Table of Contents
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Experiment 5: Molar Volume of a Gas (Mg + Hcl)
1 CHEM 30A EXPERIMENT 5: MOLAR VOLUME OF A GAS (MG + HCL) Learning Outcomes Upon completion of this lab, the student will be able to: 1) Demonstrate a single replacement reaction. 2) Calculate the molar volume of a gas at STP using experimental data. 3) Calculate the molar mass of a metal using experimental data. Introduction Metals that are above hydrogen in the activity series will displace hydrogen from an acid and produce hydrogen gas. Magnesium is an example of a metal that is more active than hydrogen in the activity series. The reaction between magnesium metal and aqueous hydrochloric acid is an example of a single replacement reaction (a type of redox reaction). The chemical equation for this reaction is shown below: Mg(s) + 2HCl(aq) è MgCl2(aq) + H2(g) Equation 1 When the reaction between the metal and the acid is conducted in a eudiometer, the volume of the hydrogen gas produced can be easily determined. In the experiment described below magnesium metal will be reacted with an excess of hydrochloric acid and the volume of hydrogen gas produced at the experimental conditions will be determined. According to Avogadro’s law, the volume of one mole of any gas at Standard Temperature and Pressure (STP = 273 K and 1 atm) is 22.4 L. Two important Gas Laws are required in order to convert the experimentally determined volume of hydrogen gas to that at STP. 1. Dalton’s law of partial pressures. 2. Combined gas law. Dalton’s Law of Partial Pressures According to Dalton’s law of partial pressures in a mixture of non-reacting gases, the total pressure exerted is equal to the sum of the partial pressures of the individual gases. -

Use of Laboratory Equipement
USE OF LABORATORY EQUIPEMENT C. Laboratory Thermometers Most thermometers are based upon the principle that liquids expand when heated. Most common thermometers use mercury or colored alcohol as the liquid. These thermometers are constructed as that a uniform-diameter capillary tube surmounts a liquid reservoir. To calibrate a thermometer, one defines two reference points, normally the freezing point of water (0°C, 32°F) and the boiling point of water (100°C, 212°F) at 1 tam of pressure (1 tam = 760 mm Hg). Once these points are marked on the capillary, its length is then subdivided into uniform divisions called degrees. There are 100° between these two points on the Celsius (°C, or centigrade) scale and 180° between those two points on the Fahrenheit (°F) scale. °F = 1.8 °C + 32 The Thermometer and Its Calibration This section describes the proper technique for checking the accuracy of your thermometer. These measurements will show how measured temperatures (read from thermometer) compare with true temperatures (the boiling and freezing points of water). The freezing point of water is 0°C; the boiling point depends upon atmospheric pressure but at sea level it is 100°C. Option 1: Place approximately 50 mL of ice in a 250-mL beaker and cover the ice with distilled water. Allow about 15 min for the mixture to come to equilibrium and then measure and record the temperature of the mixture. Theoretically, this temperature is 0°C. Option 2: Set up a 250-mL beaker on a wire gauze and iron ring. Fill the beaker about half full with distilled water. -

Determination of the Identity of an Unknown Liquid Group # My Name the Date My Period Partner #1 Name Partner #2 Name
Determination of the Identity of an unknown liquid Group # My Name The date My period Partner #1 name Partner #2 name Purpose: The purpose of this lab is to determine the identity of an unknown liquid by measuring its density, melting point, boiling point, and solubility in both water and alcohol, and then comparing the results to the values for known substances. Procedure: 1) Density determination Obtain a 10mL sample of the unknown liquid using a graduated cylinder Determine the mass of the 10mL sample Save the sample for further use 2) Melting point determination Set up an ice bath using a 600mL beaker Obtain a ~5mL sample of the unknown liquid in a clean dry test tube Place a thermometer in the test tube with the sample Place the test tube in the ice water bath Watch for signs of crystallization, noting the temperature of the sample when it occurs Save the sample for further use 3) Boiling point determination Set up a hot water bath using a 250mL beaker Begin heating the water in the beaker Obtain a ~10mL sample of the unknown in a clean, dry test tube Add a boiling stone to the test tube with the unknown Open the computer interface software, using a graph and digit display Place the temperature sensor in the test tube so it is in the unknown liquid Record the temperature of the sample in the test tube using the computer interface Watch for signs of boiling, noting the temperature of the unknown Dispose of the sample in the assigned waste container 4) Solubility determination Obtain two small (~1mL) samples of the unknown in two small test tubes Add an equal amount of deionized into one of the samples Add an equal amount of ethanol into the other Mix both samples thoroughly Compare the samples for solubility Dispose of the samples in the assigned waste container Observations: The unknown is a clear, colorless liquid. -

