Historical Background Prepared by Dr, Robin Chaplin Professor of Power Plant Engineering (Retired) University of New Brunswick
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

1 the Equation of a Light Leptonic Magnetic Monopole and Its
The equation of a Light Leptonic Magnetic Monopole and its Experimental Aspects Georges Lochak Fondation Louis de Broglie 23, rue Marsoulan F-75012 Paris [email protected] Abstract. The present theory is closely related to Dirac’s equation of the electron, but not to his magnetic monopole theory, except for his relation between electric and magnetic charge. The theory is based on the fact, that the massless Dirac equation admits a second electromagnetic coupling, deduced from a pseudo-scalar gauge invariance. The equation thus obtained has the symmetry laws of a massless leptonic, magnetic monopole, able to interact weakly. We give a more precise form of the Dirac relation between electric and magnetic charges and a quantum form of the Poincaré first integral. In the Weyl representation our equation splits into P-conjugated monopole and antimonopole equations with the correct electromagnetic coupling and opposite chiralities, predicted by P. Curie. Charge conjugated monopoles are symmetric in space and not in time (contrary to the electric particles) : an important fact for the vacuum polarization. Our monopole is a magnetically excited neutrino, which leads to experimental consequences. These monopoles are assumed to be produced by electromagnetic pulses or arcs, leading to nuclear transmutations and, for beta radioactive elements, a shortening of the life time and the emission of monopoles instead of neutrinos in a magnetic field. A corresponding discussion is given in section 15. 1. Introduction. The hypothesis of separated magnetic poles is very old. In the 2nd volume of his famous Treatise of Electricity and Magnetism [1], devoted to Magnetism, Maxwell considered the existence of free magnetic charges as an evidence, just as the evidence of electric charges. -

Unerring in Her Scientific Enquiry and Not Afraid of Hard Work, Marie Curie Set a Shining Example for Generations of Scientists
Historical profile Elements of inspiration Unerring in her scientific enquiry and not afraid of hard work, Marie Curie set a shining example for generations of scientists. Bill Griffiths explores the life of a chemical heroine SCIENCE SOURCE / SCIENCE PHOTO LIBRARY LIBRARY PHOTO SCIENCE / SOURCE SCIENCE 42 | Chemistry World | January 2011 www.chemistryworld.org On 10 December 1911, Marie Curie only elements then known to or ammonia, having a water- In short was awarded the Nobel prize exhibit radioactivity. Her samples insoluble carbonate akin to BaCO3 in chemistry for ‘services to the were placed on a condenser plate It is 100 years since and a chloride slightly less soluble advancement of chemistry by the charged to 100 Volts and attached Marie Curie became the than BaCl2 which acted as a carrier discovery of the elements radium to one of Pierre’s electrometers, and first person ever to win for it. This they named radium, and polonium’. She was the first thereby she measured quantitatively two Nobel prizes publishing their results on Boxing female recipient of any Nobel prize their radioactivity. She found the Marie and her husband day 1898;2 French spectroscopist and the first person ever to be minerals pitchblende (UO2) and Pierre pioneered the Eugène-Anatole Demarçay found awarded two (she, Pierre Curie and chalcolite (Cu(UO2)2(PO4)2.12H2O) study of radiactivity a new atomic spectral line from Henri Becquerel had shared the to be more radioactive than pure and discovered two new the element, helping to confirm 1903 physics prize for their work on uranium, so reasoned that they must elements, radium and its status. -

