Principles Governing DNA Methylation During Neuronal Lineage and Subtype Specification
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Synaptic Modulators Nrxn1 and Nrxn3 Are Disregulated in a Disc1 Mouse
Letters to the Editor 585 National University of Ireland Galway, Galway, Ireland; 4Laboratory of Clinical Science, National Institute of Mental Health, National Institutes of Health, Bethesda, MD, USA; 5Center for Integrated Molecular Brain Imaging, Rigshospitalet, University of Copenhagen, Copenhagen, Denmark and 6Department of Psychiatry, Laureate Institute for Brain Research, University of Oklahoma College of Medicine, Tulsa, OK, USA E-mail: [email protected] 7These senior authors contributed equally to this paper. References 1 Ichimiya T, Suhara T, Sudo Y, Okubo Y, Nakayama K, Nankai M et al. Biological Psychiatry 2002; 51: 715–722. 2 Cannon DM, Ichise M, Rollis D, Klaver JM, Gandhi SK, Charney DS et al. Biol Psychiatry 2007; 15: 870–877. 3 Cannon DM, Ichise M, Fromm SJ, Nugent AC, Rollis D, Gandhi SK et al. Biol Psychiatry 2006; 60: 207–217. 4 Purcell S, Neale B, Todd-Brown K, Thomas L, Ferreira MA, Bender D et al. American Journal of Human Genetics 2007; 81: 559–575. Figure 1 Map of t-values from voxel-wise analysis of 5 Shioe K, Ichimiya T, Suhara T, Takano A, Sudo Y, Yasuno F et al. Synapse 2003; 48: 184–188. rs6741892, overlaid on a sample axial magnetic resonance 6 Kalbitzer J, Frokjaer VG, Erritzoe D, Svarer C, Cumming P, imaging slice at the level of the medial thalamus (z = 6 mm). Nielsen FA et al. Neuroimage 2009; 45: 280–285. Bilaterally, T-allele carriers (n = 13) have greater serotonin- 7 Erritzoe D, Holst K, Frokjaer VG, Licht CL, Kalbitzer J, Nielsen FA transporter-binding potential than AA homozygotes (n = 42). -

ADHD) Gene Networks in Children of Both African American and European American Ancestry
G C A T T A C G G C A T genes Article Rare Recurrent Variants in Noncoding Regions Impact Attention-Deficit Hyperactivity Disorder (ADHD) Gene Networks in Children of both African American and European American Ancestry Yichuan Liu 1 , Xiao Chang 1, Hui-Qi Qu 1 , Lifeng Tian 1 , Joseph Glessner 1, Jingchun Qu 1, Dong Li 1, Haijun Qiu 1, Patrick Sleiman 1,2 and Hakon Hakonarson 1,2,3,* 1 Center for Applied Genomics, Children’s Hospital of Philadelphia, Philadelphia, PA 19104, USA; [email protected] (Y.L.); [email protected] (X.C.); [email protected] (H.-Q.Q.); [email protected] (L.T.); [email protected] (J.G.); [email protected] (J.Q.); [email protected] (D.L.); [email protected] (H.Q.); [email protected] (P.S.) 2 Division of Human Genetics, Department of Pediatrics, The Perelman School of Medicine, University of Pennsylvania, Philadelphia, PA 19104, USA 3 Department of Human Genetics, Children’s Hospital of Philadelphia, Philadelphia, PA 19104, USA * Correspondence: [email protected]; Tel.: +1-267-426-0088 Abstract: Attention-deficit hyperactivity disorder (ADHD) is a neurodevelopmental disorder with poorly understood molecular mechanisms that results in significant impairment in children. In this study, we sought to assess the role of rare recurrent variants in non-European populations and outside of coding regions. We generated whole genome sequence (WGS) data on 875 individuals, Citation: Liu, Y.; Chang, X.; Qu, including 205 ADHD cases and 670 non-ADHD controls. The cases included 116 African Americans H.-Q.; Tian, L.; Glessner, J.; Qu, J.; Li, (AA) and 89 European Americans (EA), and the controls included 408 AA and 262 EA. -

Association of Neurexin 3 Polymorphisms with Smoking Behavior
