1 Nitrogen Cost Minimization Is Promoted by Structural Changes in the Transcriptome of N 1 Deprived Prochlorococcus Cells 2 3 Ro
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

UNIVERSITY of CALIFORNIA Los Angeles
UNIVERSITY OF CALIFORNIA Los Angeles Dissecting transcriptional control by Klf4 in somatic cell reprogramming A dissertation submitted in partial satisfaction of the requirements for the degree Doctor of Philosophy in Biological Chemistry by Huajun Zhou 2017 ABSTRACT OF THE DISSERTATION Dissecting transcriptional control by Klf4 in somatic cell reprogramming by Huajun Zhou Doctor of Philosophy in Biological Chemistry University of California, Los Angeles, 2017 Professor Gregory S. Payne, Chair Ectopic expression of four transcription factors, Oct4, Sox2, Klf4, and c-Myc, coverts somatic cells directly into induced pluripotent stem cells (iPSCs), which are functionally equivalent to embryonic stem cells (ESCs). The discovery of iPSC has been reshaping the methodology of disease modeling and drug screening in the past decade, and provides tremendous promise for regenerative medicine. However, the mechanism underlying this conversion process, reprogramming, is not yet fully understood. I aimed to dissect the reprogramming process, by characterizing the functional domains of one reprogramming factor Klf4. The transcriptional activation domain (TAD) of Klf4 was revealed to be critical for reprogramming. To search for the factors that mediates the functionality of Klf4 TAD, I identified transcriptional coactivators CBP/p300 and Mediator complex as the physical interaction partners of Klf4 TAD, and further showed that this interaction is functionally required ii for Klf4 mediated transcriptional activation in reprograming. Clathrin heavy chain, initially identified as a physical interaction partner of Klf4 TAD, was shown to be not required for Klf4 transcriptional activation. Clathrin heavy chain was furthered characterized for its potential transcriptional activation activity in CHC-TFE3, a chromosomal fusion discovered in renal cell carcinoma. -

A Computational Approach for Defining a Signature of Β-Cell Golgi Stress in Diabetes Mellitus
Page 1 of 781 Diabetes A Computational Approach for Defining a Signature of β-Cell Golgi Stress in Diabetes Mellitus Robert N. Bone1,6,7, Olufunmilola Oyebamiji2, Sayali Talware2, Sharmila Selvaraj2, Preethi Krishnan3,6, Farooq Syed1,6,7, Huanmei Wu2, Carmella Evans-Molina 1,3,4,5,6,7,8* Departments of 1Pediatrics, 3Medicine, 4Anatomy, Cell Biology & Physiology, 5Biochemistry & Molecular Biology, the 6Center for Diabetes & Metabolic Diseases, and the 7Herman B. Wells Center for Pediatric Research, Indiana University School of Medicine, Indianapolis, IN 46202; 2Department of BioHealth Informatics, Indiana University-Purdue University Indianapolis, Indianapolis, IN, 46202; 8Roudebush VA Medical Center, Indianapolis, IN 46202. *Corresponding Author(s): Carmella Evans-Molina, MD, PhD ([email protected]) Indiana University School of Medicine, 635 Barnhill Drive, MS 2031A, Indianapolis, IN 46202, Telephone: (317) 274-4145, Fax (317) 274-4107 Running Title: Golgi Stress