Fekerte Thesis
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

An Analysis of the Afar-Somali Conflict in Ethiopia and Djibouti
Regional Dynamics of Inter-ethnic Conflicts in the Horn of Africa: An Analysis of the Afar-Somali Conflict in Ethiopia and Djibouti DISSERTATION ZUR ERLANGUNG DER GRADES DES DOKTORS DER PHILOSOPHIE DER UNIVERSTÄT HAMBURG VORGELEGT VON YASIN MOHAMMED YASIN from Assab, Ethiopia HAMBURG 2010 ii Regional Dynamics of Inter-ethnic Conflicts in the Horn of Africa: An Analysis of the Afar-Somali Conflict in Ethiopia and Djibouti by Yasin Mohammed Yasin Submitted in partial fulfilment of the requirements for the degree PHILOSOPHIAE DOCTOR (POLITICAL SCIENCE) in the FACULITY OF BUSINESS, ECONOMICS AND SOCIAL SCIENCES at the UNIVERSITY OF HAMBURG Supervisors Prof. Dr. Cord Jakobeit Prof. Dr. Rainer Tetzlaff HAMBURG 15 December 2010 iii Acknowledgments First and foremost, I would like to thank my doctoral fathers Prof. Dr. Cord Jakobeit and Prof. Dr. Rainer Tetzlaff for their critical comments and kindly encouragement that made it possible for me to complete this PhD project. Particularly, Prof. Jakobeit’s invaluable assistance whenever I needed and his academic follow-up enabled me to carry out the work successfully. I therefore ask Prof. Dr. Cord Jakobeit to accept my sincere thanks. I am also grateful to Prof. Dr. Klaus Mummenhoff and the association, Verein zur Förderung äthiopischer Schüler und Studenten e. V., Osnabruck , for the enthusiastic morale and financial support offered to me in my stay in Hamburg as well as during routine travels between Addis and Hamburg. I also owe much to Dr. Wolbert Smidt for his friendly and academic guidance throughout the research and writing of this dissertation. Special thanks are reserved to the Department of Social Sciences at the University of Hamburg and the German Institute for Global and Area Studies (GIGA) that provided me comfortable environment during my research work in Hamburg. -
Districts of Ethiopia
Region District or Woredas Zone Remarks Afar Region Argobba Special Woreda -- Independent district/woredas Afar Region Afambo Zone 1 (Awsi Rasu) Afar Region Asayita Zone 1 (Awsi Rasu) Afar Region Chifra Zone 1 (Awsi Rasu) Afar Region Dubti Zone 1 (Awsi Rasu) Afar Region Elidar Zone 1 (Awsi Rasu) Afar Region Kori Zone 1 (Awsi Rasu) Afar Region Mille Zone 1 (Awsi Rasu) Afar Region Abala Zone 2 (Kilbet Rasu) Afar Region Afdera Zone 2 (Kilbet Rasu) Afar Region Berhale Zone 2 (Kilbet Rasu) Afar Region Dallol Zone 2 (Kilbet Rasu) Afar Region Erebti Zone 2 (Kilbet Rasu) Afar Region Koneba Zone 2 (Kilbet Rasu) Afar Region Megale Zone 2 (Kilbet Rasu) Afar Region Amibara Zone 3 (Gabi Rasu) Afar Region Awash Fentale Zone 3 (Gabi Rasu) Afar Region Bure Mudaytu Zone 3 (Gabi Rasu) Afar Region Dulecha Zone 3 (Gabi Rasu) Afar Region Gewane Zone 3 (Gabi Rasu) Afar Region Aura Zone 4 (Fantena Rasu) Afar Region Ewa Zone 4 (Fantena Rasu) Afar Region Gulina Zone 4 (Fantena Rasu) Afar Region Teru Zone 4 (Fantena Rasu) Afar Region Yalo Zone 4 (Fantena Rasu) Afar Region Dalifage (formerly known as Artuma) Zone 5 (Hari Rasu) Afar Region Dewe Zone 5 (Hari Rasu) Afar Region Hadele Ele (formerly known as Fursi) Zone 5 (Hari Rasu) Afar Region Simurobi Gele'alo Zone 5 (Hari Rasu) Afar Region Telalak Zone 5 (Hari Rasu) Amhara Region Achefer -- Defunct district/woredas Amhara Region Angolalla Terana Asagirt -- Defunct district/woredas Amhara Region Artuma Fursina Jile -- Defunct district/woredas Amhara Region Banja -- Defunct district/woredas Amhara Region Belessa -- -

