E2F, HSF2, and Mir-26 in Thyroid Carcinoma: Bioinformatic Analysis of RNA-Sequencing Data
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Multifactorial Erβ and NOTCH1 Control of Squamous Differentiation and Cancer
Multifactorial ERβ and NOTCH1 control of squamous differentiation and cancer Yang Sui Brooks, … , Karine Lefort, G. Paolo Dotto J Clin Invest. 2014;124(5):2260-2276. https://doi.org/10.1172/JCI72718. Research Article Oncology Downmodulation or loss-of-function mutations of the gene encoding NOTCH1 are associated with dysfunctional squamous cell differentiation and development of squamous cell carcinoma (SCC) in skin and internal organs. While NOTCH1 receptor activation has been well characterized, little is known about how NOTCH1 gene transcription is regulated. Using bioinformatics and functional screening approaches, we identified several regulators of the NOTCH1 gene in keratinocytes, with the transcription factors DLX5 and EGR3 and estrogen receptor β (ERβ) directly controlling its expression in differentiation. DLX5 and ERG3 are required for RNA polymerase II (PolII) recruitment to the NOTCH1 locus, while ERβ controls NOTCH1 transcription through RNA PolII pause release. Expression of several identified NOTCH1 regulators, including ERβ, is frequently compromised in skin, head and neck, and lung SCCs and SCC-derived cell lines. Furthermore, a keratinocyte ERβ–dependent program of gene expression is subverted in SCCs from various body sites, and there are consistent differences in mutation and gene-expression signatures of head and neck and lung SCCs in female versus male patients. Experimentally increased ERβ expression or treatment with ERβ agonists inhibited proliferation of SCC cells and promoted NOTCH1 expression and squamous differentiation both in vitro and in mouse xenotransplants. Our data identify a link between transcriptional control of NOTCH1 expression and the estrogen response in keratinocytes, with implications for differentiation therapy of squamous cancer. Find the latest version: https://jci.me/72718/pdf Research article Multifactorial ERβ and NOTCH1 control of squamous differentiation and cancer Yang Sui Brooks,1,2 Paola Ostano,3 Seung-Hee Jo,1,2 Jun Dai,1,2 Spiro Getsios,4 Piotr Dziunycz,5 Günther F.L. -

A Computational Approach for Defining a Signature of Β-Cell Golgi Stress in Diabetes Mellitus
Page 1 of 781 Diabetes A Computational Approach for Defining a Signature of β-Cell Golgi Stress in Diabetes Mellitus Robert N. Bone1,6,7, Olufunmilola Oyebamiji2, Sayali Talware2, Sharmila Selvaraj2, Preethi Krishnan3,6, Farooq Syed1,6,7, Huanmei Wu2, Carmella Evans-Molina 1,3,4,5,6,7,8* Departments of 1Pediatrics, 3Medicine, 4Anatomy, Cell Biology & Physiology, 5Biochemistry & Molecular Biology, the 6Center for Diabetes & Metabolic Diseases, and the 7Herman B. Wells Center for Pediatric Research, Indiana University School of Medicine, Indianapolis, IN 46202; 2Department of BioHealth Informatics, Indiana University-Purdue University Indianapolis, Indianapolis, IN, 46202; 8Roudebush VA Medical Center, Indianapolis, IN 46202. *Corresponding Author(s): Carmella Evans-Molina, MD, PhD ([email protected]) Indiana University School of Medicine, 635 Barnhill Drive, MS 2031A, Indianapolis, IN 46202, Telephone: (317) 274-4145, Fax (317) 274-4107 Running Title: Golgi Stress Response in Diabetes Word Count: 4358 Number of Figures: 6 Keywords: Golgi apparatus stress, Islets, β cell, Type 1 diabetes, Type 2 diabetes 1 Diabetes Publish Ahead of Print, published online August 20, 2020 Diabetes Page 2 of 781 ABSTRACT The Golgi apparatus (GA) is an important site of insulin processing and granule maturation, but whether GA organelle dysfunction and GA stress are present in the diabetic β-cell has not been tested. We utilized an informatics-based approach to develop a transcriptional signature of β-cell GA stress using existing RNA sequencing and microarray datasets generated using human islets from donors with diabetes and islets where type 1(T1D) and type 2 diabetes (T2D) had been modeled ex vivo. To narrow our results to GA-specific genes, we applied a filter set of 1,030 genes accepted as GA associated. -