Response in Diabetes Word Count: 4358 Number of Figures: 6 Keywords: Golgi apparatus stress, Islets, β cell, Type 1 diabetes, Type 2 diabetes 1 Diabetes Publish Ahead of Print, published online August 20, 2020 Diabetes Page 2 of 781 ABSTRACT The Golgi apparatus (GA) is an important site of insulin processing and granule maturation, but whether GA organelle dysfunction and GA stress are present in the diabetic β-cell has not been tested. We utilized an informatics-based approach to develop a transcriptional signature of β-cell GA stress using existing RNA sequencing and microarray datasets generated using human islets from donors with diabetes and islets where type 1(T1D) and type 2 diabetes (T2D) had been modeled ex vivo. To narrow our results to GA-specific genes, we applied a filter set of 1,030 genes accepted as GA associated. -

Supplementary Table S4. FGA Co-Expressed Gene List in LUAD
Supplementary Table S4. FGA co-expressed gene list in LUAD tumors Symbol R Locus Description FGG 0.919 4q28 fibrinogen gamma chain FGL1 0.635 8p22 fibrinogen-like 1 SLC7A2 0.536 8p22 solute carrier family 7 (cationic amino acid transporter, y+ system), member 2 DUSP4 0.521 8p12-p11 dual specificity phosphatase 4 HAL 0.51 12q22-q24.1histidine ammonia-lyase PDE4D 0.499 5q12 phosphodiesterase 4D, cAMP-specific FURIN 0.497 15q26.1 furin (paired basic amino acid cleaving enzyme) CPS1 0.49 2q35 carbamoyl-phosphate synthase 1, mitochondrial TESC 0.478 12q24.22 tescalcin INHA 0.465 2q35 inhibin, alpha S100P 0.461 4p16 S100 calcium binding protein P VPS37A 0.447 8p22 vacuolar protein sorting 37 homolog A (S. cerevisiae) SLC16A14 0.447 2q36.3 solute carrier family 16, member 14 PPARGC1A 0.443 4p15.1 peroxisome proliferator-activated receptor gamma, coactivator 1 alpha SIK1 0.435 21q22.3 salt-inducible kinase 1 IRS2 0.434 13q34 insulin receptor substrate 2 RND1 0.433 12q12 Rho family GTPase 1 HGD 0.433 3q13.33 homogentisate 1,2-dioxygenase PTP4A1 0.432 6q12 protein tyrosine phosphatase type IVA, member 1 C8orf4 0.428 8p11.2 chromosome 8 open reading frame 4 DDC 0.427 7p12.2 dopa decarboxylase (aromatic L-amino acid decarboxylase) TACC2 0.427 10q26 transforming, acidic coiled-coil containing protein 2 MUC13 0.422 3q21.2 mucin 13, cell surface associated C5 0.412 9q33-q34 complement component 5 NR4A2 0.412 2q22-q23 nuclear receptor subfamily 4, group A, member 2 EYS 0.411 6q12 eyes shut homolog (Drosophila) GPX2 0.406 14q24.1 glutathione peroxidase -

Supplementary Material
BMJ Publishing Group Limited (BMJ) disclaims all liability and responsibility arising from any reliance Supplemental material placed on this supplemental material which has been supplied by the author(s) J Neurol Neurosurg Psychiatry Page 1 / 45 SUPPLEMENTARY MATERIAL Appendix A1: Neuropsychological protocol. Appendix A2: Description of the four cases at the transitional stage. Table A1: Clinical status and center proportion in each batch. Table A2: Complete output from EdgeR. Table A3: List of the putative target genes. Table A4: Complete output from DIANA-miRPath v.3. Table A5: Comparison of studies investigating miRNAs from brain samples. Figure A1: Stratified nested