1 Summary of Product Characteristics 1. Name Of
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Download Leaflet View the Patient Leaflet in PDF Format
Package leaflet: Information for the patient Fosinopril Sodium 10 mg Tablets Fosinopril Sodium 20 mg Tablets Fosinopril sodium Read all of this leaflet carefully before you start taking this medicine because it contains important information for you. - Keep this leaflet. You may need to read it again. - If you have any further questions, ask your doctor or pharmacist. - This medicine has been prescribed for you only. Do not pass it on to others. It may harm them, even if their signs of illness are the same as yours. - If you get any side effects, talk to your doctor or pharmacist. This includes any possible side effects not listed in this leaflet. See section 4. What is in this leaflet 1. What Fosinopril is and what it is used for 2. What you need to know before you take Fosinopril 3. How to take Fosinopril 4. Possible side effects 5. How to store Fosinopril 6. Contents of the pack and other information. 1. What Fosinopril is and what is it used for Fosinopril belongs to the class of medicines called Angiotensin Converting Enzyme (ACE) inhibitors that act on the heart and blood vessels. You may have been given Fosinopril to: • lower your blood pressure if it is too high (a condition called hypertension) • help your heart pump blood around your body if you have a condition known as heart failure and are also being treated with diuretics (medicines which help to remove excess fluid from the body). 2. What you need to know before you take Fosinopril Do not take Fosinopril: • if you are allergic to fosinopril or any other ACE inhibitor, or any of the other ingredients of this medicine (listed in section 6). -

Angiotensin-Converting Enzyme Inhibition but Not Angiotensin II Receptor Blockade Regulates Matrix Metalloproteinase Activity in Patients with Glomerulonephritis
J Am Soc Nephrol 14: 2861–2872, 2003 Angiotensin-Converting Enzyme Inhibition but not Angiotensin II Receptor Blockade Regulates Matrix Metalloproteinase Activity in Patients with Glomerulonephritis NADE` GE LODS,* PAOLO FERRARI,* FELIX J. FREY,* ANDREAS KAPPELER,† CELINE BERTHIER,* BRUNO VOGT,* and HANS-PETER MARTI* *Division of Nephrology and Hypertension, Inselspital Bern, Bern, Switzerland; and †Institute of Pathology, University of Bern, Bern, Switzerland Abstract. Equivalent long-term effects on the kidney are attrib- periods of 4 wk each without therapy. Untreated patients with uted to angiotensin-converting enzyme inhibitors (ACEI) and glomerulonephritis displayed distinctively higher serum levels angiotensin II type 1 receptor blockers (ARB). Nevertheless, it of MMP-2 but much lower MMP-1/-8/-9 concentrations com- is unknown to which degree effects of these compounds on pared with healthy control subjects. Immunohistology of individual inflammatory mediators, including matrix metallo- MMP-2 and MMP-9 in kidney biopsy specimen was accord- proteinases (MMP), are comparable. On the basis of structural ingly. However, these patients excreted higher amounts of and functional differences, it was hypothesized that ACEI and MMP-2 and MMP-9 in urine than healthy control subjects, ARB differentially regulate MMP activity. In a randomized, possibly reflecting ongoing glomerular inflammation. In pa- prospective crossover trial, the effect of an ACEI (fosinopril; tients with glomerulonephritis, ACEI significantly reduced 20 mg/d) and of an ARB (irbesartan; 150 mg/d) on MMP overall MMP serum activity to 25%, whereas ARB did not activity was evaluated. Ten hypertensive patients with glomer- show any effect. Activities of MMP-1/-2/-8/-9 were also sig- ulonephritis and normal or mildly reduced creatinine clearance nificantly inhibited by fosinopril but not by irbesartan. -
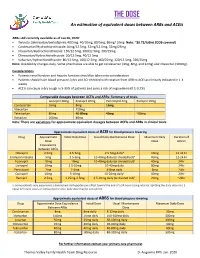
THE DOSE an Estimation of Equivalent Doses Between Arbs and Aceis
THE DOSE An estimation of equivalent doses between ARBs and ACEIs ARBs still currently available as of Jan 26, 2020: Twynsta (telmisartan/amlodipine): 40/5mg. 40/10mg, 80/5mg, 80mg/ 10mg Note: ~$0.73/tablet (ODB covered) Candesartan/Hydrochlorothiazide:16mg/12.5mg, 32mg/12.5mg, 32mg/25mg Irbesartan/Hydrochlorothiazide: 150/12.5mg, 300/12.5mg, 300/25mg Olmesartan/Hydrochlorothiaizde: 20/12.5mg, 40/12.5mg Valsartan/Hydrochlorothiazide: 80/12.5mg, 160/12.5mg, 160/25mg, 320/12.5mg, 320/25mg Note: Availability changes daily. Some pharmacies are able to get candesartan (4mg, 8mg, and 32mg) and irbesartan (300mg). Considerations Patients renal function and hepatic function should be taken into consideration Patients should have blood pressure, lytes and SCr checked with rotation from ARB to ACEI as clinically indicated in 1-4 weeks ACEIs can cause a dry cough in 5-35% of patients and carry a risk of angioedema (0.1-0.2%) Comparable dosages between ACEIs and ARBs- Summary of trials Lisinopril 20mg Enalapril 20mg Perindopril 4mg Ramipril 10mg Candesartan 16mg 8mg 16mg Irbesartan 150mg Telmisartan 80mg 40-80mg 40mg ~80mg Valsartan 160mg 80mg Note: There are variations for approximate equivalent dosages between ACEIs and ARBs in clinical trials. Approximate equivalent doses of ACEI for blood pressure lowering Drug Approximate Initial Daily Dose Usual Daily Maintenance Dose Maximum Daily Duration of Dose Dose Action Equivalence Between ACEIs Cilazapril 2.5mg 2.5-5mg 2.5-5mg dailya 10mg 12-24 hr Enalapril maleate 5mg 2.5-5mg 10-40mg daily (or divided bid)a 40mg 12-24 hr Fosinopril 10mg 10mg 10-40mg daily (or divided bid)a 40mg 24hr Lisinopril 10mg 2.5-10mg 10-40mg daily 80mg 24hr Perindopril 2mg 2-4mg 4-8mg daily 8mg 24hr Quinapril 10mg 5-10mg 10-20mg dailya 40mg 24hr Ramipril 2.5mg 1.25mg-2.5mg 2.5-10mg daily (or divided bid)a 20mg ~24hr a: Some patients may experience a diminished antihypertensive effect toward the end of a 24-hour dosing interval. -