A Century of X-Ray Crystallography and 2014 International Year of X-Ray Crystallography
Macedonian Journal of Chemistry and Chemical Engineering, Vol. 34, No. 1, pp. 19–32 (2015) MJCCA9 – 658 ISSN 1857-5552 Received: January 15, 2015 UDC: 631.416.865(497.7:282) Accepted: February 13, 2015 Review A CENTURY OF X-RAY CRYSTALLOGRAPHY AND 2014 INTERNATIONAL YEAR OF X-RAY CRYSTALLOGRAPHY Biserka Kojić-Prodić Rudjer Bošković Institute, Bijenička c. 54, Zagreb, Croatia [email protected] The 100th anniversary of the Nobel prize awarded to Max von Laue in 1914 for his discovery of diffraction of X-rays on a crystal marked the beginning of a new branch of science - X-ray crystallog- raphy. The experimental evidence of von Laue's discovery was provided by physicists W. Friedrich and P. Knipping in 1912. In the same year, W. L. Bragg described the analogy between X-rays and visible light and formulated the Bragg's law, a fundamental relation that connected the wave nature of X-rays and fine structure of a crystal at atomic level. In 1913 the first simple diffractometer was constructed and structure determination started by the Braggs, father and son. In 1915 their discoveries were acknowl- edged by a Nobel Prize in physics. Since then, X-ray diffraction has been the basic method for determina- tion of three-dimensional structures of synthetic and natural compounds. The three-dimensional structure of a substances defines its physical, chemical, and biological properties. Over the past century the signifi- cance of X-ray crystallography has been recognized by about forty Nobel prizes. X-ray structure analysis of simple crystals of rock salt, diamond and graphite, and later of complex biomolecules such as B12- vitamin, penicillin, haemoglobin/myoglobin, DNA, and biomolecular complexes such as viruses, chroma- tin, ribozyme, and other molecular machines have illustrated the development of the method. -

Luis Alvarez: the Ideas Man
CERN Courier March 2012 Commemoration Luis Alvarez: the ideas man The years from the early 1950s to the late 1980s came alive again during a symposium to commemorate the birth of one of the great scientists and inventors of the 20th century. Luis Alvarez – one of the greatest experimental physicists of the 20th century – combined the interests of a scientist, an inventor, a detective and an explorer. He left his mark on areas that ranged from radar through to cosmic rays, nuclear physics, particle accel- erators, detectors and large-scale data analysis, as well as particles and astrophysics. On 19 November, some 200 people gathered at Berkeley to commemorate the 100th anniversary of his birth. Alumni of the Alvarez group – among them physicists, engineers, programmers and bubble-chamber film scanners – were joined by his collaborators, family, present-day students and admirers, as well as scientists whose professional lineage traces back to him. Hosted by the Lawrence Berkeley National Laboratory (LBNL) and the University of California at Berkeley, the symposium reviewed his long career and lasting legacy. A recurring theme of the symposium was, as one speaker put it, a “Shakespeare-type dilemma”: how could one person have accom- plished all of that in one lifetime? Beyond his own initiatives, Alvarez created a culture around him that inspired others to, as George Smoot put it, “think big,” as well as to “think broadly and then deep” and to take risks. Combined with Alvarez’s strong scientific standards and great care in execut- ing them, these principles led directly to the awarding of two Nobel Luis Alvarez celebrating the announcement of his 1968 Nobel prizes in physics to scientists at Berkeley – George Smoot in 2006 prize. -
![I. I. Rabi Papers [Finding Aid]. Library of Congress. [PDF Rendered Tue Apr](https://docslib.b-cdn.net/cover/8589/i-i-rabi-papers-finding-aid-library-of-congress-pdf-rendered-tue-apr-428589.webp)
I. I. Rabi Papers [Finding Aid]. Library of Congress. [PDF Rendered Tue Apr
I. I. Rabi Papers A Finding Aid to the Collection in the Library of Congress Manuscript Division, Library of Congress Washington, D.C. 1992 Revised 2010 March Contact information: http://hdl.loc.gov/loc.mss/mss.contact Additional search options available at: http://hdl.loc.gov/loc.mss/eadmss.ms998009 LC Online Catalog record: http://lccn.loc.gov/mm89076467 Prepared by Joseph Sullivan with the assistance of Kathleen A. Kelly and John R. Monagle Collection Summary Title: I. I. Rabi Papers Span Dates: 1899-1989 Bulk Dates: (bulk 1945-1968) ID No.: MSS76467 Creator: Rabi, I. I. (Isador Isaac), 1898- Extent: 41,500 items ; 105 cartons plus 1 oversize plus 4 classified ; 42 linear feet Language: Collection material in English Location: Manuscript Division, Library of Congress, Washington, D.C. Summary: Physicist and educator. The collection documents Rabi's research in physics, particularly in the fields of radar and nuclear energy, leading to the development of lasers, atomic clocks, and magnetic resonance imaging (MRI) and to his 1944 Nobel Prize in physics; his work as a consultant to the atomic bomb project at Los Alamos Scientific Laboratory and as an advisor on science policy to the United States government, the United Nations, and the North Atlantic Treaty Organization during and after World War II; and his studies, research, and professorships in physics chiefly at Columbia University and also at Massachusetts Institute of Technology. Selected Search Terms The following terms have been used to index the description of this collection in the Library's online catalog. They are grouped by name of person or organization, by subject or location, and by occupation and listed alphabetically therein. -

