Organic Chemistry II / CHEM 252 Chapter 19 – Synthesis And
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Development of Novel Peptide Nucleic Acids A
b-Dicarbonyl compounds Compounds having two carbonyl groups separated by an intervening carbon atom are called b-dicarbonyl compounds, and these compounds are highly versatile reagents for organic synthesis. O O O O C C C R C C C OR' b b b-Dicarbonyl system b-Keto ester ** The pKa for such a proton is in the range 9-11, acidic enough to be removed easily by an alkoxide base to form an enolate. O O O H O OR C C C C C.. C + HOR H enolate anion pKa = 9-11 .. .. .. .. .. .. O . O . O O . O O C C C .. C C C R C OR' R C OR' R C OR' H H H Resonance structure of the anion of a b-keto ester Synthesis of b-keto ester (Claisen condensation) O O O NaOC H 2 2 5 CH3COC2H5 CH3CC..HCOC2H5 + C2H5OH + Na (removed by distillation) Sodiumacetoacetic ester HCl O O CH3CCH2COC2H5 Ethyl acetoacetate (acetoacetic ester) (76%) O O O O (1) NaOC2H5 R CH C + H CHC R CH2C CHCOC H + C H OH 2 OC2H5 OC2H5 + 2 5 2 5 (2) H3O R R (R may also be H) b-Keto ester Mechanism Step1 .. O O . .. + . OHC H + C H OH R CHC OC2H5 .. 2 5 RC.. H C OC2H5 2 5 H .. O . RCH C OC2H5 Step 2 .. .. .. .. O . O O O . RCH2C + . RCH2C CH C OC2H5 HC C OC2H5 . C H O . OC2H5 R 2 5 .. R .. O . O .. + . OC2H5 RCH2C CH C OC2H5 .. R Step 3 .. .. O . O H O O . -

Lonza's Chemical Network Diketene and HCN Derivatives, Heterocycles
Lonza’s chemical network Diketene and HCN derivatives, heterocycles and basic chemicals catalog Pharma&Biotech Nutrition Agriculture MaterialsScience PersonalCare Diketene and HCN derivatives, heterocycles and basic chemicals catalog Content About Lonza 3 Introduction 4 Diketene / ketene derivatives 6 Esters 6 Arylides 7 Alkylamides 8 Pyrazolones 9 Dehydroacetic acid 9 Lonzamon monomers 10 Other diketene derivatives 10 HCN derivatives 12 Heterocycles 14 Basic chemicals 16 Others 17 Alphabetical index 18 About Lonza Lonza is the global leader in the production and support of active phar- From 1897 to the present day combining Swiss tradition with global maceutical ingredients both chemically and biotechnologically. Biophar- experience, the company has had an enterprising character, adapting maceuticals are one of the key growth drivers of the pharmaceutical and its offerings and services to the needs of customers and to changing biotechnology industries. technologies. Throughout our history, we have maintained a strong culture of performance, results and dependability that is valued by all Lonza has strong capabilities in large and small molecules, peptides, of our customers. amino acids and niche bioproducts which play an important role in the development of novel medicines and healthcare products. In addition, Lonza’s cracker in Visp is the back bone of a comprehensive fully back- Lonza is a leader in cell-based research, endotoxin detection and cell ward integrated chemical network. Our product portfolio consists of therapy manufacturing. Furthermore, the company is a leading provider HCN- and diketene derivatives as well as basic chemicals which are key of chemical and biotech ingredients to the nutrition, hygiene, preserva- raw materials and intermediates for many sophisticated applications. -

