What Can Lycophytes Teach Us About Plant Evolution and Development? Modern Perspectives on an Ancient Lineage
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

History and Philosophy of Systematic Biology
History and Philosophy of Systematic Biology Bock, W. J. (1973) Philosophical foundations of classical evolutionary classification Systematic Zoology 22: 375-392 Part of a general symposium on "Contemporary Systematic Philosophies," there are some other interesting papers here. Brower, A. V. Z. (2000) Evolution Is Not a Necessary Assumption of Cladistics Cladistics 16: 143- 154 Dayrat, Benoit (2005) Ancestor-descendant relationships and the reconstruction of the Tree of Lif Paleobiology 31: 347-353 Donoghue, M.J. and J.W. Kadereit (1992) Walter Zimmermann and the growth of phylogenetic theory Systematic Biology 41: 74-84 Faith, D. P. and J. W. H. Trueman (2001) Towards an inclusive philosophy for phylogenetic inference Systematic Biology 50: 331-350 Gaffney, E. S. (1979) An introduction to the logic of phylogeny reconstruction, pp. 79-111 in Cracraft, J. and N. Eldredge (eds.) Phylogenetic Analysis and Paleontology Columbia University Press, New York. Gilmour, J. S. L. (1940) Taxonomy and philosophy, pp. 461-474 in J. Huxley (ed.) The New Systematics Oxford Hull, D. L. (1978) A matter of individuality Phil. of Science 45: 335-360 Hull, D. L. (1978) The principles of biological classification: the use and abuse of philosophy Hull, D. L. (1984) Cladistic theory: hypotheses that blur and grow, pp. 5-23 in T. Duncan and T. F. Stuessy (eds.) Cladistics: Perspectives on the Reconstruction of Evolutionary History Columbia University Press, New York * Hull, D. L. (1988) Science as a process: an evolutionary account of the social and conceptual development of science University of Chicago Press. An already classic work on the recent, violent history of systematics; used as data for Hull's general theories about scientific change. -

RI Equisetopsida and Lycopodiopsida.Indd
IIntroductionntroduction byby FFrancisrancis UnderwoodUnderwood Rhode Island Equisetopsida, Lycopodiopsida and Isoetopsida Special Th anks to the following for giving permission for the use their images. Robbin Moran New York Botanical Garden George Yatskievych and Ann Larson Missouri Botanical Garden Jan De Laet, plantsystematics.org Th is pdf is a companion publication to Rhode Island Equisetopsida, Lycopodiopsida & Isoetopsida at among-ri-wildfl owers.org Th e Elfi n Press 2016 Introduction Formerly known as fern allies, Horsetails, Club-mosses, Fir-mosses, Spike-mosses and Quillworts are plants that have an alternate generation life-cycle similar to ferns, having both sporophyte and gametophyte stages. Equisetopsida Horsetails date from the Devonian period (416 to 359 million years ago) in earth’s history where they were trees up to 110 feet in height and helped to form the coal deposits of the Carboniferous period. Only one genus has survived to modern times (Equisetum). Horsetails Horsetails (Equisetum) have jointed stems with whorls of thin narrow leaves. In the sporophyte stage, they have a sterile and fertile form. Th ey produce only one type of spore. While the gametophytes produced from the spores appear to be plentiful, the successful reproduction of the sporophyte form is low with most Horsetails reproducing vegetatively. Lycopodiopsida Lycopodiopsida includes the clubmosses (Dendrolycopodium, Diphasiastrum, Lycopodiella, Lycopodium , Spinulum) and Fir-mosses (Huperzia) Clubmosses Clubmosses are evergreen plants that produce only microspores that develop into a gametophyte capable of producing both sperm and egg cells. Club-mosses can produce the spores either in leaf axils or at the top of their stems. Th e spore capsules form in a cone-like structures (strobili) at the top of the plants. -