Ear and Forehead Thermometer
Ear and Forehead Thermometer INSTRUCTION MANUAL Item No. 91807 19.PJN174-14_GA-USA_HHD-Ohrthermometer_DSO364_28.07.14 Montag, 28. Juli 2014 14:37:38 USAD TABLE OF CONTENTS No. Topic Page 1.0 Definition of symbols 5 2.0 Application and functionality 6 2.1 Intended use 6 2.2 Field of application 7 3.0 Safety instructions 7 3.1 General safety instructions 7 3.3 Environment for which the DSO 364 device is not suited 9 3.4 Usage by children and adolescents 10 3.5 Information on the application of the device 10 4.0 Questions concerning body temperature 13 4.1 What is body temperature? 13 4.2 Advantages of measuring the body tempera- ture in the ear 14 4.3 Information on measuring the body tempera- ture in the ear 15 5.0 Scope of delivery / contents 16 6.0 Designation of device parts 17 7.0 LCD display 18 8.0 Basic functions 19 8.1 Commissioning of the device 19 8.2 Warning indicator if the body temperature is 21 too high 2 19.PJN174-14_GA-USA_HHD-Ohrthermometer_DSO364_28.07.14 Montag, 28. Juli 2014 14:37:38 TABLE OF CONTENTS USA No. Topic Page 8.3 Backlighting / torch function 21 8.4 Energy saving mode 22 8.5 Setting °Celsius / °Fahrenheit 22 9.0 Display / setting time and date 23 9.1 Display of time and date 23 9.2 Setting time and date 23 10.0 Memory mode 26 11.0 Measuring the temperature in the ear 28 12.0 Measuring the temperature on the forehead 30 13.0 Object temperature measurement 32 14.0 Disposal of the device 33 15.0 Battery change and information concerning batteries 33 16.0 Cleaning and care 36 17.0 “Cleaning” warning indicator 37 18.0 Calibration 38 19.0 Malfunctions 39 20.0 Technical specification 41 21.0 Warranty 44 3 19.PJN174-14_GA-USA_HHD-Ohrthermometer_DSO364_28.07.14 Montag, 28. -

Laboratory Equipment Reference Sheet
Laboratory Equipment Stirring Rod: Reference Sheet: Iron Ring: Description: Glass rod. Uses: To stir combinations; To use in pouring liquids. Evaporating Dish: Description: Iron ring with a screw fastener; Several Sizes Uses: To fasten to the ring stand as a support for an apparatus Description: Porcelain dish. Buret Clamp/Test Tube Clamp: Uses: As a container for small amounts of liquids being evaporated. Glass Plate: Description: Metal clamp with a screw fastener, swivel and lock nut, adjusting screw, and a curved clamp. Uses: To hold an apparatus; May be fastened to a ring stand. Mortar and Pestle: Description: Thick glass. Uses: Many uses; Should not be heated Description: Heavy porcelain dish with a grinder. Watch Glass: Uses: To grind chemicals to a powder. Spatula: Description: Curved glass. Uses: May be used as a beaker cover; May be used in evaporating very small amounts of Description: Made of metal or porcelain. liquid. Uses: To transfer solid chemicals in weighing. Funnel: Triangular File: Description: Metal file with three cutting edges. Uses: To scratch glass or file. Rubber Connector: Description: Glass or plastic. Uses: To hold filter paper; May be used in pouring Description: Short length of tubing. Medicine Dropper: Uses: To connect parts of an apparatus. Pinch Clamp: Description: Glass tip with a rubber bulb. Uses: To transfer small amounts of liquid. Forceps: Description: Metal clamp with finger grips. Uses: To clamp a rubber connector. Test Tube Rack: Description: Metal Uses: To pick up or hold small objects. Beaker: Description: Rack; May be wood, metal, or plastic. Uses: To hold test tubes in an upright position. -

