky-intertidal ecological stud- ies have been conducted on rocky platforms where the substrate is composed of stable basement rock. Projec- tiles tend to be uncommon in these types of habitats, and damage from projectiles is usually light. Perhaps for this reason the role of projectiles in intertidal ecology has received little attention. Boulder-fi eld intertidal zones are as common as, if not more common than, rock plat- forms. In boulder fi elds, projectiles are abundant, and the evidence of damage due to projectiles is obvious. Here projectiles may be one of the most important defi ning physical forces in the habitat. SEE ALSO THE FOLLOWING ARTICLES Geology, Coastal / Habitat Alteration / Hydrodynamic Forces / Wave Exposure FURTHER READING Carstens. T. 1968. Wave forces on boundaries and submerged bodies. Sarsia FIGURE 6 The intertidal zone on the north side of Cape Blanco, 34: 37–60. Oregon. The large, smooth boulders are made of serpentine, while Dayton, P. K. 1971. Competition, disturbance, and community organi- the surrounding rock from which the intertidal platform is formed zation: the provision and subsequent utilization of space in a rocky is sandstone. The smooth boulders are from a source outside the intertidal community. Ecological Monographs 45: 137–159. intertidal zone and were carried into the intertidal zone by waves. Levin, S. A., and R. -

Algae and Cyanobacteria in Coastal and Estuarine Waters
CHAPTER 7 Algae and cyanobacteria in coastal and estuarine waters n coastal and estuarine waters, algae range from single-celled forms to the seaweeds. ICyanobacteria are organisms with some characteristics of bacteria and some of algae. They are similar in size to the unicellular algae and, unlike other bacteria, contain blue-green or green pigments and are able to perform photosynthesis; thus, they are also termed blue-green algae. Algal blooms in the sea have occurred throughout recorded history but have been increasing during recent decades (Anderson, 1989; Smayda, 1989a; Hallegraeff, 1993). In several areas (e.g., the Baltic and North seas, the Adriatic Sea, Japanese coastal waters and the Gulf of Mexico), algal blooms are a recurring phe- nomenon. The increased frequency of occurrence has accompanied nutrient enrich- ment of coastal waters on a global scale (Smayda, 1989b). Blooms of non-toxic phytoplankton species and mass occurrences of macro-algae can affect the amenity value of recreational waters due to reduced transparency, dis- coloured water and scum formation. Furthermore, bloom degradation can be accom- panied by unpleasant odours, resulting in aesthetic problems (see chapter 9). Several human diseases have been reported to be associated with many toxic species of dinoflagellates, diatoms, nanoflagellates and cyanobacteria that occur in the marine environment (CDC, 1997). The effects of these algae on humans are due to some of their constituents, principally algal toxins. Marine algal toxins become a problem primarily because they may concentrate in shellfish and fish that are subsequently eaten by humans (CDR, 1991; Lehane, 2000), causing syndromes known as para- lytic shellfish poisoning (PSP), diarrhetic shellfish poisoning (DSP), amnesic shellfish poisoning (ASP), neurotoxic shellfish poisoning (NSP) and ciguatera fish poisoning (CFP). -

Yu-Chen Ling and John W. Moreau
Microbial Distribution and Activity in a Coastal Acid Sulfate Soil System Introduction: Bioremediation in Yu-Chen Ling and John W. Moreau coastal acid sulfate soil systems Method A Coastal acid sulfate soil (CASS) systems were School of Earth Sciences, University of Melbourne, Melbourne, VIC 3010, Australia formed when people drained the coastal area Microbial distribution controlled by environmental parameters Microbial activity showed two patterns exposing the soil to the air. Drainage makes iron Microbial structures can be grouped into three zones based on the highest similarity between samples (Fig. 4). Abundant populations, such as Deltaproteobacteria, kept constant activity across tidal cycling, whereas rare sulfides oxidize and release acidity to the These three zones were consistent with their geological background (Fig. 5). Zone 1: Organic horizon, had the populations changed activity response to environmental variations. Activity = cDNA/DNA environment, low pH pore water further dissolved lowest pH value. Zone 2: surface tidal zone, was influenced the most by tidal activity. Zone 3: Sulfuric zone, Abundant populations: the heavy metals. The acidity and toxic metals then Method A Deltaproteobacteria Deltaproteobacteria this area got neutralized the most. contaminate coastal and nearby ecosystems and Method B 1.5 cause environmental problems, such as fish kills, 1.5 decreased rice yields, release of greenhouse gases, Chloroflexi and construction damage. In Australia, there is Gammaproteobacteria Gammaproteobacteria about a $10 billion “legacy” from acid sulfate soils, Chloroflexi even though Australia is only occupied by around 1.0 1.0 Cyanobacteria,@ Acidobacteria Acidobacteria Alphaproteobacteria 18% of the global acid sulfate soils. Chloroplast Zetaproteobacteria Rare populations: Alphaproteobacteria Method A log(RNA(%)+1) Zetaproteobacteria log(RNA(%)+1) Method C Method B 0.5 0.5 Cyanobacteria,@ Bacteroidetes Chloroplast Firmicutes Firmicutes Bacteroidetes Planctomycetes Planctomycetes Ac8nobacteria Fig. -