Achieving Water Security: Lessons from Research in Water Supply, Sanitation and Hygiene in Ethiopia, Practical Action Publishing, Rugby
Achieving Water Security Praise for the book ‘Water security matters, and it matters most to the most vulnerable individuals, households, communities and nations. Ethiopia is home to many millions of people who are anything but water-secure. This publication shines a light on such people, and it provides numerous inter-disciplinary insights into the causes of water insecurity and into some of the measures which are needed to relieve that insecurity. There are too few research programmes of this type which focus on situations of real need in the world, and which grapple with the difficult issues associated with the relief of poverty and vulnerability. I warmly welcome this contribution to the literature, and hope that this work will inform the practices and policies of government and development part- ners in Ethiopia.’ Richard C. Carter, Visiting Professor, Cranfield University, UK ‘Consolidating these findings from the RiPPLE programme into a single volume to address water security in Ethiopia has generated an essential primer and benchmark in realizing domestic water supply and sanitation in Africa’s second most populous country. Being informed by five years of research and learning, this is a valuable insight into a critical dimension of Ethiopia’s future security.’ Melvin Woodhouse, Associate Director, LTS International, UK Achieving Water Security Lessons from research in water supply, sanitation and hygiene in Ethiopia Edited by Roger Calow, Eva Ludi and Josephine Tucker Practical Action Publishing Ltd The Schumacher Centre Bourton on Dunsmore, Rugby, Warwickshire CV23 9QZ, UK www.practicalactionpublishing.org Copyright © Overseas Development Institute, 2013 ISBN 978 1 85339 7639 Hardback ISBN 978 1 85339 7646 Paperback ISBN 978 1 78044 763 6 Library Ebook ISBN 978 1 78044 764 3 Ebook All rights reserved. -

Somali Region
Federalism and ethnic conflict in Ethiopia. A comparative study of the Somali and Benishangul-Gumuz regions Adegehe, A.K. Citation Adegehe, A. K. (2009, June 11). Federalism and ethnic conflict in Ethiopia. A comparative study of the Somali and Benishangul-Gumuz regions. Retrieved from https://hdl.handle.net/1887/13839 Version: Not Applicable (or Unknown) Licence agreement concerning inclusion of doctoral thesis in the License: Institutional Repository of the University of Leiden Downloaded from: https://hdl.handle.net/1887/13839 Note: To cite this publication please use the final published version (if applicable). 8 Inter-regional Conflicts: Somali Region 8.1 Introduction The previous chapter examined intra-regional conflicts within the Benishangul-Gumuz region. This and the next chapter (chapter 9) deal with inter-regional conflicts between the study regions and their neighbours. The federal restructuring carried out by dismantling the old unitary structure of the country led to territorial and boundary disputes. Unlike the older federations created by the union of independent units, which among other things have stable boundaries, creating a federation through federal restructuring leads to controversies and in some cases to violent conflicts. In the Ethiopian case, violent conflicts accompany the process of intra-federal boundary making. Inter-regional boundaries that divide the Somali region from its neighbours (Oromia and Afar) are ill defined and there are violent conflicts along these borders. In some cases, resource conflicts involving Somali, Afar and Oromo clans transformed into more protracted boundary and territorial conflicts. As will be discussed in this chapter, inter-regional boundary making also led to the re-examination of ethnic identity. -

