RET Signaling and Ureteric Bud Development 1377
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Structure of Pronephros and Development of Mesonephric Kidney in Larvae of Russian Sturgeon, Acipenser Gueldenstaedtii Brandt (Acipenseridae)
Zoologica5 PRONEPHROS Poloniae-AND (2012)-MESONEPHRIC 57/1-4: 5-20-KIDNEY-IN-LARVAE-OF-A.-GUELDENSTAEDTII 5 DOI: 10.2478/v10049-012-0001-6 STRUCTURE OF PRONEPHROS AND DEVELOPMENT OF MESONEPHRIC KIDNEY IN LARVAE OF RUSSIAN STURGEON, ACIPENSER GUELDENSTAEDTII BRANDT (ACIPENSERIDAE) L.S. KRAYUSHKINA*1, A.A. GERASIMOV1, A.A. KIRSANOV1, M.V. MOSYAGINA1, A. OGORZA£EK2 1Department of Ichthyology and Hydrobiology, St. Petersburg State University, 16-th Line 29, 199178, St. Petersburg, Russia, [email protected] 2 Department of Animal Developmental Biology, Zoological Institute, University of Wroclaw, Sienkiewicza 21, 50-335 Wroclaw, Poland. *Corresponding author Abstract. The structure of the pronephros and development of mesonephric kidney in Russian sturgeon larvae, Acipenser gueldenstaedtii Brandt at different stages of early postembryonic development (from hatching to 14 days), were studied with histological and electronic microscopy methods. The larval pronephros is represented by the system of bilaterally located pronephric tubules with ciliated nephrostomes and funnels and exog- enous single glomus, which is not integrated directly into pronephric tubules and located in the pronephric chamber. The glomus is positioned below the dorsal aorta and vascular- ized by its capillaries. The glomus has the same features of the thin structure that are typical of and necessary for the function of a filtering organ. The structure of the prone- phros in acipenserids is discussed and compared with teleosts and amphibians. Histogen- esis of the mesonephric kidney is observed during the period of pronephros functioning; it is complete by the time the larvae transfer to exogenous feeding. At this moment, the pronephros undergoes significant structural degradation. -

Stem Cells in the Embryonic Kidney R Nishinakamura1
View metadata, citation and similar papers at core.ac.uk brought to you by CORE provided by Elsevier - Publisher Connector http://www.kidney-international.org mini review & 2008 International Society of Nephrology Stem cells in the embryonic kidney R Nishinakamura1 1Division of Integrative Cell Biology, Institute of Molecular Embryology and Genetics, Kumamoto University, 2-2-1 Honjo, Kumamoto, Japan The mammalian kidney, the metanephros, is formed by a STRATEGY TOWARD KIDNEY RECONSTITUTION USING reciprocally inductive interaction between two precursor PROGENITOR CELLS tissues, the metanephric mesenchyme and the ureteric bud. Stem cells are defined by two criteria: self-renewal and The ureteric bud induces the metanephric mesenchyme to multipotency. Few reports in the kidney field have addressed differentiate into the epithelia of glomeruli and renal tubules. both of these criteria at a clonal level, so it is better to use the Multipotent renal progenitors that form colonies upon Wnt4 term ‘progenitor’ rather than ‘stem cells.’ In this review, renal stimulation and strongly express Sall1 exist in the progenitors in the embryonic kidney, not those in the adult metanephric mesenchyme; these cells can partially kidney, from the viewpoint of developmental biology and reconstitute a three-dimensional structure in an organ stem/progenitor cell biology will be discussed. To generate culture setting. Six2 maintains this mesenchymal progenitor multiple cell lineages for kidney regeneration, the identifica- population by opposing Wnt4-mediated epithelialization. tion of renal progenitors is a prerequisite. Furthermore, there Upon epithelial tube formation, Notch2 is required for the exist three obstacles to be overcome: (1) derivation of the differentiation of proximal nephron structures (podocyte and renal progenitors; (2) expansion of the renal progenitors; and proximal tubules). -