Nobel Prizes Social Network
Nobel prizes social network Marie Skłodowska Curie (Phys.1903, Chem.1911) Nobel prizes social network Henri Becquerel (Phys.1903) Pierre Curie (Phys.1903) = Marie Skłodowska Curie (Phys.1903, Chem.1911) Nobel prizes social network Henri Becquerel (Phys.1903) Pierre Curie (Phys.1903) = Marie Skłodowska Curie (Phys.1903, Chem.1911) Irène Joliot-Curie (Chem.1935) Nobel prizes social network Henri Becquerel (Phys.1903) Pierre Curie (Phys.1903) = Marie Skłodowska Curie (Phys.1903, Chem.1911) Irène Joliot-Curie (Chem.1935) = Frédéric Joliot-Curie (Chem.1935) Nobel prizes social network Henri Becquerel (Phys.1903) Pierre Curie (Phys.1903) = Marie Skłodowska Curie (Phys.1903, Chem.1911) Paul Langevin Irène Joliot-Curie (Chem.1935) = Frédéric Joliot-Curie (Chem.1935) Nobel prizes social network Henri Becquerel (Phys.1903) Pierre Curie (Phys.1903) = Marie Skłodowska Curie (Phys.1903, Chem.1911) Paul Langevin Maurice de Broglie Louis de Broglie (Phys.1929) Irène Joliot-Curie (Chem.1935) = Frédéric Joliot-Curie (Chem.1935) Nobel prizes social network Sir J. J. Thomson (Phys.1906) Henri Becquerel (Phys.1903) Pierre Curie (Phys.1903) = Marie Skłodowska Curie (Phys.1903, Chem.1911) Paul Langevin Maurice de Broglie Louis de Broglie (Phys.1929) Irène Joliot-Curie (Chem.1935) = Frédéric Joliot-Curie (Chem.1935) Nobel prizes social network (more) Sir J. J. Thomson (Phys.1906) Nobel prizes social network (more) Sir J. J. Thomson (Phys.1906) Owen Richardson (Phys.1928) Nobel prizes social network (more) Sir J. J. Thomson (Phys.1906) Owen Richardson (Phys.1928) Clinton Davisson (Phys.1937) Nobel prizes social network (more) Sir J. J. Thomson (Phys.1906) Owen Richardson (Phys.1928) Charlotte Richardson = Clinton Davisson (Phys.1937) Nobel prizes social network (more) Sir J. -

Chem 103, Section F0F Unit I
Lecture 4 - Observations that Led to the Chem 103, Section F0F Nuclear Model of the Atom Unit I - An Overview of Chemistry Dalton’s theory proposed that atoms were indivisible particles. Lecture 4 • By the late 19th century, this aspect of Dalton’s theory was being challenged. • Work with electricity lead to the discovery of the electron, • Some observations that led to the nuclear model as a particle that carried a negative charge. for the structure of the atom • The modern view of the atomic structure and the elements • Arranging the elements into a (periodic) table 2 Lecture 4 - Observations that Led to the Lecture 4 - Observations that Led to the Nuclear Model of the Atom Nuclear Model of the Atom The cathode ray In 1897, J.J. Thomson (1856-1940) studies how cathode rays • Cathode rays were shown to be electrons are affected by electric and magnetic fields • This allowed him to determine the mass/charge ration of an electron Cathode rays are released by metals at the cathode 3 4 Lecture 4 - Observations that Led to the Lecture 4 - Observations that Led to the Nuclear Model of the Atom Nuclear Model of the Atom In 1897, J.J. Thomson (1856-1940) studies how cathode rays In 1897, J.J. Thomson (1856-1940) studies how cathode rays are affected by electric and magnetic fields are affected by electric and magnetic fields • Thomson estimated that the mass of an electron was less • Thomson received the 1906 Nobel Prize in Physics for his that 1/1000 the mass of the lightest atom, hydrogen!! work. -