LOWERING COSTS and SIMPLIFYING WORKFLOW with the Automate 2550 SYSTEM
HACETTEPE UNIVERSITY FACULTY OF MEDICINE HOSPITAL CENTRAL AND STAT LABORATORIES LOWERING COSTS AND SIMPLIFYING WORKFLOW WITH THE AutoMate 2550 SYSTEM Laboratory profile › 1,040-bed hospital located in Ankara, Turkey › Provides uninterrupted 24/7 inpatient, outpatient and emergency services › Employs 6 professors, 6 associate Hacettepe University Faculty of Medicine Hospital is one of the leading professors and 130 operators university hospitals in Turkey. The university is well known for its › Performs up to 6.5 million geographical location and adoption of ground breaking technologies. biochemistry and hormone While the reference site is preferred by a large number of patients, it is tests annually of particular interest to specific patient populations seeking test results › Processes up to 2 million tests from a variety of panels. With many different systems performing on instruments in more than hundreds of standard and specialized tests, the hospital’s laboratories 20 different units are driven to ensure the delivery of results without error, while › Operates a Beckman Coulter containing costs and managing workload burden. standalone AutoMate 2550 pre- For more than 16 years, the facility—which has been held up as an and post-analytical automation system in the biochemistry example of excellence in its own region, as well as all over the country— laboratory, along with a Power has been using a standalone automation system with an aliquoter Processor system with two feature. With this technology, it has enhanced patient satisfaction, -
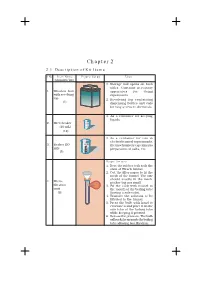
Chapter 2 2.1 Description of Kit Items
Manual of SEAT Friendly MCL Kit 7 Chapter 2 2.1 Description of Kit Items S.No. Item Name Figure/Setup Uses (Quantity/ kit) 1. Storage box opens on both sides. Contains necessary 1. Wooden box apparatus for doing with revolving experiments. top 2. Revolving top containing (1) dispensing bottles and vials for easy access to chemicals. 1. As a container for keeping liquids. 2. Microbeaker (10 mL) (12) 1. As a container for use in electrochemical experiments, 3. Beaker (50 thermochemistry experiments mL) preparation of salts, etc. (3) Steps for use 1. Bore the rubber cork to fit the stem of Hirsch funnel. 2. Cut the filter paper to fit the mesh of the funnel. The size should exactly fit the mesh, 4. Micro- neither big nor small. filtration 3. Fit the cork with funnel in unit the mouth of the boiling tube (2) (having a side tube). 4. Transfer the solution to be filtrated to the funnel. 5. Press the bulb with hand to evacuate it and place it on the side tube of the boiling tube while keeping it pressed. 6. Release the pressure. The bulb will suck the air inside the boiling tube allowing fast filtration. 8 Manual of SEAT Friendly MCL Kit S.No. Item Name Figure/Setup Uses (Quantity/Kit) 1. The watch glass is used to hold solids when being 5. Watch glass weighed. (2) 2. It should never be heated. 1. Flat plate with multiple “wells” used as micro test tubes. 2. Contains 96 rectangular matrix. 3. Each well of a micro plate typically holds somewhere between a few to a few hundred microlitres of liquid. -

Laboratory Supplies and Equipment
Laboratory Supplies and Equipment Beakers: 9 - 12 • Beakers with Handles • Printed Square Ratio Beakers • Griffin Style Molded Beakers • Tapered PP, PMP & PTFE Beakers • Heatable PTFE Beakers Bottles: 17 - 32 • Plastic Laboratory Bottles • Rectangular & Square Bottles Heatable PTFE Beakers Page 12 • Tamper Evident Plastic Bottles • Concertina Collapsible Bottle • Plastic Dispensing Bottles NEW Straight-Side Containers • Plastic Wash Bottles PETE with White PP Closures • PTFE Bottle Pourers Page 39 Containers: 38 - 42 • Screw Cap Plastic Jars & Containers • Snap Cap Plastic Jars & Containers • Hinged Lid Plastic Containers • Dispensing Plastic Containers • Graduated Plastic Containers • Disposable Plastic Containers Cylinders: 45 - 48 • Clear Plastic Cylinder, PMP • Translucent Plastic Cylinder, PP • Short Form Plastic Cylinder, PP • Four Liter Plastic Cylinder, PP NEW Polycarbonate Graduated Bottles with PP Closures Page 21 • Certified Plastic Cylinder, PMP • Hydrometer Jar, PP • Conical Shape Plastic Cylinder, PP Disposal Boxes: 54 - 55 • Bio-bin Waste Disposal Containers • Glass Disposal Boxes • Burn-upTM Bins • Plastic Recycling Boxes • Non-Hazardous Disposal Boxes Printed Cylinders Page 47 Drying Racks: 55 - 56 • Kartell Plastic Drying Rack, High Impact PS • Dynalon Mega-Peg Plastic Drying Rack • Azlon Epoxy Coated Drying Rack • Plastic Draining Baskets • Custom Size Drying Racks Available Burn-upTM Bins Page 54 Dynalon® Labware Table of Contents and Introduction ® Dynalon Labware, a leading wholesaler of plastic lab supplies throughout -