Secreted Molecules in Metanephric Induction
J Am Soc Nephrol 11: S116–S119, 2000 Secreted Molecules in Metanephric Induction THOMAS J. CARROLL and ANDREW P. McMAHON Department of Molecular and Cellular Biology, Biological Laboratories, Harvard University, Cambridge, Massachusetts. Abstract. Nearly 50 yr ago, Clifford Grobstein made the ob- the classic model of metanephric induction. The studies of the servation that the ureteric bud induced the nephrogenic mes- classic ureteric inducer performed to date have most likely enchyme to undergo tubulogenesis. Since that discovery, sci- been characterizations of a mesenchyme-specific inducer, entists have attempted to characterize the molecular nature of Wnt-4, and its role in tubulogenesis. Ureteric induction most the inducer. To date, no single molecule that is both necessary likely involves a series of distinct events that provide prolif- and sufficient for nephric induction has been identified. Be- erative, survival, and condensation signals to the mesenchyme, cause of recent insights regarding the role of several secreted integrating the growth of the ureteric system with tubulogen- molecules in tubulogenesis, it has become necessary to revise esis. The developmental biologic processes of the kidney have been logenesis. The conclusion drawn from this discovery was that the subject of intense study for more than 100 yr (for review, the ureteric bud induces tubulogenesis within the surrounding see reference (1). All three vertebrate kidney types (pro- mesenchyme. During further investigation, it was discovered nephros, mesonephros, and metanephros) are derivatives of a that a number of tissues, including, most notably, a dorsal region of the embryo known as the intermediate mesoderm. In portion of the embryonic spinal cord, are able to substitute for mice, a portion of the mesonephric duct, known as the meta- the ureter in this inductive interaction. -

Urinary System Intermediate Mesoderm
Urinary System Intermediate mesoderm lateral mesoderm: somite ectoderm neural NOTE: Intermediate mesoderm splanchnic groove somatic is situated between somites and lateral mesoderm (somatic and splanchnic mesoderm bordering the coelom). All mesoderm is derived from the primary mesen- intermediate mesoderm endoderm chyme that migrated through the notochord coelom (becomes urogenital ridge) primitive streak. Intermediate mesoderm (plus adjacent mesothelium lining the coelom) forms a urogenital ridge, which consists of a laterally-positioned nephrogenic cord (that forms kidneys & ureter) and a medially-positioned gonadal ridge (for ovary/testis & female/male genital tract formation). Thus urinary & genital systems have a common embryonic origin; also, they share common ducts. NOTE: Urine production essentially requires an increased capillary surface area (glomeruli), epithelial tubules to collect plasma filtrate and extract desirable constituents, and a duct system to convey urine away from the body. Kidneys Bilateraly, three kid- mesonephric duct neys develop from the neph- metanephros pronephros rogenic cord. They develop mesonephric tubules chronologically in cranial- mesonephros caudal sequence, and are designated pro—, meso—, Nephrogenic Cord (left) and meta—, respectively. cloaca The pronephros and mesonephros develop similarly: the nephrogenic cord undergoes seg- mentation, segments become tubules, tubules drain into a duct & eventually tubules disintegrate. spinal ganglion 1] Pronephros—consists of (7-8) primitive tubules and a pronephric duct that grows caudally and terminates in the cloaca. The tubules soon degenerate, but the pronephric duct persists as the neural tube mesonephric duct. (The pronephros is not functional, somite except in sheep.) notochord mesonephric NOTE tubule The mesonephros is the functional kidney for fish and am- aorta phibians. The metanephros is the functional kidney body of reptiles, birds, & mammals. -