Genes, Brain and Behavior (2012) 11: 704–711 doi: 10.1111/j.1601-183X.2012.00815.x Association of Neurexin 3 polymorphisms with smoking behavior E. Docampo†,M.Ribases´ ‡,§,¶, M. Gratacos` †,E. smoking behavior, and strongly implicate this gene in Bruguera¶, C. Cabezas∗∗,C.Sanchez-Mora´ ‡,G. genetic vulnerability to addictive behaviors. Nieva§,¶, D. Puente††, J. M. Argimon-Pallas` ‡‡, Keywords: Addiction, association, NRXN3, smoking, SNP M. Casas§,¶,§§, R. Rabionet∗,† and X. Estivill† Received 26 Jan 2012, revised 21 Mar 2012 and 22 May 2012, accepted for publication 11 June 2012 †Genes and Disease Program, Centre for Genomic Regulation (CRG) and UPF and Centro de Investigacion´ Biomedica´ en Red en Epidemiología y Salud Publica´ (CIBERESP), ‡Psychiatric Genetics Unit, Vall d’Hebron Research Institute (VHIR), According to the 2011 World Health Organization report on §Biomedical Network Research Centre on Mental Health the global tobacco epidemic, tobacco kills nearly 6 million (CIBERSAM), ¶Department of Psychiatry, Hospital Universitari people each year, causing hundreds of billions of dollars of Vall d’Hebron, **Subdireccio´ General de PromociodelaSalut,´ economic damage worldwide (WHO 2011). In fact, smoking Direccio´ General de Salut Publica,´ Departament de Salut, is the single greatest contributor to preventable ill health and ††Institut Universitari d’InvestigacioenAtenci´ oPrim´ aria` Jordi premature death (reviewed in Bierut 2011). The development Gol, ‡‡Divisio´ d’Avaluacio,´ Servei CataladelaSalut,and` of nicotine addiction is influenced by environmental and §§Department of Psychiatry and Legal Medicine, Universitat genetic factors, and while environmental factors have a Autonoma` de Barcelona, Catalonia, Spain stronger influence on initiation, genetic factors play a more *Corresponding author: R. Rabionet, Genes and Disease Pro- significant role in the transition from regular use to addiction gram, Center for Genomic Regulation (CRG-UPF), C/Dr. -

Transcriptomic Causal Networks Identified Patterns of Differential Gene Regulation in Human Brain from Schizophrenia Cases Versus Controls
Transcriptomic Causal Networks identified patterns of differential gene regulation in human brain from Schizophrenia cases versus controls Akram Yazdani1, Raul Mendez-Giraldez2, Michael R Kosorok3, Panos Roussos1,4,5 1Department of Genetics and Genomic Science, Icahn School of Medicine at Mount Sinai, New York, NY, USA 2Lineberger Comprehensive Cancer Center, School of Medicine, University of North Carolina at Chapel Hill, NC, USA 3Department of Biostatistics, University of North Carolina at Chapel Hill, NC, USA 4Department of Psychiatry and Friedman Brain Institute, Icahn School of Medicine at Mount Sinai, New York, NY 10029, USA 5Mental Illness Research Education and Clinical Center (MIRECC), James J. Peters VA Medical Center, Bronx, New York, 10468, USA Abstract Common and complex traits are the consequence of the interaction and regulation of multiple genes simultaneously, which work in a coordinated way. However, the vast majority of studies focus on the differential expression of one individual gene at a time. Here, we aim to provide insight into the underlying relationships of the genes expressed in the human brain in cases with schizophrenia (SCZ) and controls. We introduced a novel approach to identify differential gene regulatory patterns and identify a set of essential genes in the brain tissue. Our method integrates genetic, transcriptomic, and Hi-C data and generates a transcriptomic-causal network. Employing this approach for analysis of RNA-seq data from CommonMind Consortium, we identified differential regulatory patterns for SCZ cases and control groups to unveil the mechanisms that control the transcription of the genes in the human brain. Our analysis identified modules with a high number of SCZ-associated genes as well as assessing the relationship of the hubs with their down-stream genes in both, cases and controls. -

Cartography of Neurexin Alternative Splicing Mapped by Single