Heliorhodopsins Are Absent in Diderm (Gram-Negative) Bacteria
bioRxiv preprint doi: https://doi.org/10.1101/356287; this version posted June 26, 2018. The copyright holder for this preprint (which was not certified by peer review) is the author/funder, who has granted bioRxiv a license to display the preprint in perpetuity. It is made available under aCC-BY-NC-ND 4.0 International license. Heliorhodopsins are absent in diderm (Gram-negative) bacteria: Some thoughts and possible implications for activity José Flores-Uribe*1, Gur Hevroni*1, Rohit Ghai2, Alina Pushkarev1, Keiichi 5 Inoue3-6, Hideki Kandori3,4 and Oded Béjà†1 1Faculty of Biology, Technion – Israel Institute of Technology, Haifa, Israel; 2Institute of Hydrobiology, Department of Aquatic Microbial Ecology, Biology Center of the Academy of Sciences of the Czech Republic, České Budějovice, Czech Republic; 3Department of Life Science and Applied Chemistry, Nagoya Institute of Technology, 10 Showa-ku, Aichi 466-8555, Japan; 4OptoBioTechnology Research Center, Nagoya Institute of Technology, Showa-ku, Nagoya 466-8555, Japan; 5The Institute for Solid State Physics, The University of Tokyo, 5-1-5 Kashiwanoha, Kashiwa, Chiba 277- 8581, Japan; 6PRESTO, Japan Science and Technology Agency, 4-1-8 Honcho, Kawaguchi, Saitama 332-0012, Japan. 15 † Corresponding author. Email: [email protected] (O.B.) * Equal contribution bioRxiv preprint doi: https://doi.org/10.1101/356287; this version posted June 26, 2018. The copyright holder for this preprint (which was not certified by peer review) is the author/funder, who has granted bioRxiv a license to display the preprint in perpetuity. It is made available under aCC-BY-NC-ND 4.0 International license. -

Culturomics of Anaerobic Sludge
Culturomics of Anaerobic Sludge Jack Matthew Murgatroyd MSc by Research University of York Biology January 2019 Abstract Ever increasing concerns surrounding climate change are making alternative energy sources a focus of global discussion. Anaerobic digestion holds promise as one method of generating renewable energy with a low carbon footprint. The simple principle that underlies anaerobic digestion is the degradation of organic material by microorganisms to produce digestate and biogas. While this process occurs without human intervention in a variety of environments, there is still large scope for refinement of the process in order to increase its efficiency in industry. A key problem that limits technology advancement is the understanding of the microbiomes within anaerobic digesters. This can be attributed to the fastidious growth requirements of many of the organisms involved and as such, novel methods for culturing must be adopted if we are to advance our understanding. Work undertaken used a number of different growth backgrounds and isolation chips as a novel method for the enrichment of methanogens and anaerobic fungi from anaerobic sludge samples. 16S rRNA gene sequencing technology was used to assign taxonomy to 1,152 samples. A potential novel bacterial species was further investigated using fluorescent in-situ hybridisation and Sanger sequencing with a view to demonstrating the efficacy of this culturing method for the isolation and culturing of novel microbes. This alternative approach for the cultivation of multiple microbes in isolation has shown promise in terms of the number of novel species grown. Given that 16 samples were investigated at an individual level and one of these has been identified as a novel species, it is likely that novel species have also been cultured within the other 1,136 samples that have not yet been studied individually.