cross-validation. Figure A2: Expression heatmap of miRNA signature. Figure A3: Bootstrapped ROC AUC scores. Figure A4: ROC AUC scores with 100 different fold splits. Figure A5: Presymptomatic subjects probability scores. Figure A6: Heatmap of the level of enrichment in KEGG pathways. Kmetzsch V, et al. J Neurol Neurosurg Psychiatry 2021; 92:485–493. doi: 10.1136/jnnp-2020-324647 BMJ Publishing Group Limited (BMJ) disclaims all liability and responsibility arising from any reliance Supplemental material placed on this supplemental material which has been supplied by the author(s) J Neurol Neurosurg Psychiatry Appendix A1. Neuropsychological protocol The PREV-DEMALS cognitive evaluation included standardized neuropsychological tests to investigate all cognitive domains, and in particular frontal lobe functions. The scores were provided previously (Bertrand et al., 2018). Briefly, global cognitive efficiency was evaluated by means of Mini-Mental State Examination (MMSE) and Mattis Dementia Rating Scale (MDRS). Frontal executive functions were assessed with Frontal Assessment Battery (FAB), forward and backward digit spans, Trail Making Test part A and B (TMT-A and TMT-B), Wisconsin Card Sorting Test (WCST), and Symbol-Digit Modalities test. -

"Description"" ""Generatio"
TABLE S4 "ID ""Description"" ""GeneRatio"" ""BgRatio"" ""pvalue"" ""p.adjust"" ""qvalue"" ""geneID"" ""Count""" "GO:0003712 ""GO:0003712"" ""transcription coregulator activity"" ""84/1859"" ""454/22744"" 9.49597175224444e-13 9.80933882006851e-10 8.20651874588704e-10 ""Ncoa2/Zfp451/Dhx9/Hnrnpu/Cited2/Ncoa7/Ccar1/ Sirt1/Arid5b/Sirt6/Med1/Rara/Atxn7l3/Ddx5/Wbp2/Hdac9/Zmynd11/Cdyl/ Mier3/Sfmbt1/Gata4/Med4/Basp1/Zfpm2/Zhx2/Ddx17/Mkl2/Hes1/Nrip1/Usp16/ Elob/Rrp1b/Rxrb/Kat2b/Mta3/Hsbp1l1/Tle4/Sfr1/Eid1/Cops2/Sox12/Raly/ Ncoa6/Rbm39/Lpin3/Skil/Jade1/Maml3/Supt20/Med12l/Hdgf/Glmp/Nfib/Jun/ Pex14/Rere/Psmd9/Ncor2/Trim24/Ruvbl1/Rybp/Bhlhe40/Atf7ip/Ube3a/Mef2a/ Nrg1/Rbpms/Cnot7/Sin3b/Pou4f2/Pkn1/Cdyl2/Taf5l/Irf2bp2/Birc2/Yap1/ Skor1/Tfdp2/Rad54l2/Ctnnb1/Limd1/Med14/Rap2c/Tbl1x"" 84" "GO:0003779 ""GO:0003779"" ""actin binding"" ""65/1859"" ""414/22744"" 2.57546466939442e-07 8.86818334494813e-05 7.41914559148359e-05 ""Actr3/Cxcr4/Hnrnpu/Enah/Utrn/Epb41l2/Marcks/Ctnna3/Eef2/Pawr/ Ccdc88a/Anxa6/Gas7/Lasp1/Tns4/Syne2/Sipa1l1/Syne3/Phactr1/Enc1/Pxk/ Vcl/Ang/Myo10/Mtss1/Triobp/Mkl2/Afdn/Daam2/Svil/Ctnna1/Synpo/Myo5b/ Nrap/Ablim1/Shtn1/Fmnl2/Itprid2/Ino80/Pfn2/Myoz2/Pdlim5/Cap1/Macf1/ Epb41/Wasf2/Myom3/Ywhah/Coro1c/Ssh1/Hip1/Ppp1r9a/Wasl/Ctnna2/Mical3/ Eps8/Tlnrd1/Myom2/Klhl2/Sntb2/Spire2/Coro2b/Clasp2/Hdac6/Diaph2"" 65" "GO:0046332 ""GO:0046332"" ""SMAD binding"" ""22/1859"" ""84/22744"" 6.87941766027971e-07 0.000177660961076723 0.000148631628923412 ""Bmpr2/Cited2/Usp15/Ddx5/Axin2/Ppm1a/Yy1/Gata4/Tgif1/Ldlrad4/Smad7/ Acvr2a/Pmepa1/Skil/Trim33/Jun/Mef2a/Ipo7/Skor1/Rnf111/Tcf12/Ctnnb1"" -

REVIEWS Transcriptional Regulation Through Mediator-Like Coactivators