Infrared Forehead Thermometer (4DET-306) Quick Start Guide
Infrared Forehead Thermometer (4DET-306) Quick Start Guide To take a forehead temperature: 1” (2-3cm) 1 Press the power button , wait for 2 beeps. 2 Aim the thermometer at the center of the forehead about 1 inch (2-3 cm) from the skin and press the start button S T A R T . 2 START If the body temperature is below 100°F (38.7°C) you will hear one short beep and a 1 happy face will appear on the display. If the body temperature is above 100°F (38.7°C) you will hear one long beep followed by three short beeps and a sad face will appear on the display. Detailed instructions are included in the packaging Temperature Taking Hints and Tips • All body site temperatures are not the same. The • Temporal artery temperature changes faster than a temperature taken at different sites on the body will vary temperature taken rectally, orally or under the arm. A significantly. A forehead (temporal) temperature, for difference of 1°F (.6°C) is normal. Body temperature can example, is usually 0.5°F (0.3°C) to 1.0°F (0.6°C) lower change throughout the day. than an oral temperature. • Consistently low temperature readings may be caused by a • Make certain the thermometer is in FOREHEAD MODE. dirty instrument window. Use an alcohol wipe or a cotton • Non-contact infrared thermometers measure surface swab moistened with alcohol (70% isopropyl) to clean the temperature. Hold the device approximately 1 inch (2-3 instrument window and housing. Gently wipe and let air dry. -

High School Chemistry
RECOMMENDED MINIMUM CORE INVENTORY TO SUPPORT STANDARDS-BASED INSTRUCTION HIGH SCHOOL GRADES SCIENCES High School Chemistry Quantity per Quantity per lab classroom/ Description group adjacent work area SAFETY EQUIPMENT 2 Acid storage cabinet (one reserved exclusively for nitric acid) 1 Chemical spill kit 1 Chemical storage reference book 5 Chemical waste containers (Categories: corrosives, flammables, oxidizers, air/water reactive, toxic) 1 Emergency shower 1 Eye wash station 1 Fire blanket 1 Fire extinguisher 1 First aid kit 1 Flammables cabinet 1 Fume hood 1/student Goggles 1 Goggles sanitizer (holds 36 pairs of goggles) 1/student Lab aprons COMPUTER ASSISTED LEARNING 1 Television or digital projector 1 VGA Adapters for various digital devices EQUIPMENT/SUPPLIES 1 box Aluminum foil 100 Assorted rubber stoppers 1 Balance, analytical (0.001g precision) 5 Balance, electronic or manual (0.01g precision) 1 pkg of 50 Balloons, latex 4 Beakers, 50 mL 4 Beakers, 100 mL 2 Beakers, 250 mL Developed by California Science Teachers Association to support the implementation of the California Next Generation Science Standards. Approved by the CSTA Board of Directors November 17, 2015. Quantity per Quantity per lab classroom/ Description group adjacent work area 2 Beakers, 400 or 600 mL 1 Beakers, 1000 mL 1 Beaker tongs 1 Bell jar 4 Bottle, carboy round, LDPE 10 L 4 Bottle, carboy round, LDPE 4 L 10 Bottle, narrow mouth, 1000 mL 20 Bottle, narrow mouth, 125 mL 20 Bottle, narrow mouth, 250 mL 20 Bottle, narrow mouth, 500 mL 10 Bottle, wide mouth, 125 -

Model 914-604 Infra-Red Thermometer
Not recommended for use in measuring shiny or polished metal surfaces (stainless steel, aluminum, etc.) The reading will not be accurate. It is not possible to take measurements through transparent materials (glass, Plexiglas, etc.). It will Model 914-604 measure the temperature of the transparent surface Infra-red thermometer instead. It is not possible to measure air temperatures. Measurement errors can occur due to air contaminated with dust, steam, smoke, etc. Battery Replacement: Battery is low and no more measurements are possible. Replace the battery immediately with a CR2032 lithium button cell. 1- Power off the thermometer before replacing the battery A malfunction may occur if the power is on during battery replacement. If a malfunction occurs, restart the device. 2-Twist the battery cover on the back of thermometer to loosen the cover and remove the old battery. A coin will work well. 3- The new CR2032 battery is installed positive side up. Take care that the metal contacts at the top of the battery Congratulations on your purchase of the Infrared compartment are not bent. Thermometer! One-click for a temperature reading. Metal Contacts Metal Contacts No surface contact required, prevents contamination. Take a Measurement: 4-Replace the battery cover and tighten. Place the thermometer close to the item to measure. Click the button once to measure the surface temperature. The Troubleshooting: reading will display for 15 seconds. Problem Solution Hold the button for ongoing readings. No Display: Press the button Auto Shut Off: Ensure battery polarity is correct When the button has not been pressed for 15 seconds, the Change the battery thermometer will automatically shut off to save power. -

ELISA Technical Guide Table of Contents
ELISA Technical Guide Table of contents Introduction ................................................................................................................3 ELISA technology ......................................................................................................4 ELISA components ....................................................................................................6 ELISA equipment .......................................................................................................7 Equipment maintenance and calibration ..................................................................8 Reagent handling and preparation ...........................................................................9 Test component handling and preparation .............................................................10 Quality control .........................................................................................................11 Sample handling ......................................................................................................12 Pipetting methods ...................................................................................................14 ELISA plate timing ...................................................................................................16 ELISA plate washing ................................................................................................17 Plate reading and data management ......................................................................19 ELISA