Transcriptional Control of Tissue-Resident Memory T Cell Generation
Transcriptional control of tissue-resident memory T cell generation Filip Cvetkovski Submitted in partial fulfillment of the requirements for the degree of Doctor of Philosophy in the Graduate School of Arts and Sciences COLUMBIA UNIVERSITY 2019 © 2019 Filip Cvetkovski All rights reserved ABSTRACT Transcriptional control of tissue-resident memory T cell generation Filip Cvetkovski Tissue-resident memory T cells (TRM) are a non-circulating subset of memory that are maintained at sites of pathogen entry and mediate optimal protection against reinfection. Lung TRM can be generated in response to respiratory infection or vaccination, however, the molecular pathways involved in CD4+TRM establishment have not been defined. Here, we performed transcriptional profiling of influenza-specific lung CD4+TRM following influenza infection to identify pathways implicated in CD4+TRM generation and homeostasis. Lung CD4+TRM displayed a unique transcriptional profile distinct from spleen memory, including up-regulation of a gene network induced by the transcription factor IRF4, a known regulator of effector T cell differentiation. In addition, the gene expression profile of lung CD4+TRM was enriched in gene sets previously described in tissue-resident regulatory T cells. Up-regulation of immunomodulatory molecules such as CTLA-4, PD-1, and ICOS, suggested a potential regulatory role for CD4+TRM in tissues. Using loss-of-function genetic experiments in mice, we demonstrate that IRF4 is required for the generation of lung-localized pathogen-specific effector CD4+T cells during acute influenza infection. Influenza-specific IRF4−/− T cells failed to fully express CD44, and maintained high levels of CD62L compared to wild type, suggesting a defect in complete differentiation into lung-tropic effector T cells. -

Development and Validation of a Novel Immune-Related Prognostic Model
Wang et al. J Transl Med (2020) 18:67 https://doi.org/10.1186/s12967-020-02255-6 Journal of Translational Medicine RESEARCH Open Access Development and validation of a novel immune-related prognostic model in hepatocellular carcinoma Zheng Wang1, Jie Zhu1, Yongjuan Liu3, Changhong Liu2, Wenqi Wang2, Fengzhe Chen1* and Lixian Ma1* Abstract Background: Growing evidence has suggested that immune-related genes play crucial roles in the development and progression of hepatocellular carcinoma (HCC). Nevertheless, the utility of immune-related genes for evaluating the prognosis of HCC patients are still lacking. The study aimed to explore gene signatures and prognostic values of immune-related genes in HCC. Methods: We comprehensively integrated gene expression data acquired from 374 HCC and 50 normal tissues in The Cancer Genome Atlas (TCGA). Diferentially expressed genes (DEGs) analysis and univariate Cox regression analysis were performed to identify DEGs that related to overall survival. An immune prognostic model was constructed using the Lasso and multivariate Cox regression analyses. Furthermore, Cox regression analysis was applied to identify independent prognostic factors in HCC. The correlation analysis between immune-related signature and immune cells infltration were also investigated. Finally, the signature was validated in an external independent dataset. Results: A total of 329 diferentially expressed immune‐related genes were detected. 