Gene Expression Data Support the Hypothesis That Isoetes Rootlets Are True Roots and Not Modifed Leaves Alexander J
www.nature.com/scientificreports OPEN Gene expression data support the hypothesis that Isoetes rootlets are true roots and not modifed leaves Alexander J. Hetherington1,2, David M. Emms1, Steven Kelly1 & Liam Dolan1,3* Rhizomorphic lycopsids are the land plant group that includes the frst giant trees to grow on Earth and extant species in the genus Isoetes. Two mutually exclusive hypotheses account for the evolution of terminal rooting axes called rootlets among the rhizomorphic lycopsids. One hypothesis states that rootlets are true roots, like roots in other lycopsids. The other states that rootlets are modifed leaves. Here we test predictions of each hypothesis by investigating gene expression in the leaves and rootlets of Isoetes echinospora. We assembled the de novo transcriptome of axenically cultured I. echinospora. Gene expression signatures of I. echinospora rootlets and leaves were diferent. Furthermore, gene expression signatures of I. echinospora rootlets were similar to gene expression signatures of true roots of Selaginella moellendorfi and Arabidopsis thaliana. RSL genes which positively regulate cell diferentiation in roots were either exclusively or preferentially expressed in the I. echinospora rootlets, S. moellendorfi roots and A. thaliana roots compared to the leaves of each respective species. Taken together, gene expression data from the de-novo transcriptome of I. echinospora are consistent with the hypothesis that Isoetes rootlets are true roots and not modifed leaves. Te frst giant (> 50 m) trees to grow on Earth, the arborescent clubmosses, were tethered to the ground by rooting structures termed stigmarian systems whose homology has been debated for more than 150 years1–9. Stigmarian rooting systems consisted of two components, a central axis (rhizomorph) on which developed large numbers of fne axes (rootlets). -
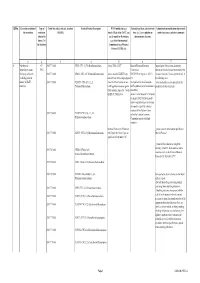
Qrno. 1 2 3 4 5 6 7 1 CP 2903 77 100 0 Cfcl3
QRNo. General description of Type of Tariff line code(s) affected, based on Detailed Product Description WTO Justification (e.g. National legal basis and entry into Administration, modification of previously the restriction restriction HS(2012) Article XX(g) of the GATT, etc.) force (i.e. Law, regulation or notified measures, and other comments (Symbol in and Grounds for Restriction, administrative decision) Annex 2 of e.g., Other International the Decision) Commitments (e.g. Montreal Protocol, CITES, etc) 12 3 4 5 6 7 1 Prohibition to CP 2903 77 100 0 CFCl3 (CFC-11) Trichlorofluoromethane Article XX(h) GATT Board of Eurasian Economic Import/export of these ozone destroying import/export ozone CP-X Commission substances from/to the customs territory of the destroying substances 2903 77 200 0 CF2Cl2 (CFC-12) Dichlorodifluoromethane Article 46 of the EAEU Treaty DECISION on August 16, 2012 N Eurasian Economic Union is permitted only in (excluding goods in dated 29 may 2014 and paragraphs 134 the following cases: transit) (all EAEU 2903 77 300 0 C2F3Cl3 (CFC-113) 1,1,2- 4 and 37 of the Protocol on non- On legal acts in the field of non- _to be used solely as a raw material for the countries) Trichlorotrifluoroethane tariff regulation measures against tariff regulation (as last amended at 2 production of other chemicals; third countries Annex No. 7 to the June 2016) EAEU of 29 May 2014 Annex 1 to the Decision N 134 dated 16 August 2012 Unit list of goods subject to prohibitions or restrictions on import or export by countries- members of the -

The Origin and Early Evolution of Vascular Plant Shoots and Leaves Rstb.Royalsocietypublishing.Org C
Downloaded from http://rstb.royalsocietypublishing.org/ on January 22, 2018 The origin and early evolution of vascular plant shoots and leaves rstb.royalsocietypublishing.org C. Jill Harrison 1 and Jennifer L. Morris 2 1School of Biological Sciences, and 2School of Earth Sciences, University of Bristol, 24 Tyndall Avenue, Bristol BS8 1TQ, UK Review CJH, 0000-0002-5228-600X; JLM, 0000-0002-7453-3841 Cite this article: Harrison CJ, Morris JL. 2017 The morphology of plant fossils from the Rhynie chert has generated long- standing questions about vascular plant shoot and leaf evolution, for The origin and early evolution of vascular plant instance, which morphologies were ancestral within land plants, when did shoots and leaves. Phil. Trans. R. Soc. B 373 : vascular plants first arise and did leaves have multiple evolutionary origins? 20160496. Recent advances combining insights from molecular phylogeny, palaeobotany http://dx.doi.org/10.1098/rstb.2016.0496 and evo–devo research address these questions and suggest the sequence of morphological innovation during vascular plant shoot and leaf evolution. The evidence pinpoints testable developmental and genetic hypotheses relat- Accepted: 11 August 2017 ing to the origin of branching and indeterminate shoot architectures prior to the evolution of leaves, and demonstrates underestimation of polyphyly in One contribution of 18 to a discussion meeting the evolution of leaves from branching forms in ‘telome theory’ hypotheses issue ‘The Rhynie cherts: our earliest terrestrial of leaf evolution. This review discusses fossil, developmental and genetic ecosystem revisited’. evidence relating to the evolution of vascular plant shoots and leaves in a phylogenetic framework. This article is part of a discussion meeting issue ‘The Rhynie cherts: our Subject Areas: earliest terrestrial ecosystem revisited’. -