Acetoacetic Ester Synthesis
Programme: B.Sc. B.ed. (Integrated) Course: ORGANIC CHEMISTRY- III Semester: VI Code: CHE-352 Topic: ETHYLACETOACETATEE Date- 07/04/2020 y Only PPe Dr. Angad Kumar Singh ForF Department of Chemistry, Central University of South Bihar, Gaya (Bihar) Note: These materials are only for classroom teaching purpose at Central University of South Bihar. All the data taken from several books, research articles including Wikipedia. Note: These materials are only for classroom teaching purpose at Central University of South Bihar. All the data taken from several books, research articles including Wikipedia. Ethylacetoacetate The Claisen Condensation between esters containing - hdhydrogens, promotdted byabase such as sodium ethoxy ide, toproduce a !- ketoester. One equiv serves as the nucleophilele (enolate)(eno and the other is OnlyO the electrophile which undergoes additiontion andae elimination. The use of stronger bases, e.g. sodium amide or sodiumsodUse hydride instead of sodium ethoxide, often increases the yield.d. al onal Ethylacetoacetate Note: These materials are only for classroom teaching purpose at Central University of South Bihar. All the data taken from several books, research articles including Wikipedia. Mechanism of the Claisen Condensation The reaction is driven to product by the final deprotonation step. Note: These materials are only for classroom teaching purpose at Central University of South Bihar. All the data taken from several books, research articles including Wikipedia. Mixed Claisen Condensation Like mixed aldol reactions, mixed Claisen condensations are useful if differences in reactivity exist betweenn they two esters as for example when one of the esters has no -hydrogenydrogOnlyO . Examples of such esters are: e Use ers excess Note: These materials are only for classroom teaching purpose at Central University of South Bihar. -
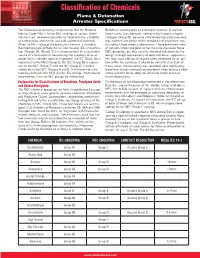
Classification of Chemicals
Classification of Chemicals Flame & Detonation Arrester Specifications PROTECTOSEAL ® The Protectoseal Company recommends that the National Butadiene would qualify as a Group D material. In each of Electric Code (NEC) Article 500, rankings of various chemi - these cases, the chemicals were primarly listed in a higher cals be used, whenever possible, to determine the suitability category (Group B), because of relatively high pressure read - of a detonation arrester for use with a particular chemical. ings noted in one phase of the standard test procedure con - When no NEC rating of the particular chemical is available, ducted by Underwriters Laboratories. These pressures were the International Electrotechnical Commission (IEC) classifica - of concern when categorizing the chemicals because these tion (Groups IIA, IIB and IIC) is recommended as a secondary NEC groupings are also used as standard indicators for the source of information for determining the suitability of an ar - design strength requirements of electrical boxes, apparatus, rester for its intended service. In general, the IEC Group IIA is etc. that must withstand the pressures generated by an igni - equivalent to the NEC Group D; the IEC Group IIB is equiva - tion within the container. It should be noted that, in each of lent to the NEC Group C; and the IEC Group IIC includes these cases, the test pressures recorded were significantly chemicals in the NEC Groups A and B. In the event of a dis - lower than those commonly encountered when testing a deto - crepancy between the NEC and the IEC ratings, Protectoseal nation arrester for its ability to withstand stable and over - recommends that the NEC groups be referenced. -

Regioselective Addition of 1,3-Dicarbonyl Dianion to N-Sulfonyl Aldimines: an Expedient Route to N-Sulfonyl Piperidines and N-Sulfonyl Azetidines
Tetrahedron 63 (2007) 4779–4787 Regioselective addition of 1,3-dicarbonyl dianion to N-sulfonyl aldimines: an expedient route to N-sulfonyl piperidines and N-sulfonyl azetidines Manas K. Ghorai,* Amit Kumar and Sandipan Halder Department of Chemistry, Indian Institute of Technology, Kanpur 208 016, India Received 26 June 2006; revised 23 February 2007; accepted 15 March 2007 Available online 18 March 2007 Abstract—A simple route for the synthesis of d-amino-b-keto esters and d-amino-b-diketones is reported. This involves regioselective addition of 1,3-dianions derived from ethyl acetoacetate and acetyl acetone to N-sulfonyl aldimines. The d-amino-b-keto ester derivatives were further converted into the corresponding N-sulfonyl piperidines and N-sulfonyl azetidines. Ó 2007 Elsevier Ltd. All rights reserved. 1. Introduction In continuation of our research on enolate chemistry and also supported by the related literature on the reactivity of 1,3-di- 1,3-Dicarbonyl dianions have been used extensively in or- carbonyl dianions, it was envisaged that dianions derived ganic synthesis for building ring systems. The anion on the from suitable precursors could be regioselectively added to terminal carbon of the dianion can be regioselectively reacted aldimines to give an easy access to d-amino-b-keto esters18 with 1 equiv of an electrophile resulting in an enolate ion, and their diketone analogues. These compounds could easily which can be trapped by the addition of a second electrophile be converted into various piperidine and azetidine deriva- -

