Role of Intermediate Filament Desmin in Development of Desmin- Desmin in Development of Desmin- Related Myopathy Related Myopathy
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

A Case of Prenatal Diagnosis of 18P Deletion Syndrome Following
Zhao et al. Molecular Cytogenetics (2019) 12:53 https://doi.org/10.1186/s13039-019-0464-y CASE REPORT Open Access A case of prenatal diagnosis of 18p deletion syndrome following noninvasive prenatal testing Ganye Zhao, Peng Dai, Shanshan Gao, Xuechao Zhao, Conghui Wang, Lina Liu and Xiangdong Kong* Abstract Background: Chromosome 18p deletion syndrome is a disease caused by the complete or partial deletion of the short arm of chromosome 18, there were few cases reported about the prenatal diagnosis of 18p deletion syndrome. Noninvasive prenatal testing (NIPT) is widely used in the screening of common fetal chromosome aneuploidy. However, the segmental deletions and duplications should also be concerned. Except that some cases had increased nuchal translucency or holoprosencephaly, most of the fetal phenotype of 18p deletion syndrome may not be evident during the pregnancy, 18p deletion syndrome was always accidentally discovered during the prenatal examination. Case presentations: In our case, we found a pure partial monosomy 18p deletion during the confirmation of the result of NIPT by copy number variation sequencing (CNV-Seq). The result of NIPT suggested that there was a partial or complete deletion of X chromosome. The amniotic fluid karyotype was normal, but result of CNV-Seq indicated a 7.56 Mb deletion on the short arm of chromosome 18 but not in the couple, which means the deletion was de novo deletion. Finally, the parents chose to terminate the pregnancy. Conclusions: To our knowledge, this is the first case of prenatal diagnosis of 18p deletion syndrome following NIPT.NIPT combined with ultrasound may be a relatively efficient method to screen chromosome microdeletions especially for the 18p deletion syndrome. -

Large-Scale Opening of Utrophints Tandem Calponin Homology (CH
Large-scale opening of utrophin’s tandem calponin homology (CH) domains upon actin binding by an induced-fit mechanism Ava Y. Lin, Ewa Prochniewicz, Zachary M. James, Bengt Svensson, and David D. Thomas1 Department of Biochemistry, Molecular Biology and Biophysics, University of Minnesota, Minneapolis, MN 55455 Edited by James A. Spudich, Stanford University School of Medicine, Stanford, CA, and approved June 20, 2011 (received for review April 21, 2011) We have used site-directed spin labeling and pulsed electron has prevented the development of a reliable structural model for paramagnetic resonance to resolve a controversy concerning the any of these complexes. A major unresolved question concerns structure of the utrophin–actin complex, with implications for the the relative disposition of the tandem CH domains (CH1 and pathophysiology of muscular dystrophy. Utrophin is a homolog of CH2) (9, 10). Crystal structures of the tandem CH domains dystrophin, the defective protein in Duchenne and Becker muscular showed a closed conformation for fimbrin (11) and α-actinin (12), dystrophies, and therapeutic utrophin derivatives are currently but an open conformation for both utrophin (Utr261) (Fig. 1A) being developed. Both proteins have a pair of N-terminal calponin and dystrophin (Dys246) (16). The crystal structure of Utr261 homology (CH) domains that are important for actin binding. suggests that the central helical region connecting CH1 and CH2 Although there is a crystal structure of the utrophin actin-binding is highly flexible. Even for α-actinin, which has a closed crystal domain, electron microscopy of the actin-bound complexes has structure, computational analysis suggests the potential for a high produced two very different structural models, in which the CH do- degree of dynamic flexibility that facilitates actin binding (17). -
The Desmoplakin Carboxyl Terminus Coaligns with and Specifically Disrupts Intermediate Filament Networks When Expressed in Cultured Cells Thaddeus S
View metadata, citation and similar papers at core.ac.uk brought to you by CORE provided by PubMed Central The Desmoplakin Carboxyl Terminus Coaligns with and Specifically Disrupts Intermediate Filament Networks When Expressed in Cultured Cells Thaddeus S. Stappenbeck and Kathleen J. Green Department of Pathology and the Cancer Center, Northwestern University Medical School, Chicago, Illinois 60611 Abstract. Specific interactions between desmoplakins tides including the 90-kD carboxy-terminal globular I and 11 (DP I and II) and other desmosomal or cyto- domain of DP I specifically colocalized with and ulti- skeletal molecules have been difficult to determine in mately resulted in the complete disruption of IF in part because of the complexity and insolubility of the both cell lines. This effect was specific for IF as micro- desmosome and its constituents . We have used a mo- tubule and microfilament networks were unaltered . lecular genetic approach to investigate the role that This effect was also specific for the carboxyl terminus DP I and 11 may play in the association of the desmo- of DP, as the expression of the 95-kD rod domain of somal plaque with cytoplasmic intermediate filaments DP I did not visibly alter IF networks. Immunogold (IF) . A series of mammalian expression vectors en- localization of COS-7 cells transfected with constructs coding specific predicted domains of DP I were tran- including the carboxyl terminus of DP demonstrated siently expressed in cultured cells that form (COS-7) an accumulation of mutant protein in perinuclear aggre- and do not form (NIH-3T3) desmosomes. Sequence gates within which IF subunits were sequestered. -