64 immune‐related genes were identifed to be markedly related to overall survival in HCC patients using univariate Cox regression analysis. Then we established a TF-mediated network for exploring the regulatory mechanisms of these genes. Lasso and multivariate Cox regression analyses were applied to construct the immune-based prognostic model, which consisted of nine immune‐related genes. -

SUPPLEMENTAL FIGURE LEGENDS Supplemental Figure S1. RBPJ
Xie et al. SUPPLEMENTAL FIGURE LEGENDS Supplemental Figure S1. RBPJ correlates with BTIC marker expression. A-D. The TCGA GBM dataset was downloaded and correlations analyzed by R. RBPJ mRNA levels were highly correlated with (A) Olig2, (B) Sox2, (C) CD133, and (D) Sox4 levels. E. RBPJ is preferentially expressed in proneural glioblastomas. The glioblastoma TCGA dataset was interrogated for RBPJ mRNA expression segregated by transcriptional profile. The proneural tumors were further divided into G-CIMP (glioma CpG-island methylator phenotype) or non-G-CIMP. **, p < 0.01. ****, p < 0.0001. *****, p < 0.00001. Supplemental Figure S2. Targeting RBPJ induces BTIC apoptosis. A. 3691 BTICs were transduced with shCONT, shRBPJ-1, or shRBPJ-2. Lysates were prepared and immunoblotted with the indicated antibodies. shRNA-mediated knockdown of RBPJ was associated with increased cleaved (activated) PARP. B. 3691 BTICs were transduced with shCONT, shRBPJ-1, or shRBPJ-2. Apoptosis measured by Annexin V staining. Data are presented as mean ± SEM (two- way ANOVA; **, p < 0.01; n = 3). Supplemental Figure S3. Targeting RBPJ does not affect non-BTIC proliferation. Non-BTICs (Top, 3691; Bottom, 4121) were transduced with shCONT, shRBPJ-1, or shRBPJ-2. Cell proliferation was measured by CellTiter-Glo. 42 Xie et al. Supplemental Figure S4. RBPJ induces transcriptional profiles in BTICs distinct from Notch activation. A. In parallel experiments, 3691 BTICs were either treated with DAPT (at either 5 μM or 10 μM) vs. vehicle control (DMSO) or transduced with shRBPJ vs. shCONT. RNA-Seq was performed and the results displayed as a heat map with normalization to the relevant control. -

HSF1) During Macrophage Differentiation of Monocytes
Leukemia (2014) 28, 1676–1686 & 2014 Macmillan Publishers Limited All rights reserved 0887-6924/14 www.nature.com/leu ORIGINAL ARTICLE Dual regulation of SPI1/PU.1 transcription factor by heat shock factor 1 (HSF1) during macrophage differentiation of monocytes G Jego1,2, D Lanneau1,2, A De Thonel1,2, K Berthenet1,2, A Hazoume´ 1,2, N Droin3,4, A Hamman1,2, F Girodon1,2, P-S Bellaye1,2, G Wettstein1,2, A Jacquel1,2,5, L Duplomb6,7, A Le Moue¨l8,9, C Papanayotou10, E Christians11, P Bonniaud1,2, V Lallemand-Mezger8,9, E Solary3,4 and C Garrido1,2,12 In addition to their cytoprotective role in stressful conditions, heat shock proteins (HSPs) are involved in specific differentiation pathways, for example, we have identified a role for HSP90 in macrophage differentiation of human peripheral blood monocytes that are exposed to macrophage colony-stimulating factor (M-CSF). Here, we show that deletion of the main transcription factor involved in heat shock gene regulation, heat shock factor 1 (HSF1), affects M-CSF-driven differentiation of mouse bone marrow cells. HSF1 transiently accumulates in the nucleus of human monocytes undergoing macrophage differentiation, including M-CSF- treated peripheral blood monocytes and phorbol ester-treated THP1 cells. We demonstrate that HSF1 has a dual effect on SPI1/ PU.1, a transcription factor essential for macrophage differentiation and whose deregulation can lead to the development of leukemias and lymphomas. Firstly, HSF1 regulates SPI1/PU.1 gene expression through its binding to a heat shock element within the intron 2 of this gene. -