TIBS 25 – JUNE 2000 REVIEWS termed Mediator, that was necessary to support activated transcription in vitro in conjunction with general initiation Transcriptional regulation factors8, but apparently distinct from the TAF (Ref. 5) and USA (Ref. 7) coacti- through Mediator-like vators identified in metazoans (see below). Concurrent genetic analysis also implicated two broad groups of pro- coactivators in yeast and teins in transcriptional regulation in vivo. The SRB (suppressor of RNA metazoan cells polymerase B) proteins first emerged from genetic screens for suppressors of partial truncations in the C-terminal domain (CTD) of the largest subunit of Sohail Malik and Robert G. Roeder Pol II (Ref. 9). Other gene products, including GAL11, RGR1 and SIN4, emerged from genetic screens for regulatory A novel multiprotein complex has recently been identified as a coactivator for factors that function in other path- transcriptional control of protein-encoding genes by RNA polymerase II in ways10 (see below). Subsequent studies higher eukaryotic cells. This complex is evolutionarily related to the Mediator aimed at purifying the Mediator activity complex from yeast and, on the basis of its structural and functional charac- and the SRB proteins resulted in the teristics, promises to be a key target of diverse regulatory circuits. identification of a Pol II holoenzyme that contained the 12-subunit core Pol II, SRBs, other genetically identified TRANSCRIPTIONAL REGULATION IN factors that mediate, and might integrate, polypeptides (including GAL11, RGR1, eukaryotes is achieved through multi- the effects of transcriptional activators SIN4) and novel Mediator polypeptides protein complexes assembled at the on the Pol II basal machinery2. -

Recombinant Human MED4 Protein Catalog Number: ATGP0552
Recombinant human MED4 protein Catalog Number: ATGP0552 PRODUCT INPORMATION Expression system E.coli Domain 1-270aa UniProt No. Q9NPJ6 NCBI Accession No. AAH05189 Alternative Names Mediator complex subunit 4, ARC36, DRIP36, VDRIP, Mediator complex subunit 4 Activator-recruited cofactor 36 kDa component, HSPC126, Mediator of RNA polymerase II transcription subunit 4, TRAP/SMCC/PC2 subunit p36 subunit, Vitamin D3 receptor-interacting protein complex 36 kDa component. PRODUCT SPECIFICATION Molecular Weight 30.7 kDa (278aa) confirmed by MALDI-TOF Concentration 0.5mg/ml (determined by Bradford assay) Formulation Liquid in. 20mM Tris-HCl buffer (pH 8.0) containing 20% glycerol, 1mM DTT, 100mM NaCl Purity > 80% by SDS-PAGE Tag His-Tag Application SDS-PAGE Storage Condition Can be stored at +2C to +8C for 1 week. For long term storage, aliquot and store at -20C to -80C. Avoid repeated freezing and thawing cycles. BACKGROUND Description Mediator complex subunit 4 (MED4) is also known as mediator of RNA polymerase II transcription subunit 4 or vitamin D3 receptor-interacting protein complex 36 kDa component (DRIP36). This protein is a component of the vitamin D receptor-interacting protein (DRIP) complex which functions as a nuclear receptor coactivator. The DRIP complex is capable of activating nuclear receptors in a ligand-dependent manner. Recombinant human 1 Recombinant human MED4 protein Catalog Number: ATGP0552 MED4, fused to His-tag at C-terminus, was expressed in E. coli and purified by using conventional chromatography techniques. Amino acid Sequence MAASSSGEKE KERLGGGLGV AGGNSTRERL LSALEDLEVL SRELIEMLAI SRNQKLLQAG EENQVLELLI HRDGEFQELM KLALNQGKIH HEMQVLEKEV EKRDGDIQQL QKQLKEAEQI LATAVYQAKE KLKSIEKARK GAISSEEIIK YAHRISASNA VCAPLTWVPG DPRRPYPTDL EMRSGLLGQM NNPSTNGVNG HLPGDALAAG RLPDVLAPQY PWQSNDMSMN MLPPNHSSDF LLEPPGHNKE DEDDVEIMST DSSSSSSESD <LEHHHHHH> General References Rachez C., et al. -