Keratin 19 Regulates Cell Shape and Cell-Cell Adhesion of MCF7 Cells While Maintaining E-Cadherin Localization at the Cell Surface
Keratin 19 regulates cell shape and cell-cell adhesion of MCF7 cells while maintaining E-cadherin localization at the cell surface Welcome to my poster. This is Sarah Alsharif, a PhD student from the biology department. I am glad to present the work our lab has been doing in the breast cancer field. In fact, after lung cancer, breast cancer is the second cause of death in women worldwide (1). It is estimated that every 18 seconds, approximately one new case of breast cancer is documented (2). No one dies due to cancer itself. The death is because of metastasis which takes place when cancer cells leave a breast in which they are formed and reach other sites such as the brain or lung. Our lab is interested in investigating the mechanism behind metastasis of breast cancer. Metastasis is associated with what is called epithelial to mesenchymal transition (EMT), the process characterized by loss of cell to cell adhesion and expression of epithelial markers such as keratin intermediate filament proteins, as you can see in the first three images of cells. Those filaments are keratins and they are critical for the shape and for maintaining mechanical integrity of epithelial cells via cell to cell complexes called desmosomes. Among different keratins, keratin 19 (K19) is highly expressed in many types of cancer including breast cancer, and is correlated with a worse prognosis (3). Consistently, K19 expression has been reported to be significantly higher in metastatic breast cancer tumor cells compared to primary tumors (4). The role of K19 on mechanical properties of cancer cells for cell migration and possible impact on metastasis in breast cancer patients is still unknown. -

A Significant Soluble Keratin Fraction In
Journal of Cell Science 105, 433-444 (1993) 433 Printed in Great Britain © The Company of Biologists Limited 1993 A significant soluble keratin fraction in ‘simple’ epithelial cells Lack of an apparent phosphorylation and glycosylation role in keratin solubility Chih-Fong Chou*, Carrie L. Riopel, Lusijah S. Rott and M. Bishr Omary† Palo Alto Veterans Administration Medical Center and the Digestive Disease Center at Stanford University, School of Medicine, 3801 Miranda Avenue, GI 111, Palo Alto, CA 94304, USA *Author for reprint requests †Author for correspondence SUMMARY We studied the solubility of keratin polypeptides 8 and aments in vitro as determined by electron microscopy. 18 (K8/18), which are the predominant intermediate fil- Cross-linking of soluble K8/18 followed by immunopre- aments in the human colonic epithelial cell line HT29. cipitation resulted in dimeric and tetrameric forms, We find that asynchronously growing cells (G0/G1 stage based on migration in SDS-polyacrylamide gels. In of the cell cycle) have a substantial pool of soluble ker- addition, cross-linked and native soluble K8/18 showed atin that constitutes approx. 5% of total cellular ker- similar migration on nondenaturing gels and similar atin. This soluble keratin pool was observed after sedimentation after sucrose density gradient centrifu- immunoprecipitation of K8/18 from the cytosolic frac- gation. Our results indicate that simple epithelial ker- tion of cells disrupted using three detergent-free meth- atins are appreciably more soluble than previously rec- ods. Several other cell lines showed a similar significant ognized. The soluble keratin form is assembly competent soluble cytosolic K8/18 pool. -