SOX4 Is Activated by C-MYC in Prostate Cancer
Medical Oncology (2019) 36:92 https://doi.org/10.1007/s12032-019-1317-6 ORIGINAL PAPER SOX4 is activated by C‑MYC in prostate cancer Hongyan Dong1,2 · Jing Hu1 · Lin Wang3 · Mei Qi1 · Ning Lu1 · Xiao Tan2 · Muyi Yang1 · Xinnuo Bai4 · Xuemei Zhan2 · Bo Han1,5 Received: 26 July 2019 / Accepted: 12 September 2019 / Published online: 27 September 2019 © Springer Science+Business Media, LLC, part of Springer Nature 2019 Abstract Although MYC proto-oncogene (C-MYC) amplifcation has been consistently reported to be a potential marker for prostate cancer (PCa) progression and prognosis, the clinicopathological and prognostic signifcance of C-MYC protein expression remains controversial. Overexpression of SOX4 has been shown to play important roles in multiple cancers including PCa. However, the link between these two critical genetic aberrations is unclear. In the current study, we showed that C-MYC was overexpressed in 16.2% (17/105) of Chinese patients with localized PCa. Overexpression of C-MYC was signifcantly associated with high Gleason scores (P = 0.012) and high Ki67 labeling index (P = 0.005). C-MYC overexpression was cor- related with cancer-related mortality and suggested to be an unfavorable prognostic factor in Chinese PCa patients (P = 0.018). Overexpression of C-MYC is associated with SOX4 overexpression in PCa tissues. Notably, SOX4 is a direct target gene of C-MYC; C-MYC activates SOX4 expression via binding to its promoter. In addition, Co-IP analysis demonstrated a physi- cal interaction between C-MYC and SOX4 protein in PCa cells. Clinically, C-MYC+/SOX4+ characterized poor prognosis in a subset of PCa patients. -

Whole-Exome Sequencing of Metastatic Cancer and Biomarkers of Treatment Response
Supplementary Online Content Beltran H, Eng K, Mosquera JM, et al. Whole-exome sequencing of metastatic cancer and biomarkers of treatment response. JAMA Oncol. Published online May 28, 2015. doi:10.1001/jamaoncol.2015.1313 eMethods eFigure 1. A schematic of the IPM Computational Pipeline eFigure 2. Tumor purity analysis eFigure 3. Tumor purity estimates from Pathology team versus computationally (CLONET) estimated tumor purities values for frozen tumor specimens (Spearman correlation 0.2765327, p- value = 0.03561) eFigure 4. Sequencing metrics Fresh/frozen vs. FFPE tissue eFigure 5. Somatic copy number alteration profiles by tumor type at cytogenetic map location resolution; for each cytogenetic map location the mean genes aberration frequency is reported eFigure 6. The 20 most frequently aberrant genes with respect to copy number gains/losses detected per tumor type eFigure 7. Top 50 genes with focal and large scale copy number gains (A) and losses (B) across the cohort eFigure 8. Summary of total number of copy number alterations across PM tumors eFigure 9. An example of tumor evolution looking at serial biopsies from PM222, a patient with metastatic bladder carcinoma eFigure 10. PM12 somatic mutations by coverage and allele frequency (A) and (B) mutation correlation between primary (y- axis) and brain metastasis (x-axis) eFigure 11. Point mutations across 5 metastatic sites of a 55 year old patient with metastatic prostate cancer at time of rapid autopsy eFigure 12. CT scans from patient PM137, a patient with recurrent platinum refractory metastatic urothelial carcinoma eFigure 13. Tracking tumor genomics between primary and metastatic samples from patient PM12 eFigure 14. -