Ethyl Acetoacetate (Acetoacetic Ester) (B) Diethyl Molonote (Molonic Ester) the Structure of (A) and (B) Are As
PRESENTATION OF ENOLATES Dr. Susmita Bajpai Department of Chemistry B.N.D. College, Kanpur ENOLATES The class of compounds which contain a methylene group (–CH2–) directly bonded to the electron withdrawing groups such as –COCH3, –COOC2H5, –CN, are called active methylene compounds. This is so because the –CH2 group in them is acidic and reactive. The two examples are (a) Ethyl acetoacetate (Acetoacetic ester) (b) Diethyl molonote (Molonic ester) The structure of (a) and (b) are as This reaction is known as claisen condensation Ethyl acetoacetate (CH3COOCH2COOC2H5) • It's IUPAC name is ethyl 3-oxobutanoate • Preparation: Ethyl acetoacetate is prepared by heating ethyl acetate with sodium ethoxide in ethanol, followed by acidification. Claisen condensation • It is a condensation reaction in which, two ester molecules condensed to form an alcohol and a b-Keto ester. Therefore ethyl acetoacetate is a b-Keto ester. • Mechanism: The mechanism involves three steps: + • Step-I :- First sodium ethoxide (C2H5O–Na ) breaks into ethoxide ion and sodium ion. This ethoxide ion attacks ethyl acetoacetate to give ethyl alcohol and ester anion. Step -II: Ester anion attacks the carbonyl group of a second molecules of ethyl acetate. • Step-III: Ethoxide ion is eliminated Properties • It is a colourless pleasant smelling liquid • b.p. - 180.4oC • It is sparingly soluble in water but freely so in organic solvent. • It is neutral to litmus. Chemical properties • It is a tautomeric mixture of Keto and enol forms. Therefore it gives the reaction of the various functional groups present in the two forms. Acidity of methylene hydrogen (Formation of salt) • In ethyl acetoacetate methylene group (–CH2–) flank by two carbonyl group. -

Ethyl Acetoacetate
21.6 The Acetoacetic Ester Synthesis Acetoacetic Ester O O C C H3C C OCH2CH3 H H Acetoacetic ester is another name for ethyl acetoacetate. The "acetoacetic ester synthesis" uses acetoacetic ester as a reactant for the preparation of ketones. Deprotonation of Ethyl Acetoacetate O O – C C + CH3CH2O H3C C OCH2CH3 H H Ethyl acetoacetate pKa ~ 11 can be converted readily to its anion with bases such as sodium ethoxide. Deprotonation of Ethyl Acetoacetate O O – C C + CH3CH2O H3C C OCH2CH3 H H Ethyl acetoacetate pKa ~ 11 can be converted readily to its anion K ~ 105 K ~ 10 with bases such as O O sodium ethoxide. C •• C + CH3CH2OH H3C –C OCH2CH3 3 2 H pKa ~ 16 Alkylation of Ethyl Acetoacetate O O The anion of ethyl C •• C acetoacetate can be H C C OCH CH 3 – C OCH2CH3 alkylated using an H alkyl halide (SN2: primary and R X secondary alkyl halides work best; tertiary alkyl halides undergo elimination). Alkylation of Ethyl Acetoacetate O O The anion of ethyl C •• C acetoacetate can be H C C OCH CH 3 – C OCH2CH3 alkylated using an H alkyl halide (SN2: primary and R X secondary alkyl O O halides work best; tertiary alkyl halides C C undergo elimination). H3C C OCH2CH3 H R Conversion to Ketone O O Saponification and C C acidification convert H3C C OH the alkylated H R derivative to the – 1. HO , H2O corresponding b-keto 2. H+ acid. O O The b-keto acid then undergoes C C decarboxylation to H C 3 C OCH2CH3 form a ketone. -