Embryology of the Kidney Rizaldy Paz Scott | Yoshiro Maezawa | Jordan Kreidberg | Susan E
1 Embryology of the Kidney Rizaldy Paz Scott | Yoshiro Maezawa | Jordan Kreidberg | Susan E. Quaggin CHAPTER OUTLINE MAMMALIAN KIDNEY DEVELOPMENT, 2 MOLECULAR GENETICS OF MODEL SYSTEMS TO STUDY KIDNEY NEPHROGENESIS, 22 DEVELOPMENT, 8 GENETIC ANALYSIS OF MAMMALIAN KIDNEY DEVELOPMENT, 15 KEY POINTS • The development of the kidney relies on reciprocal signaling and inductive interactions between neighboring cells. • Epithelial cells that comprise the tubular structures of the kidney are derived from two distinct cell lineages: the ureteric epithelia lineage that branches and gives rise to collecting ducts and the nephrogenic mesenchyme lineage that undergoes mesenchyme to epithelial transition to form connecting tubules, distal tubules, the loop of Henle, proximal tubules, parietal epithelial cells, and podocytes. • Nephrogenesis and nephron endowment requires an epigenetically regulated balance between nephron progenitor self-renewal and epithelial differentiation. • The timing of incorporation of nephron progenitor cells into nascent nephrons predicts their positional identity within the highly patterned mature nephron. • Stromal cells and their derivatives coregulate ureteric branching morphogenesis, nephrogenesis, and vascular development. • Endothelial cells track the development of the ureteric epithelia and establish the renal vasculature through a combination of vasculogenic and angiogenic processes. • Collecting duct epithelia have an inherent plasticity enabling them to switch between principal and intercalated cell identities. MAMMALIAN KIDNEY DEVELOPMENT The filtration function of the kidneys is accomplished by basic units called nephrons (Fig. 1.1). Humans on average have 1 million nephrons per adult kidney but the range of ANATOMIC OVERVIEW OF THE 4 MAMMALIAN KIDNEY total nephrons is highly variable across human populations. Each mouse kidney may contain up to 12,000–16,000 nephrons The kidney is a sophisticated, highly vascularized organ that depending on the strain.5 This wide range in nephron number plays a central role in overall body homeostasis. -
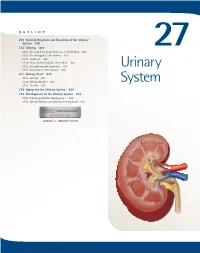
Urinary System
OUTLINE 27.1 General Structure and Functions of the Urinary System 818 27.2 Kidneys 820 27 27.2a Gross and Sectional Anatomy of the Kidney 820 27.2b Blood Supply to the Kidney 821 27.2c Nephrons 824 27.2d How Tubular Fluid Becomes Urine 828 27.2e Juxtaglomerular Apparatus 828 Urinary 27.2f Innervation of the Kidney 828 27.3 Urinary Tract 829 27.3a Ureters 829 27.3b Urinary Bladder 830 System 27.3c Urethra 833 27.4 Aging and the Urinary System 834 27.5 Development of the Urinary System 835 27.5a Kidney and Ureter Development 835 27.5b Urinary Bladder and Urethra Development 835 MODULE 13: URINARY SYSTEM mck78097_ch27_817-841.indd 817 2/25/11 2:24 PM 818 Chapter Twenty-Seven Urinary System n the course of carrying out their specific functions, the cells Besides removing waste products from the bloodstream, the uri- I of all body systems produce waste products, and these waste nary system performs many other functions, including the following: products end up in the bloodstream. In this case, the bloodstream is ■ Storage of urine. Urine is produced continuously, but analogous to a river that supplies drinking water to a nearby town. it would be quite inconvenient if we were constantly The river water may become polluted with sediment, animal waste, excreting urine. The urinary bladder is an expandable, and motorboat fuel—but the town has a water treatment plant that muscular sac that can store as much as 1 liter of urine. removes these waste products and makes the water safe to drink. -

Renal Development
RENAL DEVELOPMENT Jon Barasch M.D., Ph.D. Telephone: 305-1890 e-mail: [email protected] SUGGESTED READING: Larsen, 3rd edition, pp 265 (first three paragraphs) - 266, 268-276 and figure 10-10 LEARNING OBJECTIVES: You should be able to: 1. Describe the three kidneys that are produced during development and know what happens to each one. 2. Explain what is meant by ‘reciprocal induction’ and why it poses problems in interpreting experiments in developing kidney. 3. Describe the stages of nephron formation from the renal vesicle. 4. Discuss the regulators of mesenchymal to epithelial transition in the intermediate mesoderm and metanephric mesenchyme and name three molecules mediating conversion. 5. Describe branching morphogenesis and name the three patterns in the developing metanephros. 6. Discuss three key important ligands and their receptors. 7. Discuss the classification of congenital renal abnormalities that are associated with urological abnormalities and the possible underlying mechanisms for their association. SUMMARY: The urogenital system derives from mesenchymal cells by a process of conversion to epithelia. The development of the kidney relies on three mechanisms of epithelial morphogenesis. 1. Some newborn epithelia migrate extensively (Wolffian duct), 2. some undergo branching morphogenesis (ureteric bud) and 3. some produce highly segmented tubules (nephrons). GLOSSARY: Angiotensin II: your favorite vasoconstrictor and regulator of proximal tubule reclamation of NaCl and water by receptor type 1. Receptor type-2 modulates cell growth and seems to play a role in congenital abnormalities. Arcade: a tubule of ureteric bud that induces a few nephrons simultaneously. The nephrons join to a common drainage called a connecting tubule that feeds into the ureteric’s collecting duct. -