Enrico Fermi
Fermi, Enrico Inventors and Inventions Enrico Fermi Italian American physicist Fermi helped develop Fermi-Dirac statistics, which liceo (secondary school) and, on the advice of Amidei, elucidate the group behavior of elementary particles. joined the Scuola Normale Superiore at Pisa. This elite He also developed the theory of beta decay and college, attached to the University of Pisa, admitted only discovered neutron-induced artificial radioactivity. forty of Italy’s top students, who were given free board Finally, he succeeded in producing the first sustained and lodging. Fermi performed exceedingly well in the nuclear chain reaction, which led to the discovery highly competitive entrance exam. He completed his of nuclear energy and the development of the university education after only four years of research and atomic bomb. studies, receiving his Ph.D. in physics from the Univer- sity of Pisa and his undergraduate diploma from the Born: September 29, 1901; Rome, Italy Scuola Normale Superiore in July, 1922. He became Died: November 28, 1954; Chicago, Illinois an expert theoretical physicist and a talented exper- Primary field: Physics imentalist. This rare combination provided a solid foun- Primary inventions: Controlled nuclear chain dation for all his subsequent inventions. reaction; Fermi-Dirac statistics; theory of beta decay Life’s Work After postdoctoral work at the University of Göttingen, Early Life in Germany (1922-1923), and the University of Leiden, Enrico Fermi (ehn-REE-koh FUR-mee) was the third in the Netherlands (fall, 1924), Fermi took an interim po- child of Alberto Fermi and Ida de Gattis. Enrico was very sition at the University of Florence in December, 1924. -
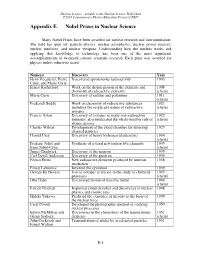
Appendix E Nobel Prizes in Nuclear Science
Nuclear Science—A Guide to the Nuclear Science Wall Chart ©2018 Contemporary Physics Education Project (CPEP) Appendix E Nobel Prizes in Nuclear Science Many Nobel Prizes have been awarded for nuclear research and instrumentation. The field has spun off: particle physics, nuclear astrophysics, nuclear power reactors, nuclear medicine, and nuclear weapons. Understanding how the nucleus works and applying that knowledge to technology has been one of the most significant accomplishments of twentieth century scientific research. Each prize was awarded for physics unless otherwise noted. Name(s) Discovery Year Henri Becquerel, Pierre Discovered spontaneous radioactivity 1903 Curie, and Marie Curie Ernest Rutherford Work on the disintegration of the elements and 1908 chemistry of radioactive elements (chem) Marie Curie Discovery of radium and polonium 1911 (chem) Frederick Soddy Work on chemistry of radioactive substances 1921 including the origin and nature of radioactive (chem) isotopes Francis Aston Discovery of isotopes in many non-radioactive 1922 elements, also enunciated the whole-number rule of (chem) atomic masses Charles Wilson Development of the cloud chamber for detecting 1927 charged particles Harold Urey Discovery of heavy hydrogen (deuterium) 1934 (chem) Frederic Joliot and Synthesis of several new radioactive elements 1935 Irene Joliot-Curie (chem) James Chadwick Discovery of the neutron 1935 Carl David Anderson Discovery of the positron 1936 Enrico Fermi New radioactive elements produced by neutron 1938 irradiation Ernest Lawrence -