Transcription Factor Gene Expression Profiling and Analysis of SOX Gene Family Transcription Factors in Human Limbal Epithelial
Transcription factor gene expression profiling and analysis of SOX gene family transcription factors in human limbal epithelial progenitor cells Der Naturwissenschaftlichen Fakultät der Friedrich-Alexander-Universität Erlangen-Nürnberg zur Erlangung des Doktorgrades Dr. rer. nat. vorgelegt von Dr. med. Johannes Menzel-Severing aus Bonn Als Dissertation genehmigt von der Naturwissenschaftlichen Fakultät der Friedrich-Alexander-Universität Erlangen-Nürnberg Tag der mündlichen Prüfung: 7. Februar 2018 Vorsitzender des Promotionsorgans: Prof. Dr. Georg Kreimer Gutachter: Prof. Dr. Andreas Feigenspan Prof. Dr. Ursula Schlötzer-Schrehardt 1 INDEX 1. ABSTRACTS Page 1.1. Abstract in English 4 1.2. Zusammenfassung auf Deutsch 7 2. INTRODUCTION 2.1. Anatomy and histology of the cornea and the corneal surface 11 2.2. Homeostasis of corneal epithelium and the limbal stem cell paradigm 13 2.3. The limbal stem cell niche 15 2.4. Cell therapeutic strategies in ocular surface disease 17 2.5. Alternative cell sources for transplantation to the corneal surface 18 2.6. Transcription factors in cell differentiation and reprogramming 21 2.7. Transcription factors in limbal epithelial cells 22 2.8. Research question 25 3. MATERIALS AND METHODS 3.1. Human donor corneas 27 3.2. Laser Capture Microdissection (LCM) 28 3.3. RNA amplification and RT2 profiler PCR arrays 29 3.4. Real-time PCR analysis 33 3.5. Immunohistochemistry 34 3.6. Limbal epithelial cell culture 38 3.7. Transcription-factor knockdown/overexpression in vitro 39 3.8. Proliferation assay 40 3.9. Western blot 40 3.10. Statistical analysis 41 2 4. RESULTS 4.1. Quality control of LCM-isolated and amplified RNA 42 4.2. -
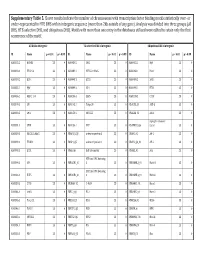
Supplementary Table 5. Clover Results Indicate the Number Of
Supplementary Table 5. Clover results indicate the number of chromosomes with transcription factor binding motifs statistically over‐ or under‐represented in HTE DHS within intergenic sequence (more than 2kb outside of any gene). Analysis was divided into three groups (all DHS, HTE‐selective DHS, and ubiquitous DHS). Motifs with more than one entry in the databases utilized were edited to retain only the first occurrence of the motif. All DHS x Intergenic TEselective DHS x Intergenic Ubiquitous DHS x Intergenic ID Name p < 0.01 p > 0.99 ID Name p < 0.01 p > 0.99 ID Name p < 0.01 p > 0.99 MA0002.2 RUNX1 23 0 MA0080.2 SPI1 23 0 MA0055.1 Myf 23 0 MA0003.1 TFAP2A 23 0 MA0089.1 NFE2L1::MafG 23 0 MA0068.1 Pax4 23 0 MA0039.2 Klf4 23 0 MA0098.1 ETS1 23 0 MA0080.2 SPI1 23 0 MA0055.1 Myf 23 0 MA0099.2 AP1 23 0 MA0098.1 ETS1 23 0 MA0056.1 MZF1_1‐4 23 0 MA0136.1 ELF5 23 0 MA0139.1 CTCF 23 0 MA0079.2 SP1 23 0 MA0145.1 Tcfcp2l1 23 0 V$ALX3_01 ALX‐3 23 0 MA0080.2 SPI1 23 0 MA0150.1 NFE2L2 23 0 V$ALX4_02 Alx‐4 23 0 myocyte enhancer MA0081.1 SPIB 23 0 MA0156.1 FEV 23 0 V$AMEF2_Q6 factor 23 0 MA0089.1 NFE2L1::MafG 23 0 V$AP1FJ_Q2 activator protein 1 23 0 V$AP1_01 AP‐1 23 0 MA0090.1 TEAD1 23 0 V$AP4_Q5 activator protein 4 23 0 V$AP2_Q6_01 AP‐2 23 0 MA0098.1 ETS1 23 0 V$AR_Q6 half‐site matrix 23 0 V$ARX_01 Arx 23 0 BTB and CNC homolog MA0099.2 AP1 23 0 V$BACH1_01 1 23 0 V$BARHL1_01 Barhl‐1 23 0 BTB and CNC homolog MA0136.1 ELF5 23 0 V$BACH2_01 2 23 0 V$BARHL2_01 Barhl2 23 0 MA0139.1 CTCF 23 0 V$CMAF_02 C‐MAF 23 0 V$BARX1_01 Barx1 23 0 MA0144.1 Stat3 23 0 -