Angiotensin-Converting Enzyme (ACE) Inhibitors
Angiotensin-Converting Enzyme (ACE) Inhibitors Summary Blood pressure reduction is similar for the ACE inhibitors class, with no clinically meaningful differences between agents. Side effects are infrequent with ACE inhibitors, and are usually mild in severity; the most commonly occurring include cough and hypotension. Captopril and lisinopril do not require hepatic conversion to active metabolites and may be preferred in patients with severe hepatic impairment. Captopril differs from other oral ACE inhibitors in its rapid onset and shorter duration of action, which requires it to be given 2-3 times per day; enalaprilat, an injectable ACE inhibitor also has a rapid onset and shorter duration of action. Pharmacology Angiotensin Converting Enzyme Inhibitors (ACE inhibitors) block the conversion of angiotensin I to angiotensin II through competitive inhibition of the angiotensin converting enzyme. Angiotensin is formed via the renin-angiotensin-aldosterone system (RAAS), an enzymatic cascade that leads to the proteolytic cleavage of angiotensin I by ACEs to angiotensin II. RAAS impacts cardiovascular, renal and adrenal functions via the regulation of systemic blood pressure and electrolyte and fluid balance. Reduction in plasma levels of angiotensin II, a potent vasoconstrictor and negative feedback mediator for renin activity, by ACE inhibitors leads to increased plasma renin activity and decreased blood pressure, vasopressin secretion, sympathetic activation and cell growth. Decreases in plasma angiotensin II levels also results in a reduction in aldosterone secretion, with a subsequent decrease in sodium and water retention.[51035][51036][50907][51037][24005] ACE is found in both the plasma and tissue, but the concentration appears to be greater in tissue (primarily vascular endothelial cells, but also present in other organs including the heart). -