Development of New Domino Reactions of Alkylidene Meldrum's
Development of New Domino Reactions of Alkylidene Meldrum’s Acids Involving Friedel-Crafts Chemistry and Catalytic Conjugate Allylation of Alkylidene Meldrum’s Acids by Aaron Michael Dumas A thesis presented to the University of Waterloo in fulfilment of the thesis requirement for the degree of Doctor of Philosophy in Chemistry Waterloo, Ontario, Canada, 2009 © Aaron Michael Dumas 2009 I hereby declare that I am the sole author of this thesis. This is a true copy of my thesis, including any required final revisions, as accepted by my examiners. I understand that my thesis may be made electronically available to the public. ii Abstract Alkylidene Meldrum’s acids are very reactive acceptors in conjugate additions, and are known to be significantly more electrophilic than other α,β-unsaturated carbonyl electrophiles. They also offer advantages in terms of ease of preparation, purification and storage. Despite this, they are relatively underused in organic synthesis, and have been treated as something of a curiousity in the literature. The goal of my research was to demonstrate the utility of these molecules in new reactions that are not readily available to other electrophiles. To facilitate this work, new conditions for the Knoevenagel condensation of aldehydes with Meldrum’s acid were developed. This allowed access to a broader range of monosubstituted alkylidenes than was previously possible from any single method. In a reaction that exploits the acylating ability of Meldrum’s acid, a domino addition of phenols to alkylidene Meldrum’s acids was developed. Here, Yb(OTf)3 catalyzed the addition of a phenol to the alkylidene as well as acylation through activation of the electrophile. -

Determination of Solvent Effects on Ketoðenol Equilibria of 1,3-Dicarbonyl Compounds Using NMR: Revisiting a Classic Physical C
In the Laboratory Determination of Solvent Effects on Keto–Enol Equilibria W of 1,3-Dicarbonyl Compounds Using NMR Revisiting a Classic Physical Chemistry Experiment Gilbert Cook* and Paul M. Feltman Department of Chemistry, Valparaiso University, Valparaiso, IN 46383; *[email protected] “The influence of solvents on chemical equilibria was discovered in 1896, simultaneously with the discovery of keto–enol tautomerism in 1,3-dicarbonyl compounds” (1). The solvents were divided into two groups according to their ability to isomerize compounds. The study of the keto–enol tautomerism of β-diketones and β-ketoesters in a variety of solvents using proton NMR has been utilized as a physical Figure 1. The β-dicarbonyl compounds studied in the experiment. chemistry experiment for many years (2, 3). The first reported use of NMR keto–enol equilibria determination was by Reeves (4). This technique has been described in detail in an experiment by Garland, Nibler, and Shoemaker (2). panded (i) to give an in-depth analysis of factors influencing The most commonly used β-diketone for these experi- solvent effects in tautomeric equilibria and (ii) to illustrate ments is acetylacetone (Scheme I). Use of proton NMR is a the use of molecular modeling in determining the origin of viable method for measuring this equilibrium because the a molecule’s polarity. The experiment’s original benefits of tautomeric keto–enol equilibrium is slow on the NMR time using proton NMR as a noninvasive method of evaluating scale, but enol (2a)–enol (2b) tautomerism is fast on this scale equilibrium are maintained. (5). It has been observed that acyclic β-diketones and β- Experimental Procedure ketoesters follow Meyer’s rule of a shift in the tautomeric equi- librium toward the keto tautomer with increasing solvent Observations of the solvent effects for three other 1,3- polarity (6). -