Getting to the Roots: a Developmental Genetic View of Root Anatomy and Function from Arabidopsis to Lycophytes
fpls-09-01410 September 21, 2018 Time: 17:3 # 1 REVIEW published: 25 September 2018 doi: 10.3389/fpls.2018.01410 Getting to the Roots: A Developmental Genetic View of Root Anatomy and Function From Arabidopsis to Lycophytes Frauke Augstein and Annelie Carlsbecker* Department of Organismal Biology, Physiological Botany and Linnean Centre for Plant Biology in Uppsala, Uppsala University, Uppsala, Sweden Roots attach plants to the ground and ensure efficient and selective uptake of water and nutrients. These functions are facilitated by the morphological and anatomical structures of the root, formed by the activity of the root apical meristem (RAM) and consecutive patterning and differentiation of specific tissues with distinct functions. Despite the importance of this plant organ, its evolutionary history is not clear, but fossils suggest that roots evolved at least twice, in the lycophyte (clubmosses and their allies) and in the euphyllophyte (ferns and seed plants) lineages. Both lycophyte and euphyllophyte roots grow indeterminately by the action of an apical meristem, which is protected by a root cap. They produce root hairs, and in most species the vascular stele is Edited by: guarded by a specialized endodermal cell layer. Hence, most of these traits must have Annette Becker, evolved independently in these lineages. This raises the question if the development Justus Liebig Universität Gießen, Germany of these apparently analogous tissues is regulated by distinct or homologous genes, Reviewed by: independently recruited from a common ancestor of lycophytes and euphyllophytes. Hongchang Cui, Currently, there are few studies of the genetic and molecular regulation of lycophyte Florida State University, United States and fern roots. -

Lake Quillwort Isoetes Lacustris
Natural Heritage Lake Quillwort & Endangered Species Isoetes lacustris L. Program www.mass.gov/nhesp State Status: Endangered Federal Status: None Massachusetts Division of Fisheries & Wildlife DESCRIPTION: Lake Quillwort is a perennial, aquatic, nonflowering member of the Quillwort family (Isoetaceae). This inconspicuous species lives submerged in ponds as a rosette of linear leaves (somewhat resembling chives), and reproduces via spores. AIDS TO IDENTIFICATION: The rosette of Lake Quillwort is composed of sharply pointed leaves, 0.7 to 2 mm wide and mostly 5 to 10 cm (2–4 in.) long, occasionally reaching 20 cm. The leaves are dark green, firm, fleshy, and brittle, emerging from a very short, thick stem that is anchored in the substrate by a subterranean corm. The leaf bases of quillworts are swollen, flattened, and concave; the rosette is arranged tightly like the bracts of an artichoke. Within the swollen Lake Quillwort is an aquatic species with a rosette of leaves that leaf bases are sporangia, sacs that house the male have swollen bases; within the leaf bases are sporangia with gametophyte-bearing microspores and the female microspores and megaspores. Photo by Robbin Moran. megaspores. A sheath, or velum, covers the sporangia; in Lake Quillwort, the velum covers up to half of the sporangia. The megaspore of Lake Quillwort, which SIMILAR SPECIES: Quillwort species are very requires a microscope to view, is mostly covered with similar in appearance and identification requires sharp, wavy crests, with a band (the girdle) encircling examination of the ornamentation of mature megaspores the spore that lacks ridges and is covered with tiny under a microscope. -