ARIE SKLODOWSKA CURIE Opened up the Science of Radioactivity
ARIE SKLODOWSKA CURIE opened up the science of radioactivity. She is best known as the discoverer of the radioactive elements polonium and radium and as the first person to win two Nobel prizes. For scientists and the public, her radium was a key to a basic change in our understanding of matter and energy. Her work not only influenced the development of fundamental science but also ushered in a new era in medical research and treatment. This file contains most of the text of the Web exhibit “Marie Curie and the Science of Radioactivity” at http://www.aip.org/history/curie/contents.htm. You must visit the Web exhibit to explore hyperlinks within the exhibit and to other exhibits. Material in this document is copyright © American Institute of Physics and Naomi Pasachoff and is based on the book Marie Curie and the Science of Radioactivity by Naomi Pasachoff, Oxford University Press, copyright © 1996 by Naomi Pasachoff. Site created 2000, revised May 2005 http://www.aip.org/history/curie/contents.htm Page 1 of 79 Table of Contents Polish Girlhood (1867-1891) 3 Nation and Family 3 The Floating University 6 The Governess 6 The Periodic Table of Elements 10 Dmitri Ivanovich Mendeleev (1834-1907) 10 Elements and Their Properties 10 Classifying the Elements 12 A Student in Paris (1891-1897) 13 Years of Study 13 Love and Marriage 15 Working Wife and Mother 18 Work and Family 20 Pierre Curie (1859-1906) 21 Radioactivity: The Unstable Nucleus and its Uses 23 Uses of Radioactivity 25 Radium and Radioactivity 26 On a New, Strongly Radio-active Substance -

EUGENE PAUL WIGNER November 17, 1902–January 1, 1995
NATIONAL ACADEMY OF SCIENCES E U G ENE PAUL WI G NER 1902—1995 A Biographical Memoir by FR E D E R I C K S E I T Z , E RICH V OG T , A N D AL V I N M. W E I NBER G Any opinions expressed in this memoir are those of the author(s) and do not necessarily reflect the views of the National Academy of Sciences. Biographical Memoir COPYRIGHT 1998 NATIONAL ACADEMIES PRESS WASHINGTON D.C. Courtesy of Atoms for Peace Awards, Inc. EUGENE PAUL WIGNER November 17, 1902–January 1, 1995 BY FREDERICK SEITZ, ERICH VOGT, AND ALVIN M. WEINBERG UGENE WIGNER WAS A towering leader of modern physics Efor more than half of the twentieth century. While his greatest renown was associated with the introduction of sym- metry theory to quantum physics and chemistry, for which he was awarded the Nobel Prize in physics for 1963, his scientific work encompassed an astonishing breadth of sci- ence, perhaps unparalleled during his time. In preparing this memoir, we have the impression we are attempting to record the monumental achievements of half a dozen scientists. There is the Wigner who demonstrated that symmetry principles are of great importance in quan- tum mechanics; who pioneered the application of quantum mechanics in the fields of chemical kinetics and the theory of solids; who was the first nuclear engineer; who formu- lated many of the most basic ideas in nuclear physics and nuclear chemistry; who was the prophet of quantum chaos; who served as a mathematician and philosopher of science; and the Wigner who was the supervisor and mentor of more than forty Ph.D. -

52 Annual Greenville County
nd 52 Annual Greenville County and South Carolina Regional 1A Science and Engineering Fair 2006 Sponsored by Rotary Club of Greenville Roper Mountain Science Center and The South Carolina Academy of Science th th st Judging- March 14 , Public Exhibition – March 15 , Award Ceremony - March 21 Palmetto EXPO Center – Greenville, South Carolina For Immediate Release March 7, 2006 Regional Science & Engineering Fair announces new Charles H. Townes Student Research Award GREENVILLE, SC – The Greenville County and South Carolina Regional Science and Engineering Fair announces a new award recognizing a local high school student for excellence in science research. The Charles H. Townes Student Research Award is the top award in the 52nd Annual Greenville County and South Carolina Regional Science & Engineering Fair to be held at the Palmetto Expo Center. The award honors the career of Greenville’s Charles H. Townes, who received the 1964 Nobel prize in Physics for his research leading to the invention of the laser. To encourage our local future scientists Dr. Townes wrote: “Being a scientist can be a wonderful career. Science involves exploration, figuring things out, understanding our universe and ourselves, and new discoveries. We live in an amazing universe, understanding it is fascinating and inspiring, and also leads to new possibilities for humans. Applied science or engineering uses our scientific understanding to provide new tools, solutions to problems, and remarkable improvements in human life. To do good science, it's helpful to learn all one can, be familiar with a variety of fields, and enjoy exploring. The more we know, the better we can fit ideas together to make new ones.