Structural Biology of the Dystrophin-Deficient Muscle Fiber
Dystrophin-deficient muscle fiber REVIEW ISSN- 0102-9010145 STRUCTURAL BIOLOGY OF THE DYSTROPHIN-DEFICIENT MUSCLE FIBER Maria Julia Marques Department of Anatomy, Institute of Biology, State University of Campinas (UNICAMP), Campinas, SP, Brazil. ABSTRACT The discovery of dystrophin and its gene has led to major advances in our understanding of the molecular basis of Duchenne, Becker and other muscular dystrophies related to the dystrophin-associated protein complex. The concept that dystrophin has a mechanical function in stabilizing the muscle fiber membrane has expanded in the last five years. The dystrophin-glycoprotein complex is now considered a multifunctional complex that contains molecules involved in signal transduction cascades important for cell survival. The roles of dystrophin and the dystrophin- glycoprotein complex in positioning and anchoring receptors and ion channels is also important, and much of what is known about these functions is based on studies of the neuromuscular synapse. In this review, we discuss the components and the cellular signaling molecules associated with the dystrophin-glycoprotein complex. We then focus on the molecular organization of the neuromuscular junction and its structural organization in the dystrophin-deficient muscle fibers of mdx mice, a well-established experimental model of Duchenne muscular dystrophy. Key words: Confocal microscopy, Duchenne muscular dystrophy, mdx, neuromuscular junction INTRODUCTION that can improve the quality of life of the patients, Duchenne muscular dystrophy (DMD) is an X- death usually occurs in the early twenties, as a result linked recessive, progressive muscle-wasting disease of cardiac and/or respiratory failures [24]. that affects primarily skeletal and cardiac muscle. Several other muscular dystrophies have been DMD was first reported in 1868 by Dr. -

Genetic Mutations and Mechanisms in Dilated Cardiomyopathy
Genetic mutations and mechanisms in dilated cardiomyopathy Elizabeth M. McNally, … , Jessica R. Golbus, Megan J. Puckelwartz J Clin Invest. 2013;123(1):19-26. https://doi.org/10.1172/JCI62862. Review Series Genetic mutations account for a significant percentage of cardiomyopathies, which are a leading cause of congestive heart failure. In hypertrophic cardiomyopathy (HCM), cardiac output is limited by the thickened myocardium through impaired filling and outflow. Mutations in the genes encoding the thick filament components myosin heavy chain and myosin binding protein C (MYH7 and MYBPC3) together explain 75% of inherited HCMs, leading to the observation that HCM is a disease of the sarcomere. Many mutations are “private” or rare variants, often unique to families. In contrast, dilated cardiomyopathy (DCM) is far more genetically heterogeneous, with mutations in genes encoding cytoskeletal, nucleoskeletal, mitochondrial, and calcium-handling proteins. DCM is characterized by enlarged ventricular dimensions and impaired systolic and diastolic function. Private mutations account for most DCMs, with few hotspots or recurring mutations. More than 50 single genes are linked to inherited DCM, including many genes that also link to HCM. Relatively few clinical clues guide the diagnosis of inherited DCM, but emerging evidence supports the use of genetic testing to identify those patients at risk for faster disease progression, congestive heart failure, and arrhythmia. Find the latest version: https://jci.me/62862/pdf Review series Genetic mutations and mechanisms in dilated cardiomyopathy Elizabeth M. McNally, Jessica R. Golbus, and Megan J. Puckelwartz Department of Human Genetics, University of Chicago, Chicago, Illinois, USA. Genetic mutations account for a significant percentage of cardiomyopathies, which are a leading cause of conges- tive heart failure. -

Transgenic Cyclooxygenase-2 Overexpression Sensitizes Mouse Skin for Carcinogenesis
Transgenic cyclooxygenase-2 overexpression sensitizes mouse skin for carcinogenesis Karin Mu¨ ller-Decker*†, Gitta Neufang*, Irina Berger‡, Melanie Neumann*, Friedrich Marks*, and Gerhard Fu¨ rstenberger* *Research Program Tumor Cell Regulation, Deutsches Krebsforschungszentrum, and ‡Department of Pathology, Ruprecht-Karls-University, 69120 Heidelberg, Germany Edited by Philip Needleman, Pharmacia Corporation, St. Louis, MO, and approved July 29, 2002 (received for review May 30, 2002) Genetic and pharmacological evidence suggests that overexpres- there is a causal relationship between COX-2 overexpression sion of cyclooxygenase-2 (COX-2) is critical for epithelial carcino- and tumor development. Recently, we have shown that the genesis and provides a major target for cancer chemoprevention keratin 5 (K5) promoter-driven overexpression of COX-2 in by nonsteroidal antiinflammatory drugs. Transgenic mouse lines basal cells of interfollicular epidermis and the pilosebaceous unit with keratin 5 promoter-driven COX-2 overexpression in basal led to a preneoplastic skin phenotype in 4 of 4 high-expression epidermal cells exhibit a preneoplastic skin phenotype. As shown mouse lines (15). here, this phenotype depends on the level of COX-2 expression and To delineate COX-2 functions for carcinogenesis, we have COX-2-mediated prostaglandin accumulation. The transgenics did used the initiation–promotion model (2) for the induction of skin not develop skin tumors spontaneously but did so after a single tumors in wild-type (wt) NMRI mice and COX-2 transgenic application of an initiating dose of the carcinogen 7,12-dimethyl- mouse lines. This multistage model allows the analysis of the benz[a]anthracene (DMBA). Long-term treatment with the tumor carcinogenic process in terms of distinct stages, i.e., initiation by promoter phorbol 12-myristate 13-acetate, as required for tumor- application of a subcarcinogenic dose of a carcinogen such as igenesis in wild-type mice, was not necessary for transgenics. -