Imaging the Developing Human External and Internal Urogenital Organs with Light Sheet fluorescence Microscopy T
Differentiation 111 (2020) 12–21 Contents lists available at ScienceDirect Differentiation journal homepage: www.elsevier.com/locate/diff Imaging the developing human external and internal urogenital organs with light sheet fluorescence microscopy T ∗ Dylan Isaacsona, , Dylan McCreedyb, Meredith Calvertc, Joel Shend, Adriane Sinclaire, Mei Caoe, Yi Lie, Todd McDevittf,g, Gerald Cunhae, Laurence Baskine,h a Department of Urology, Northwestern University Feinberg School of Medicine, Chicago, IL, USA b Department of Biology, Texas A&M University, College Station, TX, USA c Histology and Light Microscopy Core, J. David Gladstone Institutes, San Francisco, CA, USA d CytomX Therapeutics, Inc. South San Francisco, CA, USA e Department of Urology, University of California, San Francisco, San Francisco, CA, USA f Department of Bioengineering and Therapeutic Sciences, J. David Gladstone Institutes, San Francisco, CA, USA g Institute of Cardiovascular Disease, J. David Gladstone Institutes, San Francisco, CA, USA h Division of Pediatric Urology, University of California San Francisco Benioff Children's Hospital, San Francisco, CA, USA ARTICLE INFO ABSTRACT Keywords: Technological advances in three-dimensional (3D) reconstruction techniques have previously enabled paradigm Microscopy shifts in our understanding of human embryonic and fetal development. Light sheet fluorescence microscopy Light sheet fluorescence microscopy (LSFM) is a recently-developed technique that uses thin planes of light to optically section whole-mount cleared Selective plane illumination microscopy and immunolabeled biologic specimens. The advent of commercially-available light sheet microscopes has fa- Genital development cilitated a new generation of research into protein localization and tissue dynamics at extremely high resolution. Urogenital development Our group has applied LSFM to study developing human fetal external genitalia, internal genitalia and kidneys. -

Kidney Development and Function in the Fetus
Kidney Development, - and Function in the Fetus Bob Caruthers, CST, PhD The kidneys produce urine, a blood filtrate, and regulate uri- can be seen in the fourth week. The pronephros is complete1 nary volume and composition. These regulatory activities regred by the start of the fifth week. The pronephros form involve balancing water and solute transport, conserving and regresses in a cranial-to-caudal sequence. No pronephric nutrients, eliminating waste products, and regulating acid and glomeruli (cluster of capillaries) have been observed, and no bases. The primary purpose of kidney function is to maintain a vesicles are associated with the pronephric duct. The stable environment in which cellular and tissue metabolic pronephros is, therefore, not active in urine producti0n.Y activity can proceed at an optimal level. The kidneys secrete the hormone renin, erythropoietin and 1.25-dihydroxy vita- MPSONEPHROS min D. Renin helps regulate blood pressure. Erythropoietin The mesonephros originates in the nephrogenic cord that is helps regulate erythrocyte production. 1,25-dihydroxy vitamin part of the intermediate mesoderm. Early mesonephric forma- D plays a role in calcium metabolism. This article will discuss tion is evident before the pronephros has completed its regre: the development of the kidney, and its role in the fetus."' sion. The mesonephros also degenerates in a cranial-to-cauda Since the kidneys are bilateral structures, development sequence. Some of the cranial structures are degenerating in involves both right and left kidneys. During fetal develop- the fifth week of fetal development while the caudal structun ment, three separate nephric structures develop in succession; are still differentiating. -

Conserved and Divergent Features of Human and Mouse Kidney Organogenesis
BASIC RESEARCH www.jasn.org Conserved and Divergent Features of Human and Mouse Kidney Organogenesis Nils O. Lindström,1 Jill A. McMahon,1 Jinjin Guo,1 Tracy Tran,1 Qiuyu Guo,1 Elisabeth Rutledge,1 Riana K. Parvez,1 Gohar Saribekyan,1 Robert E. Schuler,2 Christopher Liao,1 Albert D. Kim,1 Ahmed Abdelhalim,1 Seth W. Ruffins,1 Matthew E. Thornton,3 Laurence Basking,4 Brendan Grubbs,3 Carl Kesselman,2,5 and Andrew P. McMahon1 1Department of Stem Cell Biology and Regenerative Medicine, Keck School of Medicine, 2Information Sciences Institute, Viterbi School of Engineering, 5Epstein Department of Industrial and Systems Engineering and Information Sciences Institute, Viterbi School of Engineering and Department of Preventive Medicine, Keck School of Medicine, and 3Maternal Fetal Medicine Division, University of Southern California, Los Angeles, California; and 4Department of Urology and Pediatrics, University of California San Francisco, San Francisco, California ABSTRACT Human kidney function is underpinned by approximately 1,000,000 nephrons, although the number varies sub- stantially, and low nephron number is linked to disease. Human kidney development initiates around 4 weeks of BASIC RESEARCH gestation and ends around 34–37 weeks of gestation. Over this period, a reiterative inductive process establishes the nephron complement. Studies have provided insightful anatomic descriptions of human kidney development, but the limited histologic views are not readily accessible to a broad audience. In this first paper in a series providing comprehensive insight into human kidney formation, we examined human kidney development in 135 anony- mously donated human kidney specimens. We documented kidney development at a macroscopic and cellular level through histologic analysis, RNA in situ hybridization, immunofluorescence studies, and transcriptional pro- filing, contrasting human development (4–23 weeks) with mouse development at selected stages (embryonic day 15.5 and postnatal day 2). -