Cartography of neurexin alternative splicing mapped PNAS PLUS by single-molecule long-read mRNA sequencing Barbara Treutleina,b,1, Ozgun Gokcec,1, Stephen R. Quakea,b,d,2, and Thomas C. Südhofc,d,2 Departments of aBioengineering and cMolecular and Cellular Physiology, School of Medicine, bDepartment of Applied Physics, and dHoward Hughes Medical Institute, Stanford University, Stanford, CA 94305 Contributed by Thomas C. Südhof, February 24, 2014 (sent for review January 24, 2014) Neurexins are evolutionarily conserved presynaptic cell-adhesion α-andβ-neurexins contain different extracellular sequences molecules that are essential for normal synapse formation and but identical transmembrane regions and short cytoplasmic tails. synaptic transmission. Indirect evidence has indicated that exten- Specifically, the extracellular sequences of α-neurexins are sive alternative splicing of neurexin mRNAs may produce hundreds composed of six LNS (laminin-α, neurexin, sex hormone-binding if not thousands of neurexin isoforms, but no direct evidence for globulin) domains with three interspersed EGF-like repeats such diversity has been available. Here we use unbiased long-read followed by an O-linked sugar attachment sequence and a con- sequencing of full-length neurexin (Nrxn)1α, Nrxn1β,Nrxn2β, served cysteine loop sequence (8, 15). In contrast, the extracel- Nrxn3α, and Nrxn3β mRNAs to systematically assess how many lular sequences of β-neurexins comprise a short β-neurexin– sites of alternative splicing are used in neurexins with a significant specific sequence, and then splice into the sixth LNS domain of frequency, and whether alternative splicing events at these sites α-neurexins, from which point on β-neurexins are identical to are independent of each other. -

Widespread Sex Differences in Gene Expression and Splicing in the Adult Human Brain
ARTICLE Received 26 Mar 2013 | Accepted 15 Oct 2013 | Published 22 Nov 2013 DOI: 10.1038/ncomms3771 OPEN Widespread sex differences in gene expression and splicing in the adult human brain Daniah Trabzuni1,2,*, Adaikalavan Ramasamy3,*, Sabaena Imran1, Robert Walker4, Colin Smith4, Michael E. Weale3, John Hardy1, Mina Ryten1,3 & North American Brain Expression Consortiumw There is strong evidence to show that men and women differ in terms of neurodevelopment, neurochemistry and susceptibility to neurodegenerative and neuropsychiatric disease. The molecular basis of these differences remains unclear. Progress in this field has been hampered by the lack of genome-wide information on sex differences in gene expression and in particular splicing in the human brain. Here we address this issue by using post-mortem adult human brain and spinal cord samples originating from 137 neuropathologically confirmed control individuals to study whole-genome gene expression and splicing in 12 CNS regions. We show that sex differences in gene expression and splicing are widespread in adult human brain, being detectable in all major brain regions and involving 2.5% of all expressed genes. We give examples of genes where sex-biased expression is both disease-relevant and likely to have functional consequences, and provide evidence suggesting that sex biases in expression may reflect sex-biased gene regulatory structures. 1 Reta Lilla Weston Laboratories, Department of Molecular Neuroscience, UCL Institute of Neurology, Queen Square, London WC1N 3BG, UK. 2 Department of Genetics, King Faisal Specialist Hospital and Research Centre, PO Box 3354, Riyadh 11211, Saudi Arabia. 3 Department of Medical and Molecular Genetics, King’s College London, Guy’s Hospital, 8th Floor, Tower Wing, London SE1 9RT, UK. -

Peripheral Nerve Single-Cell Analysis Identifies Mesenchymal Ligands That Promote Axonal Growth