A Computational Approach for Defining a Signature of Β-Cell Golgi Stress in Diabetes Mellitus
Page 1 of 781 Diabetes A Computational Approach for Defining a Signature of β-Cell Golgi Stress in Diabetes Mellitus Robert N. Bone1,6,7, Olufunmilola Oyebamiji2, Sayali Talware2, Sharmila Selvaraj2, Preethi Krishnan3,6, Farooq Syed1,6,7, Huanmei Wu2, Carmella Evans-Molina 1,3,4,5,6,7,8* Departments of 1Pediatrics, 3Medicine, 4Anatomy, Cell Biology & Physiology, 5Biochemistry & Molecular Biology, the 6Center for Diabetes & Metabolic Diseases, and the 7Herman B. Wells Center for Pediatric Research, Indiana University School of Medicine, Indianapolis, IN 46202; 2Department of BioHealth Informatics, Indiana University-Purdue University Indianapolis, Indianapolis, IN, 46202; 8Roudebush VA Medical Center, Indianapolis, IN 46202. *Corresponding Author(s): Carmella Evans-Molina, MD, PhD ([email protected]) Indiana University School of Medicine, 635 Barnhill Drive, MS 2031A, Indianapolis, IN 46202, Telephone: (317) 274-4145, Fax (317) 274-4107 Running Title: Golgi Stress Response in Diabetes Word Count: 4358 Number of Figures: 6 Keywords: Golgi apparatus stress, Islets, β cell, Type 1 diabetes, Type 2 diabetes 1 Diabetes Publish Ahead of Print, published online August 20, 2020 Diabetes Page 2 of 781 ABSTRACT The Golgi apparatus (GA) is an important site of insulin processing and granule maturation, but whether GA organelle dysfunction and GA stress are present in the diabetic β-cell has not been tested. We utilized an informatics-based approach to develop a transcriptional signature of β-cell GA stress using existing RNA sequencing and microarray datasets generated using human islets from donors with diabetes and islets where type 1(T1D) and type 2 diabetes (T2D) had been modeled ex vivo. To narrow our results to GA-specific genes, we applied a filter set of 1,030 genes accepted as GA associated. -

Plakoglobin Is Required for Effective Intermediate Filament Anchorage to Desmosomes Devrim Acehan1, Christopher Petzold1, Iwona Gumper2, David D
ORIGINAL ARTICLE Plakoglobin Is Required for Effective Intermediate Filament Anchorage to Desmosomes Devrim Acehan1, Christopher Petzold1, Iwona Gumper2, David D. Sabatini2, Eliane J. Mu¨ller3, Pamela Cowin2,4 and David L. Stokes1,2,5 Desmosomes are adhesive junctions that provide mechanical coupling between cells. Plakoglobin (PG) is a major component of the intracellular plaque that serves to connect transmembrane elements to the cytoskeleton. We have used electron tomography and immunolabeling to investigate the consequences of PG knockout on the molecular architecture of the intracellular plaque in cultured keratinocytes. Although knockout keratinocytes form substantial numbers of desmosome-like junctions and have a relatively normal intercellular distribution of desmosomal cadherins, their cytoplasmic plaques are sparse and anchoring of intermediate filaments is defective. In the knockout, b-catenin appears to substitute for PG in the clustering of cadherins, but is unable to recruit normal levels of plakophilin-1 and desmoplakin to the plaque. By comparing tomograms of wild type and knockout desmosomes, we have assigned particular densities to desmoplakin and described their interaction with intermediate filaments. Desmoplakin molecules are more extended in wild type than knockout desmosomes, as if intermediate filament connections produced tension within the plaque. On the basis of our observations, we propose a particular assembly sequence, beginning with cadherin clustering within the plasma membrane, followed by recruitment of plakophilin and desmoplakin to the plaque, and ending with anchoring of intermediate filaments, which represents the key to adhesive strength. Journal of Investigative Dermatology (2008) 128, 2665–2675; doi:10.1038/jid.2008.141; published online 22 May 2008 INTRODUCTION dense plaque that is further from the membrane and that Desmosomes are large macromolecular complexes that mediates the binding of intermediate filaments. -