Earliest Record of Megaphylls and Leafy Structures, and Their Initial Diversification
Review Geology August 2013 Vol.58 No.23: 27842793 doi: 10.1007/s11434-013-5799-x Earliest record of megaphylls and leafy structures, and their initial diversification HAO ShouGang* & XUE JinZhuang Key Laboratory of Orogenic Belts and Crustal Evolution, School of Earth and Space Sciences, Peking University, Beijing 100871, China Received January 14, 2013; accepted February 26, 2013; published online April 10, 2013 Evolutionary changes in the structure of leaves have had far-reaching effects on the anatomy and physiology of vascular plants, resulting in morphological diversity and species expansion. People have long been interested in the question of the nature of the morphology of early leaves and how they were attained. At least five lineages of euphyllophytes can be recognized among the Early Devonian fossil plants (Pragian age, ca. 410 Ma ago) of South China. Their different leaf precursors or “branch-leaf com- plexes” are believed to foreshadow true megaphylls with different venation patterns and configurations, indicating that multiple origins of megaphylls had occurred by the Early Devonian, much earlier than has previously been recognized. In addition to megaphylls in euphyllophytes, the laminate leaf-like appendages (sporophylls or bracts) occurred independently in several dis- tantly related Early Devonian plant lineages, probably as a response to ecological factors such as high atmospheric CO2 concen- trations. This is a typical example of convergent evolution in early plants. Early Devonian, euphyllophyte, megaphyll, leaf-like appendage, branch-leaf complex Citation: Hao S G, Xue J Z. Earliest record of megaphylls and leafy structures, and their initial diversification. Chin Sci Bull, 2013, 58: 27842793, doi: 10.1007/s11434- 013-5799-x The origin and evolution of leaves in vascular plants was phology and evolutionary diversification of early leaves of one of the most important evolutionary events affecting the basal euphyllophytes remain enigmatic. -

Huperzine a from Huperzia Species—An Ethnopharmacolgical Review Xiaoqiang Ma A,B, Changheng Tan A, Dayuan Zhu A, David R
Huperzine A from Huperzia species—An ethnopharmacolgical review Xiaoqiang Ma a,b, Changheng Tan a, Dayuan Zhu a, David R. Gang b, Peigen Xiao c,∗ a State Key Laboratory of Drug Research, Institute of Materia Medica, Shanghai Institutes for Biological Sciences, Chinese Academy of Sciences, Shanghai 201203, PR China b Department of Plant Sciences and BIO5 Institute, The University of Arizona, 303 Forbes Building, Tucson, AZ 85721-0036, USA c Institute of Medicinal Plant Development, Peking Union Medical College and Chinese Academy of Medical Sciences, Beijing 100094, PR China Abstract Huperzine A (HupA), isolated originally from a traditional Chinese medicine Qiang Ceng Ta, whole plant of Huperzia serrata (Thunb. ex Murray) Trev., a member of the Huperziaceae family, has attracted intense attention since its marked anticholinesterase activity was discovered by Chinese scientists. Several members of the Huperziaceae (Huperzia and Phlegmariurus species) have been used as medicines in China for contusions, strains, swellings, schizophrenia, myasthenia gravis and organophosphate poisoning. HupA has been marketed in China as a new drug for Alzheimer’s disease (AD) treatment and its derivative ZT-1 is being developed as anti-AD new drug candidate both in China and in Europe. A review of the chemistry, bioactivities, toxicology, clinical trials and natural resources of HupA source plants is presented. Keywords: Huperzine A; ZT-1; Alzheimer’s disease; Huperzia serrata; Huperziaceae; Drug discovery; Bioactivities; Clinical trials; Traditional Chinese -

Embryophytic Sporophytes in the Rhynie and Windyfield Cherts
Transactions of the Royal Society of Edinburgh: Earth Sciences http://journals.cambridge.org/TRE Additional services for Transactions of the Royal Society of Edinburgh: Earth Sciences: Email alerts: Click here Subscriptions: Click here Commercial reprints: Click here Terms of use : Click here Embryophytic sporophytes in the Rhynie and Windyeld cherts Dianne Edwards Transactions of the Royal Society of Edinburgh: Earth Sciences / Volume 94 / Issue 04 / December 2003, pp 397 - 410 DOI: 10.1017/S0263593300000778, Published online: 26 July 2007 Link to this article: http://journals.cambridge.org/abstract_S0263593300000778 How to cite this article: Dianne Edwards (2003). Embryophytic sporophytes in the Rhynie and Windyeld cherts. Transactions of the Royal Society of Edinburgh: Earth Sciences, 94, pp 397-410 doi:10.1017/S0263593300000778 Request Permissions : Click here Downloaded from http://journals.cambridge.org/TRE, IP address: 131.251.254.13 on 25 Feb 2014 Transactions of the Royal Society of Edinburgh: Earth Sciences, 94, 397–410, 2004 (for 2003) Embryophytic sporophytes in the Rhynie and Windyfield cherts Dianne Edwards ABSTRACT: Brief descriptions and comments on relationships are given for the seven embryo- phytic sporophytes in the cherts at Rhynie, Aberdeenshire, Scotland. They are Rhynia gwynne- vaughanii Kidston & Lang, Aglaophyton major D. S. Edwards, Horneophyton lignieri Barghoorn & Darrah, Asteroxylon mackiei Kidston & Lang, Nothia aphylla Lyon ex Høeg, Trichopherophyton teuchansii Lyon & Edwards and Ventarura lyonii Powell, Edwards & Trewin. The superb preserva- tion of the silica permineralisations produced in the hot spring environment provides remarkable insights into the anatomy of early land plants which are not available from compression fossils and other modes of permineralisation. -