Hum Ethio Manitar Opia Rian Re Espons E Fund D
Hum anitarian Response Fund Ethiopia OCHA, 2011 OCHA, 2011 Annual Report 2011 Office for the Coordination of Humanitarian Affairs Humanitarian Response Fund – Ethiopia Annual Report 2011 Table of Contents Note from the Humanitarian Coordinator ................................................................................................ 2 Acknowledgements ................................................................................................................................. 3 1. Executive Summary ............................................................................................................................ 4 1.1 2011 Humanitarian Context ........................................................................................................... 4 1.2 Map - 2011 HRF Supported Projects ............................................................................................. 6 2. Information on Contributors ................................................................................................................ 7 2.1 Donor Contributions to HRF .......................................................................................................... 7 3. Fund Overview .................................................................................................................................... 8 3.1 Summary of HRF Allocations in 2011 ............................................................................................ 8 3.1.1 HRF Allocation by Sector ....................................................................................................... -

Irregular Migration from Ethiopia to the Gulf States
MIGRATION POLICY CENTRE Irregular Migration from Ethiopia to the Gulf States Bina Fernandez Chapter in the volume: Skilful Survivals: Irregular Migration to the Gulf edited by Philippe Fargues and Nasra M. Shah BOOK CHAPTER This is a chapter in the volume: Philippe Fargues and Nasra M. Shah (eds.),Skilful Survivals: Irregular Migration to the Gulf, Gulf Labour Markets and Migration (GLMM) Programme, Gulf Research Center Cambridge, 2017. For other chapters and the entire volume, please refer to www.gulfmigration.eu. Terms of use: By using any information from Gulf Labour Markets and Migration programme publications, the user: (a) acknowledges having read the legal notices concerning copyright, terms of use and disclaimers and agrees to comply with them in full; (b) agrees to assure himself/herself whether and to what extent copyrights exist on information published by the GLMM prior to using this information; (c) agrees not to use information published by GLMM in any way that would result in the infringement of existing copyrights; (d) agrees to always follow precisely and fully the citation instructions provided by the GLMM. GLMM publications may be copied and distributed only in their entirety and together with any copyright statements they may contain, as long as they are properly attributed and used for non-commercial, educational, or public policy purposes. Photographs, logos, graphs, tables or any other images from the GLMM publications may not be used separately. Copyright: © European University Institute (EUI) and Gulf Research Center (GRC), 2017. All rights reserved. No part of this publication may be reproduced, stored in a retrieval system, or transmitted in any form or by any means, electronic, mechanical, photocopying, recording or otherwise, without the prior permission of European University Institute and Gulf Research Center. -

Dire Dawa Millennium Park
IOM SITE MANAGEMENT SITE PROFILE - ETHIOPIA, DIRE DAWA SUPPORT Dire Dawa Millennium Park (January 2020) Publication: 01 February 2020 ETHIOPIA IOM OIM Context The residents of Millennium Park IDP site are Somali IDPs who fled their areas of origin in Oromia. The site, which is located on Current Priority Needs land slated for development by the Dire Dawa Municipality, with World Bank funding, is included in the Somali Regional Govern- Site Overview Site Population ment's durable solutions planning, though the timeframe for potential relocations to Somali Region is not yet known. In the interim, many basic humanitarian needs of the site residents are unmet, including safe water provision and sufficient latrine coverage. Fo- 1 WASH Site Location 1,638 individuals od distribution was very irregular in the second half of 2019, but distribution did take place in Jan 2020 2 Livelihood Latitude: 9.6056860 306 households Longitude:41.859980 2 3 Health Site Area: 30,000m Data source: DTM Data, SA 20 of November 2019 Methodology Established in: Sep. 18 2017 * Priority needs as reported by residents through SMS-run Complaint The information for this site profile was collected through key informant interviews and group discussions with the Site Management and Feedback Mechanism Committees and beneficiaries by the Site Management Support (SMS) team in the area. Population figures are collected and aligned through SMS and DTM and allow for the most accurate analysis possible. Sector Overview Demographics Vulnerabilities 0 Unaccompanied children Implementing -