Research Article: New Research Development Peripheral Nerve Single-Cell Analysis Identifies Mesenchymal Ligands that Promote Axonal Growth Jeremy S. Toma,1 Konstantina Karamboulas,1,ª Matthew J. Carr,1,2,ª Adelaida Kolaj,1,3 Scott A. Yuzwa,1 Neemat Mahmud,1,3 Mekayla A. Storer,1 David R. Kaplan,1,2,4 and Freda D. Miller1,2,3,4 https://doi.org/10.1523/ENEURO.0066-20.2020 1Program in Neurosciences and Mental Health, Hospital for Sick Children, 555 University Avenue, Toronto, Ontario M5G 1X8, Canada, 2Institute of Medical Sciences University of Toronto, Toronto, Ontario M5G 1A8, Canada, 3Department of Physiology, University of Toronto, Toronto, Ontario M5G 1A8, Canada, and 4Department of Molecular Genetics, University of Toronto, Toronto, Ontario M5G 1A8, Canada Abstract Peripheral nerves provide a supportive growth environment for developing and regenerating axons and are es- sential for maintenance and repair of many non-neural tissues. This capacity has largely been ascribed to paracrine factors secreted by nerve-resident Schwann cells. Here, we used single-cell transcriptional profiling to identify ligands made by different injured rodent nerve cell types and have combined this with cell-surface mass spectrometry to computationally model potential paracrine interactions with peripheral neurons. These analyses show that peripheral nerves make many ligands predicted to act on peripheral and CNS neurons, in- cluding known and previously uncharacterized ligands. While Schwann cells are an important ligand source within injured nerves, more than half of the predicted ligands are made by nerve-resident mesenchymal cells, including the endoneurial cells most closely associated with peripheral axons. At least three of these mesen- chymal ligands, ANGPT1, CCL11, and VEGFC, promote growth when locally applied on sympathetic axons. -

Patterns of Sequence Conservation in Presynaptic Neural Genes
University of Pennsylvania ScholarlyCommons Departmental Papers (CIS) Department of Computer & Information Science November 2006 Patterns of Sequence Conservation in Presynaptic Neural Genes Dexter Hadley University of Pennsylvania Tara Murphy University of Pennsylvania Otto Valladares University of Pennsylvania Sridhar Hannenhalli University of Pennsylvania Lyle H. Ungar University of Pennsylvania, [email protected] See next page for additional authors Follow this and additional works at: https://repository.upenn.edu/cis_papers Recommended Citation Dexter Hadley, Tara Murphy, Otto Valladares, Sridhar Hannenhalli, Lyle H. Ungar, Junhyong Kim, and Maja Bucan, "Patterns of Sequence Conservation in Presynaptic Neural Genes", . November 2006. Reprinted from Genome Biology, Volume 7, Issue 11, November 2006, pages R105.1-R105.19. Publisher URL: http://genomebiology.com/2006/7/11/R105 This paper is posted at ScholarlyCommons. https://repository.upenn.edu/cis_papers/282 For more information, please contact [email protected]. Patterns of Sequence Conservation in Presynaptic Neural Genes Abstract Background: The neuronal synapse is a fundamental functional unit in the central nervous system of animals. Because synaptic function is evolutionarily conserved, we reasoned that functional sequences of genes and related genomic elements known to play important roles in neurotransmitter release would also be conserved. Results: Evolutionary rate analysis revealed that presynaptic proteins evolve slowly, although some members of large gene families exhibit accelerated evolutionary rates relative to other family members. Comparative sequence analysis of 46 megabases spanning 150 presynaptic genes identified more than 26,000 elements that are highly conserved in eight vertebrate species, as well as a small subset of sequences (6%) that are shared among unrelated presynaptic genes. -

Downloaded from 4D Nucleome Portal 1346 (