SOX4 Expression in Bladder Carcinoma: Clinical Aspects and in Vitro Functional Characterization
Research Article SOX4 Expression in Bladder Carcinoma: Clinical Aspects and In vitro Functional Characterization Mads Aaboe,1 Karin Birkenkamp-Demtroder,1 Carsten Wiuf,1,3 Flemming Brandt Sørensen,4 Thomas Thykjaer,1 Guido Sauter,5 Klaus Møller-Ernst Jensen,2 Lars Dyrskjøt,1 and Torben Ørntoft1 1Molecular Diagnostic Laboratory, Department of Clinical Biochemistry, and 2Department of Urology, Aarhus University Hospital/Skejby Sygehus, Aarhus N, Denmark; 3Bioinformatics Research Center, University of Aarhus; 4Institute of Pathology, Aarhus University Hospital, Aarhus Sygehus, Aarhus C, Denmark; and 5Department of Pathology, University Medical Center Hamburg-Eppendorf, Hamburg, Germany Abstract shown to be involved in a range of developmental processes. The SOX4À/À phenotype in mouse results in death during embryo- The human transcription factor SOX4 was 5-fold up-regulated in bladder tumors compared with normal tissue based on genesis due to lack of normal heart development, which results in whole-genome expression profiling of 166 clinical bladder circulatory failure (6, 7). Furthermore, other studies have shown tumor samples and 27 normal urothelium samples.Using a that SOX4 is expressed in hormone-responsive tissues of the SOX4-specific antibody, we found that the cancer cells reproductive system in mouse, as well as in brain and T- and B-cell expressed the SOX4 protein and, thus, did an evaluation of lymphocytes (8, 9), and may play a role in osteoblastic differenti- SOX4 protein expression in 2,360 bladder tumors using a ation (10). tissue microarray with clinical annotation.We found a SOX4 binds to the 7-bp DNA-motif AACAAAG through its high mobility group domain and thereby transcriptionally activates correlation (P < 0.05) between strong SOX4 expression and target genes (9, 11). -

A High Plane of Nutrition During Early Life Alters the Hypothalamic Transcriptome of Heifer Calves José M
www.nature.com/scientificreports OPEN A high plane of nutrition during early life alters the hypothalamic transcriptome of heifer calves José M. Sánchez 1,2*, Kate Keogh3, Alan K. Kelly1, Colin J. Byrne3, Pat Lonergan1 & David A. Kenny1,3* The aim was to examine the efect of rapid body weight gain during early calfood consistent with earlier sexual development on the transcriptional profle of the hypothalamus. Angus X Holstein– Friesian heifer calves (19 ± 5 days of age) were ofered a high (HI, n = 14) or moderate (MOD, n = 15) plane of nutrition from 3 to 21 weeks of age to achieve a growth rate of 1.2 kg/d and 0.5 kg/d, respectively. Following euthanasia at 21 weeks, the arcuate nucleus (ARC) region was separated from the remainder of the hypothalamus and both were subjected to RNA-Seq. HI calves exhibited altered expression of 80 and 39 transcripts in the ARC and the remaining hypothalamus, respectively (P < 0.05) including downregulation of AGRP and NPY and upregulation of POMC, previously implicated in precocious sexual development. Stress-signaling pathways were amongst the most highly dysregulated. Organ morphology, reproductive system development and function, and developmental disorder were amongst the networks derived from diferentially expressed genes (DEGs) in the ARC. Gene co-expression analysis revealed DEGs within the ARC (POMC, CBLN2, CHGA) and hypothalamus (PENK) as hub genes. In conclusion, enhanced nutrition during early calfood alters the biochemical regulation of the hypothalamus consistent with advanced sexual development in the prepubertal heifer. Early onset of puberty is a key trait underpinning economically efcient cattle production systems.