Ethyl Acetoacetate
EUROPEAN COMMISSION JOINT RESEARCH CENTRE Institute for Health and Consumer Protection European Chemicals Bureau I-21020 Ispra (VA) Italy ETHYL ACETOACETATE CAS No: 141-97-9 EINECS No: 205-516-1 Summary Risk Assessment Report 2002 Special Publication I.02.75 ETHYL ACETOACETATE CAS No: 141-97-9 EINECS No: 205-516-1 SUMMARY RISK ASSESSMENT REPORT 2002 Germany The Rapporteur for Ethyl acetoacetate is the Federal Institute for Occupational Safety and Health. Contact point: Bundesanstalt für Arbeitsschutz und Arbeitsmedizin Anmeldestelle Chemikaliengesetz (BAuA) (Federal Institute for Occupational Safety and Health Notification Unit) Friedrich-Henkel-Weg 1-25 44149 Dortmund (Germany) fax: +49 (231) 9071-679 e-mail: [email protected] Date of Last Literature Search : 1996 Review of report by MS Technical Experts finalised: March 2001 Final report: 2002 © European Communities, 2002 PREFACE This report provides a summary, with conclusions, of the risk assessment report of the substance ethyl acetoacetate that has been prepared by Germany in the context of Council Regulation (EEC) No. 793/93 on the evaluation and control of existing substances. For detailed information on the risk assessment principles and procedures followed, the underlying data and the literature references the reader is referred to the original risk assessment report that can be obtained from the European Chemicals Bureau1. The present summary report should preferably not be used for citation purposes. 1 European Chemicals Bureau – Existing Chemicals – http://ecb.jrc.it III -

Review: Synthetic Methods for Amphetamine
Review: Synthetic Methods for Amphetamine A. Allen1 and R. Ely2 1Array BioPharma Inc., Boulder, Colorado 80503 2Drug Enforcement Administration, San Francisco, CA Abstract: This review focuses on synthesis of amphetamine. The chemistry of these methods will be discussed, referenced and precursors highlighted. This review covers the period 1985 to 2009 with emphasis on stereoselective synthesis, classical non-chiral synthesis and bio-enzymatic reactions. The review is directed to the Forensic Community and thus highlights precursors, reagents, stereochemistry, type and name reactions. The article attempts to present, as best as possible, a list of references covering amphetamine synthesis from 1900 -2009. Although this is the same fundamental ground as the recent publication by K. Norman; “Clandestine Laboratory Investigating Chemist Association” 19, 3(2009)2-39, this current review offers another perspective. Keywords: Review, Stereoselective, Amphetamine, Syntheses, references, Introduction: It has been 20 years since our last review of the synthetic literature for the manufacture of amphetamine and methamphetamine. Much has changed in the world of organic transformation in this time period. Chiral (stereoselective) synthetic reactions have moved to the forefront of organic transformations and these stereoselective reactions, as well as regio-reactions and biotransformations will be the focus of this review. Within the synthesis of amphetamine, these stereoselective transformations have taken the form of organometallic reactions, enzymatic reactions, ring openings, - aminooxylations, alkylations and amination reactions. The earlier review (J. Forensic Sci. Int. 42(1989)183-189) addressed for the most part, the ―reductive‖ synthetic methods leading to this drug of abuse. It could be said that the earlier review dealt with ―classical organic transformations,‖ roughly covering the period from 1900-1985. -

Download Author Version (PDF)
Journal of Materials Chemistry C Accepted Manuscript This is an Accepted Manuscript, which has been through the Royal Society of Chemistry peer review process and has been accepted for publication. Accepted Manuscripts are published online shortly after acceptance, before technical editing, formatting and proof reading. Using this free service, authors can make their results available to the community, in citable form, before we publish the edited article. We will replace this Accepted Manuscript with the edited and formatted Advance Article as soon as it is available. You can find more information about Accepted Manuscripts in the Information for Authors. Please note that technical editing may introduce minor changes to the text and/or graphics, which may alter content. The journal’s standard Terms & Conditions and the Ethical guidelines still apply. In no event shall the Royal Society of Chemistry be held responsible for any errors or omissions in this Accepted Manuscript or any consequences arising from the use of any information it contains. www.rsc.org/materialsC Page 1 of 27 Journal of Materials Chemistry C Journal Name RSC Publishing ARTICLE π-Expanded Coumarins: Synthesis, Optical Properties and Applications Cite this: DOI: 10.1039/x0xx00000x Mariusz Tasior, a Dokyoung Kim, b† Subhankar Singha, b† Maciej Krzeszewski, a Kyo Han Ahn,* b and Daniel T. Gryko* ac Manuscript Received 00th January 2014, Accepted 00th January 2014 Coumarins fused with other aromatic units have recently emerged as a hot topic of DOI: 10.1039/x0xx00000x research. Their synthesis is partly based on classical methodologies such as www.rsc.org/ Pechmann reaction or Knoevenagel condensation, but it also sparked the discovery of completely new pathways.