Transiently Structured Head Domains Control Intermediate Filament Assembly
Transiently structured head domains control intermediate filament assembly Xiaoming Zhoua, Yi Lina,1, Masato Katoa,b,c, Eiichiro Morid, Glen Liszczaka, Lillian Sutherlanda, Vasiliy O. Sysoeva, Dylan T. Murraye, Robert Tyckoc, and Steven L. McKnighta,2 aDepartment of Biochemistry, University of Texas Southwestern Medical Center, Dallas, TX 75390; bInstitute for Quantum Life Science, National Institutes for Quantum and Radiological Science and Technology, 263-8555 Chiba, Japan; cLaboratory of Chemical Physics, National Institute of Diabetes and Digestive and Kidney Diseases, National Institutes of Health, Bethesda, MD 20892-0520; dDepartment of Future Basic Medicine, Nara Medical University, 840 Shijo-cho, Kashihara, Nara, Japan; and eDepartment of Chemistry, University of California, Davis, CA 95616 Contributed by Steven L. McKnight, January 2, 2021 (sent for review October 30, 2020; reviewed by Lynette Cegelski, Tatyana Polenova, and Natasha Snider) Low complexity (LC) head domains 92 and 108 residues in length are, IF head domains might facilitate filament assembly in a manner respectively, required for assembly of neurofilament light (NFL) and analogous to LC domain function by RNA-binding proteins in the desmin intermediate filaments (IFs). As studied in isolation, these IF assembly of RNA granules. head domains interconvert between states of conformational disor- IFs are defined by centrally located α-helical segments 300 to der and labile, β-strand–enriched polymers. Solid-state NMR (ss-NMR) 350 residues in length. These central, α-helical segments are spectroscopic studies of NFL and desmin head domain polymers re- flanked on either end by head and tail domains thought to be veal spectral patterns consistent with structural order. -
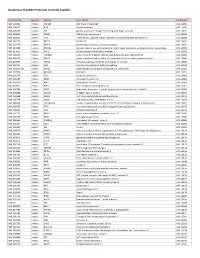
Quantigene Flowrna Probe Sets Currently Available
QuantiGene FlowRNA Probe Sets Currently Available Accession No. Species Symbol Gene Name Catalog No. NM_003452 Human ZNF189 zinc finger protein 189 VA1-10009 NM_000057 Human BLM Bloom syndrome VA1-10010 NM_005269 Human GLI glioma-associated oncogene homolog (zinc finger protein) VA1-10011 NM_002614 Human PDZK1 PDZ domain containing 1 VA1-10015 NM_003225 Human TFF1 Trefoil factor 1 (breast cancer, estrogen-inducible sequence expressed in) VA1-10016 NM_002276 Human KRT19 keratin 19 VA1-10022 NM_002659 Human PLAUR plasminogen activator, urokinase receptor VA1-10025 NM_017669 Human ERCC6L excision repair cross-complementing rodent repair deficiency, complementation group 6-like VA1-10029 NM_017699 Human SIDT1 SID1 transmembrane family, member 1 VA1-10032 NM_000077 Human CDKN2A cyclin-dependent kinase inhibitor 2A (melanoma, p16, inhibits CDK4) VA1-10040 NM_003150 Human STAT3 signal transducer and activator of transcripton 3 (acute-phase response factor) VA1-10046 NM_004707 Human ATG12 ATG12 autophagy related 12 homolog (S. cerevisiae) VA1-10047 NM_000737 Human CGB chorionic gonadotropin, beta polypeptide VA1-10048 NM_001017420 Human ESCO2 establishment of cohesion 1 homolog 2 (S. cerevisiae) VA1-10050 NM_197978 Human HEMGN hemogen VA1-10051 NM_001738 Human CA1 Carbonic anhydrase I VA1-10052 NM_000184 Human HBG2 Hemoglobin, gamma G VA1-10053 NM_005330 Human HBE1 Hemoglobin, epsilon 1 VA1-10054 NR_003367 Human PVT1 Pvt1 oncogene homolog (mouse) VA1-10061 NM_000454 Human SOD1 Superoxide dismutase 1, soluble (amyotrophic lateral sclerosis 1 (adult))