Odd-Skipped Related 1 Is Required for Development of the Metanephric Kidney and Regulates Formation and Differentiation of Kidne
DEVELOPMENT AND DISEASE RESEARCH ARTICLE 2995 Development 133, 2995-3004 (2006) doi:10.1242/dev.02442 Odd-skipped related 1 is required for development of the metanephric kidney and regulates formation and differentiation of kidney precursor cells Richard G. James1,2,*, Caramai N. Kamei1,2,*, Qingru Wang3, Rulang Jiang3 and Thomas M. Schultheiss1,2,† Formation of kidney tissue requires the generation of kidney precursor cells and their subsequent differentiation into nephrons, the functional filtration unit of the kidney. Here we report that the gene odd-skipped related 1 (Odd1) plays an important role in both these processes. Odd1 is the earliest known marker of the intermediate mesoderm, the precursor to all kidney tissue. It is localized to mesenchymal precursors within the mesonephric and metanephric kidney and is subsequently downregulated upon tubule differentiation. Mice lacking Odd1 do not form metanephric mesenchyme, and do not express several other factors required for metanephric kidney formation, including Eya1, Six2, Pax2, Sall1 and Gdnf. In transient ectopic expression experiments in the chick embryo, Odd1 can promote expression of the mesonephric precursor markers Pax2 and Lim1. Finally, persistent expression of Odd1 in chick mesonephric precursor cells inhibits differentiation of these precursors into kidney tubules. These data indicate that Odd1 plays an important role in establishing kidney precursor cells, and in regulating their differentiation into kidney tubular tissue. KEY WORDS: Kidney, Metanephros, Mesonephros, Chick embryo, Mouse, Osr1, Odd1 INTRODUCTION form a precursor cell population, which both maintains itself at the tips Vertebrate kidney tissue is derived from the intermediate mesoderm of the ureteric bud (via proliferation and/or addition from the (IM), a strip of tissue located adjacent to the somites in the surrounding non-condensed mesenchyme) and gives off cells that developing embryo (Saxen, 1987). -

FGF-7 Modulates Kidney Size
Development 126, 547-554 (1999) 547 Printed in Great Britain © The Company of Biologists Limited 1999 DEV9568 FGF-7 modulates ureteric bud growth and nephron number in the developing kidney Jizeng Qiao1,‡, Robert Uzzo2,§, Tomoko Obara-Ishihara1, Linda Degenstein3, Elaine Fuchs3 and Doris Herzlinger1,2,* 1Department of Physiology and Biophysics and 2Department of Urology, Cornell University Medical College, New York, NY 10021, USA 3Howard Hughes Medical Institute, Department of Molecular Genetics and Cell Biology, University of Chicago, Chicago, IL 60637, USA ‡Present address: Renal Division, Brigham and Women’s Hospital, Boston, MA 02115, USA §Present address: The Cleveland Clinic Foundation, Cleveland, OH 44195, USA *Author for correspondence (e-mail: [email protected]) Accepted 20 November 1998; published on WWW 7 January 1999 SUMMARY The importance of proportioning kidney size to body mature collecting system of FGF-7-null kidneys is volume was established by clinical studies which markedly smaller than wild type. Furthermore, demonstrated that in-born defecits of nephron number morphometric analyses indicate that mature FGF-7-null predispose the kidney to disease. As the kidney develops, kidneys have 30±6% fewer nephrons than wild-type the expanding ureteric bud or renal collecting system kidneys. In vitro experiments demonstrate that elevated induces surrounding metanephric mesenchyme to levels of FGF-7 augment ureteric bud growth and increase proliferate and differentiate into nephrons. Thus, it is likely the number of nephrons that form in rodent metanephric that nephron number is related to ureteric bud growth. The kidney organ cultures. Collectively, these results expression patterns of mRNAs encoding Fibroblast Growth demonstrate that FGF-7 levels modulate the extent of Factor-7 (FGF-7) and its high affinity receptor suggested ureteric bud growth during development and the number that FGF-7 signaling may play a role in regulating ureteric of nephrons that eventually form in the kidney.