bioRxiv preprint doi: https://doi.org/10.1101/2020.04.02.020990; this version posted May 12, 2021. The copyright holder for this preprint (which was not certified by peer review) is the author/funder. All rights reserved. No reuse allowed without permission. Cell-type specialization in the brain is encoded by specific long-range chromatin topologies Warren Winick-Ng1,#, Alexander Kukalev1,#, Izabela Harabula1,2,#, Luna Zea Redondo1,2,&, Dominik Szabo1,2,&, Mandy Meijer3, Leonid Serebreni1,a, Yingnan Zhang4, Simona Bianco5, Andrea M. Chiariello5, Ibai Irastorza-Azcarate1, Luca Fiorillo5, Francesco Musella5, Christoph J. Thieme1, Ehsan Irani1,6, Elena Torlai Triglia1,b, Aleksandra A. Kolodziejczyk7,8,c, Andreas Abentung9,d, Galina Apostolova9, Eleanor J. Paul10,e, Vedran Franke11, Rieke Kempfer1,2, Altuna Akalin11, Sarah A. Teichmann7,8, Georg Dechant9, Mark A. Ungless10, Mario Nicodemi5,6, Lonnie Welch4, Gonçalo Castelo-Branco3,12, Ana Pombo1,2,6,* 1Epigenetic Regulation and Chromatin Architecture Group, Berlin Institute for Medical Systems Biology, Max-Delbrück Centre for Molecular Medicine, 10115 Berlin, Germany 2Humboldt-Universität zu Berlin, 10117 Berlin, Germany 3Laboratory of Molecular Neurobiology, Department of Medical Biochemistry and Biophysics, Karolinska Institutet, 17177 Stockholm, Sweden 4School of Electrical Engineering and Computer Science, Ohio University, 45701 Athens, OH, USA 5Dipartimentio di Fisica, Università di Napoli Federico II, and INFN Napoli, Complesso Universitario di Monte Sant'Angelo, 80126 Naples, Italy -

Significance of Neurexin and Neuroligin Polymorphisms in Regulating Risk of Hirschsprung’S Disease Yanhong Li, Hui Liu, Yubin Dong
Original research J Investig Med: first published as 10.1136/jim-2017-000623 on 4 April 2018. Downloaded from Significance of neurexin and neuroligin polymorphisms in regulating risk of Hirschsprung’s disease Yanhong Li, Hui Liu, Yubin Dong ► Additional material is ABSTRact published online only. To By performing a basic case–control study among a Significance of this study view please visit the journal Chinese population, the aims of this study were to online (http:// dx. doi. org/ 10. What is already known about this subject? 1136/ jim- 2017- 000623). explore if single nucleotide polymorphisms (SNPs) within neurexin and neuroligin were associated with ► The morbidity of Hirschsprung’s disease Department of Pediatrics, (HD) was ranked high at second of all Zhoukou Central Hospital, susceptibility to Hirschsprung’s disease (HD). Eleven Zhoukou, Henan Province, SNPs within neurexin and neuroligin were selected in disorders relevant to newborn congenital China this basic case–control study, and this study recruited digestive tract malformation. 210 children with HD and 187 healthy children. The ► About 20% of patients with HD were Correspondence to t-test and Χ2 test were used to find the difference accompanied with family heredity. Dr Yanhong Li, Department between case and control in their clinical variables. ► The development of HD may be related of Pediatrics, Zhoukou with migration of neural crest cells in the Central Hospital, Henan OR and 95% CI were used to assess the association Province 466000,China; between HD susceptibility and neurexin/neuroligin embryonic period. xushuai_ yy@ 163. com polymorphisms/haplotypes. Several SNPs were What are the new findings? significantly associated with altered risk of HD in the The C allele of NRXN1 rs1421589 acted as a Accepted 29 January 2018 Chinese Han population, including rs1421589 within ► protective element for HD risk. -
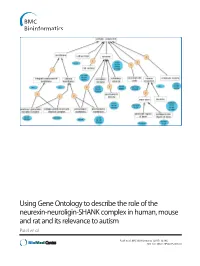
Using Gene Ontology to Describe the Role of the Neurexin-Neuroligin-SHANK Complex in Human, Mouse and Rat and Its Relevance to Autism Patel Et Al