CORE Group Polio Project FY20 Annual Report
POLIO ● core group polio project annual report fy20 Project Responding to Disruption with Resilience and Adaptation 1 core group polio project fy20 Contents Executive Summary .................................................................................................................... 3 Acknowledgements .................................................................................................................... 4 Objectives .................................................................................................................................. 4 Acronyms .................................................................................................................................. 5 Objective 1. Build effective partnerships with PVOs, NGO(s), and international, national and regional agencies involved in polio eradication ........................................................................................... 6 Children in Uganda receive Objective 2. Support PVO/NGO efforts to strengthen national and regional immunization systems to vaccines for oral polio vaccine and achieve polio eradication ............................................................................................................. 9 measles during a CGPP-supported Objective 3. Support PVO/NGO involvement in national and regional planning and implementation of SIA held in October 2020. supplemental polio immunization. ................................................................................................ 14 Photo by CGPP Uganda. Objective -

Implication of Ethiopian Federalism on the Right to Freedom of Movement and Residence: Critical Analysis of the Law and the Practice
Implication of Ethiopian Federalism on the Right to Freedom of Movement and Residence: Critical Analysis of the Law and the Practice By Yonas Girma Adimassu A Thesis Submitted to Addis Ababa University College of Law and Governance Presented in Partial Fulfillment of the Requirements for Masters of Constitutional and Public Law Addis Ababa University, Addis Ababa Ethiopia March 2013 1 DECLARATION I, Yonas Girma, hereby declare that this dissertation is original and has never been presented in any other institution. To the best of my knowledge and belief, I also declare that any information used has been duly acknowledged. LL.M Candidate Name: Yonas Girma Signature: ___________________ February 2013. This dissertation has been submitted for examination with my approval as University supervisor. Supervisor: Ass. Prof Abera Degefa Signature: _____________________ Date: March 2013. 2 Acknowledgment First and for most I would like to thank the almighty God and his mother Saint Virgin Mary for all what happened to me. I would like to express my deepest gratitude to my advisor Mr. Abera Degefa for his speedy and diligence constructive comments and suggestions on this thesis. I owe you sir! I have to confess that I would not have come this far in the absence of your support! I am also grateful to all personalities who have been voluntarily reacting for my interviews and for their valuable suggestions which made the paper more comprehensive. My gratitude also extends to all the offices and their respected staffs for their dulcet contributions by providing the necessary information and materials which I was in need of. -
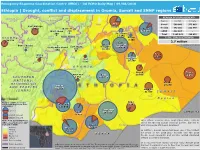
Drought, Conflict and Displacement in Oromia, Somali and SNNP Regions
Emergency Response Coordination Centre (ERCC) – DG ECHO Daily Map | 09/08/2018 Ethiopia | Drought, conflict and displacement in Oromia, Somali and SNNP regions REASON OF DISPLACEMENT* BENSHANGUL- GUMAZ AMAHARA Region Conflict Climate AFAR Siti Somali 500 003 373 663 East Wallega 78 687 Oromia 643 201 112 949 27 672 North Sheva Fafan West Sheva 220 113 593 SNNP 822 187 - 309 Dire Dawa Total 1 965 391 486 612 11 950 Kelem Wellega Harari TOTAL IDPs In ETHIOPIA 24 338 3 605 East 2.7 million West Bunno Bedele Hararghe *Excluding other causes East Sheva Hararghe 2 172 South West Sheva 169 860 180 678 40 030 Jimma Jarar 5 448 49 007 Arsi Erer zone SOMALIA 4 185 36 308 Source: Fews Net OROMIA Nogob Doolo West Arsi Bale 25 659 89 911 S O U T H E R N 4 454 121 772 NATIONS, Korahe NATIONALITIES 24 295 Gedeo A N D PEOPLES 822 187 (SNNP) Shabelle SOUTH 41 536 S O M A L I SUDAN R e g i o n Number of IDPs in Oromia Afder and Somali Region per Zone West Guji 55 963 (July 2018) 147 040 20 000 Dawa zone Guji 244 638 SOMALIA Conflict induced 51 682 Climate induced Source: Fews Net, IOM Acute Food Insecurity Phase Inter-ethnic violence since September 2017, namely DTM Round 11 | Source (IPC’s classification for July– along the Oromia-Somali regional border, has led to for Gedeo and West Guji: September 2018) Liben 500 000 people still being displaced. IOM Situation Report N.1 No Data 114 069 Borena 4-18 July 2018 Minimal 156 110 In addition, Somali region has been one of the hardest Stressed hit areas of the 2016-2017 drought and the 2018 Crisis KENYA floods. -