Table S1 Ray-Finned Fish Genes of the Non-Genomic and Genomic Androgen Receptor Signaling Pathway According to Netpath, GO0030521, Bennett Et Al
Table S1 Ray-finned fish genes of the non-genomic and genomic Androgen Receptor signaling pathway according to NetPath, GO0030521, Bennett et al. 2010, Foradori et al. 2008 Gene Species Gene Species L.oculatus D.rerio A.mexicanus G.aculeatus T.nigroviridis T.rubripes P.formosa X.maculatus O.latipes O.niloticus N.brichardi M.zebra P.nyererei A.burtoni Source of gene Pathway association AKT1 ENSLOCG00000012762 NM_001281801.1 ENSAMXG00000009230 ENSGACG00000006298 ENSTNIG00000019495 ENSTRUG00000013476 ENSPFOG00000010125 ENSXMAG00000006673 ENSORLG00000017024 gi_542229192 gi_583981523 gi_498935125 gi_548423349 gi_554812288 NetPath, Bennett et al 2010, Foradori et al 2008 non-genomic, genomic ENSAMXG00000012355 AR ENSLOCG00000014680 ARA ENSAMXG00000013256 ENSGACG00000018525 ENSTNIG00000015783 ENSTRUG00000005373 ENSPFOG00000006490 ENSXMAG00000002896 ENSORLG00000008220 ENSONIG00000017538 gi_583972812 gi_499025148 gi_545793521 gi_555943746 NetPath receptor, genomic, non-genomic ARB ENSDARG00000067976 ENSAMXG00000011395 ENSGACG00000020332 ENSTNIG00000011826 ENSTRUG00000012421 ENSPFOG00000019378 ENSXMAG00000012307 ENSORLG00000009520 ENSONIG00000012854 gi_584009291 gi_499011353 gi_548397957 gi_555943801 NetPath receptor, genomic, non-genomic ARID1 ENSLOCG00000003798 ARID1AA ENSDARG00000101710 ENSAMXG00000020237 ENSGACG00000007244 ENSTNIG00000009659 ENSTRUG00000013351 ENSPFOG00000009717 ENSXMAG00000010716 ENSORLG00000004410 ENSONIG00000006764 gi_584001874 gi_499018427 gi_548435329 gi_554870576 GO:0030521 genomic ARID1AB ENSDARG00000101891 ENSAMXG00000013606 -
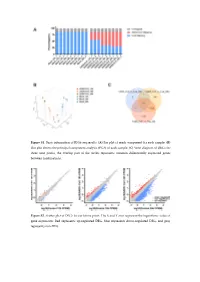
Figure S1. Basic Information of RNA-Seq Results. (A) Bar Plot of Reads Component for Each Sample
Figure S1. Basic information of RNA-seq results. (A) Bar plot of reads component for each sample. (B) Dot plot shows the principal component analysis (PCA) of each sample. (C) Venn diagram of DEGs for three time points, the overlap part of the circles represents common differentially expressed genes between combinations. Figure S2. Scatter plot of DEGs for each time point. The X and Y axes represent the logarithmic value of gene expression. Red represents up-regulated DEG, blue represents down-regulated DEG, and gray represents non-DEG. Table S1. Primers used for quantitative real-time PCR analysis of DEGs. Gene Primer Sequence Forward 5’-CTACGAGTGGATGGTCAAGAGC-3’ FOXO1 Reverse 5’-CCAGTTCCTTCATTCTGCACACG-3’ Forward 5’-GACGTCCGGCATCAGAGAAA-3’ IRS2 Reverse 5’-TCCACGGCTAATCGTCACAG-3’ Forward 5’-CACAACCAGGACCTCACACC-3’ IRS1 Reverse 5’-CTTGGCACGATAGAGAGCGT-3’ Forward 5’-AGGATACCACTCCCAACAGACCT-3’ IL6 Reverse 5’-CAAGTGCATCATCGTTGTTCATAC-3’ Forward 5’-TCACGTTGTACGCAGCTACC-3’ CCL5 Reverse 5’-CAGTCCTCTTACAGCCTTTGG-3’ Forward 5’-CTGTGCAGCCGCAGTGCCTACC-3’ BMP7 Reverse 5’-ATCCCTCCCCACCCCACCATCT-3’ Forward 5’-CTCTCCCCCTCGACTTCTGA-3’ BCL2 Reverse 5’-AGTCACGCGGAACACTTGAT-3’ Forward 5’-CTGTCGAACACAGTGGTACCTG-3’ FGF7 Reverse 5’-CCAACTGCCACTGTCCTGATTTC-3’ Forward 5’-GGGAGCCAAAAGGGTCATCA-3’ GAPDH Reverse 5’-CGTGGACTGTGGTCATGAGT-3’ Supplementary material: Differentially expressed genes log2(SADS-CoV_12h/ Qvalue (SADS-CoV _12h/ Gene Symbol Control_12h) Control_12h) PTGER4 -1.03693 6.79E-04 TMEM72 -3.08132 3.66E-04 IFIT2 -1.02918 2.11E-07 FRAT2 -1.09282 4.66E-05 -

Biochemistry. in the Article “The Med1 Subunit of the Yeast Mediator Complex Is Involved in Both Transcriptional Activation An
3330 Corrections Proc. Natl. Acad. Sci. USA 96 (1999) Biochemistry. In the article “The Med1 subunit of the yeast Neurobiology. In the article “Caspase-1 is activated in neural mediator complex is involved in both transcriptional activation cells and tissue with amyotrophic lateral sclerosis-associated and repression” by Darius Balciunas, Cecilia Ga¨lman,Hans mutations in copper-zinc superoxide dismutase” by Piera Ronne, and Stefan Bjo¨rklund, which appeared in number 2, Pasinelli, David R. Borchelt, Megan K. Houseweart, Don W. January 19, 1999, of Proc. Natl. Acad. Sci. USA (96, 376–381), Cleveland, and Robert H. Brown, Jr., which appeared in due to a printer’s error, the affiliation symbols were incorrect. number 26, December 22, 1998, of Proc. Natl. Acad. Sci. USA The correct author line, affiliation line, and address footnotes (95, 15763–15768), the following corrections should be noted. appear below. An erroneous version of Fig. 6 was published. The lane indicated as G41D represents lumbo-sacral spinal cord extract from G85R transgenic mice. In Fig. 7a, cell viability is ex- DARIUS BALCIUNAS*†,CECILIA GALMAN¨ ‡§, pressed as % of untreated cells and not as % of viability. HANS RONNE*†, AND STEFAN BJORKLUND¨ ‡¶ ‡Department of Medical Biochemistry and Biophysics, Umeå University, Physiology. In the article “Inositol 1,4,5-tris-phosphate acti- S-901 87 Umeå, Sweden; and *Department of Medical Biochemistry and vation of inositol tris-phosphate receptor Ca21 channel by Microbiology, Uppsala University Biomedical Center, Box 582, 751 23 21 Uppsala, Sweden ligand tuning of Ca inhibition” by Don-On Daniel Mak, Sean McBride, and J. Kevin Foskett, which appeared in number 26, December 22, 1998, of Proc. -

Functional Characterization of High Levels of Meningioma 1 As Collaborating Oncogene in Acute Leukemia
Leukemia (2010) 24, 601–612 & 2010 Macmillan Publishers Limited All rights reserved 0887-6924/10 $32.00 www.nature.com/leu ORIGINAL ARTICLE Functional characterization of high levels of meningioma 1 as collaborating oncogene in acute leukemia T Liu1, D Jankovic1, L Brault1, S Ehret1, F Baty1, V Stavropoulou1, V Rossi2, A Biondi2 and J Schwaller1 1Department of Biomedicine, University Hospital Basel, Basel, Switzerland and 2The Centro M. Tettamanti, Clinica Pediatrica, Universita` Milano-Bicocca, Monza, Italy Retroviral expression of leukemogenic oncogenes in the system by a conditional knock-in strategy