Bibliography of Isoetes
BIBLIOGRAPHY OF ISOETES ALLEN, B.M. 1975. A note on the distribution of Isoetes in the Cadiz Province, Spain. Fern Gaz. (U.K.) 11 (2-3): 163-164 (1975). ALONSO, PAZ, E. 1989. Notas sobre plantas nuevas o interesantes para la flora Uruguaya: 1. (Notes on new or interesting plants for the Uruguayan flora: 1.) Comun. Bot. Mus. Hist. Nat. Montevideo 5 (91): 1-4 (1989) - Isoetes pp.2-3 ALSTON, A.H.G. 1982. Isoetaceae: 1. In Steenis, C.G.G.J. van, Holttum, R. E., eds. Flora Malesiana, series 2. Pteridophytes, volume 1. The Hague, Martinus Nijhoff, Dr. W. Junk Publ. 62-64 (1982)- illus., chrom. nos., key. ANDREIS, C., RODONDI, G. 1987. Alcune stazioni di Isoetes echinospora Dur. nel Bresciano e osservazioni al SEM delle spore delle Isoetes della flora Italica. Natura Bresciana no.23: 119-130 (1986 publ. 1987) - illus., maps. 4, ANTHONY, N.C., & E.A. SCHELPE, 1985. Two new taxa and a new combination in southern African Pteridophyta. Bothalia, 15 (3 & 4): 554-555 (1985) ARREGUIN-SANCHEZ, M., 1986. Nuevos registros y taxa interesantes de pteridofitas del Valle de Mexico. (Isoetaceae, Psilotaceae y Selaginellaceae) Phytologia 59 (7): 451-453 (1986) ASH, S., & K.B. PIGG. 1991. A new Jurassic Isoetites (Isoetales) from the Wallowa Terrane in Hells Canyon Oregon and Idaho. Amer. J. Bot. 78: 1636-1642. BAJPAI, U., & H.K. MAHESHWARI,1985. EM studies on the megaspores of Isoetes coromandelina. Phytomorphology, 34 (1-4): 226-231 (1984 publ. 1985) - illus. BALDWIN, W.K.W. 1933. The organization of the young sporophyte of Isoetes engelmanni, A. -

THE EVOLUTION of XYLEM ANATOMY in EARLY TRACHEOPHYTES by ELISABETH ANNE BERGMAN
Conquering the terrestrial environment: the evolution of xylem anatomy in early tracheophytes Item Type text; Electronic Thesis Authors Bergman, Elisabeth Anne Publisher The University of Arizona. Rights Copyright © is held by the author. Digital access to this material is made possible by the University Libraries, University of Arizona. Further transmission, reproduction or presentation (such as public display or performance) of protected items is prohibited except with permission of the author. Download date 27/09/2021 03:01:29 Item License http://rightsstatements.org/vocab/InC/1.0/ Link to Item http://hdl.handle.net/10150/626731 CONQUERING THE TERRESTRIAL ENVIRONMENT: THE EVOLUTION OF XYLEM ANATOMY IN EARLY TRACHEOPHYTES By ELISABETH ANNE BERGMAN ____________________ A Thesis Submitted to The Honors College In Partial Fulfillment of the Bachelors Degree With Honors in Biology with an Emphasis in Biomedical Sciences THE UNIVERSITY OF ARIZONA D E C E M B E R 2 0 1 7 Approved by: ____________________________ Dr. Brian Enquist Department of Ecology and Evolutionary Biology Acknowledgements Many thanks go to all of those who made contributions, big and small, to my honors thesis, and more notably, my education. Foremost, I thank Dr. Brian Enquist for accepting me into his lab and serving as my mentor for two years. I appreciate all of the time he put in to meet with me and help me to develop my honors thesis. Additional thanks go to Dr. Sean Michaletz who first introduced me to the work that would eventually become my honors thesis. From the University of Santa Cruz, California, I thank Dr.