Using Gene Ontology to describe the role of the neurexin-neuroligin-SHANK complex in human, mouse and rat and its relevance to autism Patel et al. Patel et al. BMC Bioinformatics (2015) 16:186 DOI 10.1186/s12859-015-0622-0 Patel et al. BMC Bioinformatics (2015) 16:186 DOI 10.1186/s12859-015-0622-0 METHODOLOGY ARTICLE Open Access Using Gene Ontology to describe the role of the neurexin-neuroligin-SHANK complex in human, mouse and rat and its relevance to autism Sejal Patel1,2,3, Paola Roncaglia4,5 and Ruth C. Lovering3* Abstract Background: People with an autistic spectrum disorder (ASD) display a variety of characteristic behavioral traits, including impaired social interaction, communication difficulties and repetitive behavior. This complex neurodevelopment disorder is known to be associated with a combination of genetic and environmental factors. Neurexins and neuroligins play a key role in synaptogenesis and neurexin-neuroligin adhesion is one of several processes that have been implicated in autism spectrum disorders. Results: In this report we describe the manual annotation of a selection of gene products known to be associated with autism and/or the neurexin-neuroligin-SHANK complex and demonstrate how a focused annotation approach leads to the creation of more descriptive Gene Ontology (GO) terms, as well as an increase in both the number of gene product annotations and their granularity, thus improving the data available in the GO database. Conclusions: The manual annotations we describe will impact on the functional analysis of a variety of future autism-relevant datasets. Comprehensive gene annotation is an essential aspect of genomic and proteomic studies, as the quality of gene annotations incorporated into statistical analysis tools affects the effective interpretation of data obtained throughgenomewideassociationstudies, next generation sequencing, proteomic and transcriptomic datasets. -

Specification of Synaptic Connectivity by Cell Surface Interactions
REVIEWS Specification of synaptic connectivity by cell surface interactions Joris de Wit1 and Anirvan Ghosh2 Abstract | The molecular diversification of cell surface molecules has long been postulated to impart specific surface identities on neuronal cell types. The existence of unique cell surface identities would allow neurons to distinguish one another and connect with their appropriate target cells. Although progress has been made in identifying cell type-specific surface molecule repertoires and in characterizing their extracellular interactions, determining how this molecular diversity contributes to the precise wiring of neural circuitry has proven challenging. Here, we review the role of the cadherin, neurexin, immunoglobulin and leucine-rich repeat protein superfamilies in the specification of connectivity. The emerging evidence suggests that the concerted actions of these proteins may critically contribute to the assembly of neural circuits. Axon initial segment A hallmark of CNS organization is the highly precise pat- The molecular mechanisms that regulate wiring A specialized subcellular tern of connectivity between neurons. The location of a specificity and synaptic diversity in the vertebrate brain compartment in the first part synapse on a target cell and its distinctive structural and are only beginning to be understood. Classic work from of the axon that contains high 2 3 concentrations of channels, functional properties are key factors in determining the Langley and Sperry on the regeneration of nerve fibres scaffolding proteins and flow of information in a neural circuit. in the mature nervous system indicated that neurons adhesion molecules and that The nervous system uses many mechanisms to can rewire with remarkable specificity. Their work sug- initiates action potentials.