Somali Region
Food Supply Prospects FOR THE SECOND HALF OF YEAR 2013 ______________________________________________________________________________ Disaster Risk Management and Food Security Sector (DRMFSS) Ministry of Agriculture (MoA) September, 2013 Addis Ababa, Ethiopia TABLE OF CONTENTS GLOSSARY OF LOCAL NAMES .................................................................. 1 ACRONYMS ............................................................................................. 2 EXCUTIVE SUMMARY .............................................................................. 3 INTRODUCTION ....................................................................................... 7 REGIONAL SUMMARY OF FOOD SUPPLY PROSPECT ............................. 11 SOMALI .............................................................................................. 11 OROMIA ............................................................................................. 16 TIGRAY ............................................................................................... 22 AMHARA ............................................................................................ 25 AFAR .................................................................................................. 28 SNNP .................................................................................................. 32 Annex – 1: NEEDY POPULATION AND FOOD REQUIREMENT BY WOREDA (Second half of 2013) ............................................................................ 35 0 | P a g e GLOSSARY -
D.Table 9.5-1 Number of PCO Planned 1
D.Table 9.5-1 Number of PCO Planned 1. Tigrey No. Woredas Phase 1 Phase 2 Phase 3 Expected Connecting Point 1 Adwa 13 Per Filed Survey by ETC 2(*) Hawzen 12 3(*) Wukro 7 Per Feasibility Study 4(*) Samre 13 Per Filed Survey by ETC 5 Alamata 10 Total 55 1 Tahtay Adiyabo 8 2 Medebay Zana 10 3 Laelay Mayechew 10 4 Kola Temben 11 5 Abergele 7 Per Filed Survey by ETC 6 Ganta Afeshum 15 7 Atsbi Wenberta 9 8 Enderta 14 9(*) Hintalo Wajirat 16 10 Ofla 15 Total 115 1 Kafta Humer 5 2 Laelay Adiyabo 8 3 Tahtay Koraro 8 4 Asegede Tsimbela 10 5 Tselemti 7 6(**) Welkait 7 7(**) Tsegede 6 8 Mereb Lehe 10 9(*) Enticho 21 10(**) Werie Lehe 16 Per Filed Survey by ETC 11 Tahtay Maychew 8 12(*)(**) Naeder Adet 9 13 Degua temben 9 14 Gulomahda 11 15 Erob 10 16 Saesi Tsaedaemba 14 17 Alage 13 18 Endmehoni 9 19(**) Rayaazebo 12 20 Ahferom 15 Total 208 1/14 Tigrey D.Table 9.5-1 Number of PCO Planned 2. Affar No. Woredas Phase 1 Phase 2 Phase 3 Expected Connecting Point 1 Ayisaita 3 2 Dubti 5 Per Filed Survey by ETC 3 Chifra 2 Total 10 1(*) Mile 1 2(*) Elidar 1 3 Koneba 4 4 Berahle 4 Per Filed Survey by ETC 5 Amibara 5 6 Gewane 1 7 Ewa 1 8 Dewele 1 Total 18 1 Ere Bti 1 2 Abala 2 3 Megale 1 4 Dalul 4 5 Afdera 1 6 Awash Fentale 3 7 Dulecha 1 8 Bure Mudaytu 1 Per Filed Survey by ETC 9 Arboba Special Woreda 1 10 Aura 1 11 Teru 1 12 Yalo 1 13 Gulina 1 14 Telalak 1 15 Simurobi 1 Total 21 2/14 Affar D.Table 9.5-1 Number of PCO Planned 3.