resulted in the murine hematopoietic system is essential but not sufficient to formation of T-cell lymphomas as well as AML after a long induce acute leukemia. Proviral integration-mediated elevated latency, suggesting that MN1-TEL, similar to MLL-X fusions, is expression of the meningioma 1 (MN1) oncogene suggested 4–6 MN1 acting as cooperating event in mixed-lineage leukemia 1 essential but not sufficient to induce the disease. Gene (MLL) and eleven nineteen leukemia (ENL)-induced murine expression profiling studies of a large number of human leukemia leukemia. Indeed, co-expression of MN1 with MLL-ENL en- samples showed that MN1 is deregulated in cases with alterations hanced transformation in vivo, and resulted in a significantly at 3q26 leading to ecotropic virus integration-1 (EVI1) over- reduced latency for induction of an aggressive acute leukemia expression.7 In addition, elevated MN1 expression has been when compared with MN1 or MLL-ENL alone. In addition, co- associated with the presence of inv16 leading to a core-binding expression of MN1 increased the granulocyte macrophage 8 progenitor cell population with leukemia-initiating properties as factor-b/MYH11 fusion. -

Lethal Mitochondrial Cardiomyopathy in a Hypomorphic Med30 Mouse Mutant Is Ameliorated by Ketogenic Diet
Lethal mitochondrial cardiomyopathy in a hypomorphic Med30 mouse mutant is ameliorated by ketogenic diet Philippe Krebsa,1, Weiwei Fanb, Yen-Hui Chenc, Kimimasa Tobitad, Michael R. Downesb, Malcolm R. Woode, Lei Suna, Xiaohong Lia, Yu Xiaa, Ning Dingb, Jason M. Spaethf, Eva Marie Y. Morescoa, Thomas G. Boyerf, Cecilia Wen Ya Lod, Jeffrey Yenc, Ronald M. Evansb, and Bruce Beutlera,2,3 aDepartment of Genetics and eCore Microscopy Facility, The Scripps Research Institute, La Jolla, CA 92037; bThe Salk Institute, Howard Hughes Medical Institute, La Jolla, CA 92037; cInstitute of Biomedical Sciences, Academia Sinica, 11529 Taipei, Taiwan; dDepartment of Developmental Biology, Rangos Research Center, Pittsburgh, PA 15201; and fDepartment of Molecular Medicine, University of Texas Health Science Center, San Antonio, TX 78245 Contributed by Bruce Beutler, October 31, 2011 (sent for review September 24, 2011) Deficiencies of subunits of the transcriptional regulatory complex sociated with any phenotype in living organisms. In contrast to Mediator generally result in embryonic lethality, precluding study of other models with Mediator deficiencies, homozygous zeitgeist its physiological function. Here we describe a missense mutation in mice are physically indistinguishable from littermates at the time Med30 causing progressive cardiomyopathy in homozygous mice of weaning, but develop progressive cardiomyopathy that is in- that, although viable during lactation, show precipitous lethality 2– variably fatal by 7 wk of age. Mechanistically, the Med30zg mutation 3 wk after weaning. Expression profiling reveals pleiotropic changes causes a progressive and selective decline in the transcription of in transcription of cardiac genes required for oxidative phosphoryla- genes necessary for oxidative phosphorylation (OXPHOS) and tion and mitochondrial integrity.