Antidepressant Activity of Fosinopril, Ramipril and Losartan, but Not of Lisinopril in Depressive Paradigms of Albino Rats and Mice
Indian Journal of Experimental Biology Vol. 46, March 2008, pp. 180-184 Antidepressant activity of fosinopril, ramipril and losartan, but not of lisinopril in depressive paradigms of albino rats and mice Veena Nayak & P A Patil* Department of Pharmacology & Pharmacotherapeutics, J.N.Medical College, Belgaum 590 010, India Received 15 January 2007; revised 15 January 2008 Fosinopril, ramipril and losartan significantly decreased the duration (sec) of immobility in forced swim test and were comparable to amitriptyline. The duration of immobility were significantly decreased in fosinopril, ramipril and losartan in the tail suspension test and were comparable to amitriptyline. Only losartan significantly increased the rearing number of entries, time spent (sec) in open arm and in light area in comparison to control animals. Fosinopril and ramipril and not lisinopril showed significant antidepressant activity while losartan showed a significant antidepressant and anxiolytic activity. Present findings suggest that these drugs could be better antihypertensives in hypertensive patients with co- morbidity like depression or anxiety. Keywords: Anxiety, Depression, Fosinopril, Lisinopril, Losartan, Ramipril Physiological depression and anxiety when become possess antidepressant and anxiolytic activity like severe and chronic lead to a variety of psychiatric captopril and enalapril. A report regarding quinapril13 disorders. Quite often such mood disorders could be indicates that all ACE inhibitors may not share secondary to a number of cardiovascular and antidepressant and anxiolytic activity and such endocrinal disorders1,2 requiring treatment with two activities are not probed with other ACE inhibitors drugs, one for physical or metabolic and the other for used clinically. It is therefore planned to investigate associated mental disorder. -

Switching Ace-Inhibitors
Switching Ace-inhibitors http://www.ksdl.kamsc.org.au/dtp/switching_ace_inhibitors.html Change to → Enalapril Quinapril Ramipril Change from ↓ (Once daily dosing) (Once daily dosing) (Once daily dosing) Captopril Captopril 12.5mg daily Enalapril 2.5mg1 Quinapril 2.5mg Ramipril 1.25mg Captopril 25mg daily Enalapril 5mg1 Quinapril 5mg Ramipril 1.25-2.5mg Captopril 50mg daily Enalapril 7.5mg1 Quinapril 10mg Ramipril 2.5-5mg Captopril 100mg daily Enalapril 20mg1 Quinapril 20mg Ramipril 5-10mg2 Captopril 150mg daily Enalapril 40mg Quinapril 40mg Ramipril 10mg Fosinopril Fosinopril 5mg daily Enalapril 5mg Quinapril 5mg Ramipril 1.25mg Fosinopril 10mg daily Enalapril 10mg Quinapril 10mg Ramipril 2.5mg Fosinopril 20mg daily Enalapril 20mg Quinapril 20mg Ramipril 5mg Fosinopril 40mg daily Enalapril 40mg Quinapril 40mg Ramipril 10mg Lisinopril Lisinopril 5mg daily Enalapril 5mg Quinapril 5mg Ramipril 1.25mg Lisinopril 10mg daily Enalapril 10mg Quinapril 10mg Ramipril 2.5mg Lisinopril 20mg daily Enalapril 20mg Quinapril 20mg Ramipril 5mg Lisinopril 40mg Enalapril 40mg Quinapril 40mg Ramipril 10mg Perindopril Perindopril 2mg daily Enalapril 5-10mg Quinapril 5-10mg Ramipril 2.5mg Perindopril 4mg daily Enalapril 10mg-20mg Quinapril 10mg-20mg Ramipril 5mg Perindopril 8mg daily Enalapril 20-40mg Quinapril 20-40mg Ramipril 10mg Trandolapril Trandolapril 0.5mg d Enalapril 5mg Quinapril 5mg Ramipril 1.25mg Trandolapril 1mg daily Enalapril 10mg Quinapril 10mg Ramipril 2.5mg Trandolapril 2mg daily Enalapril 20mg Quinapril 20mg Ramipril 5mg Trandolapril 4mg daily Enalapril 40mg Quinapril 40mg Ramipril 10mg There are few studies comparing equivalent doses of ACE-inhibitors, for specific indications. Therefore, the above recommendations are based on clinical experiences and are not specific for any indication. -

ACE2 As a Therapeutic Target for COVID-19; Its Role in Infectious Processes and Regulation by Modulators of the RAAS System
Journal of Clinical Medicine Review ACE2 as a Therapeutic Target for COVID-19; Its Role in Infectious Processes and Regulation by Modulators of the RAAS System Veronique Michaud 1,2, Malavika Deodhar 1, Meghan Arwood 1, Sweilem B Al Rihani 1, Pamela Dow 1 and Jacques Turgeon 1,2,* 1 Tabula Rasa HealthCare Precision Pharmacotherapy Research & Development Institute, Orlando, FL 32827, USA; [email protected] (V.M.); [email protected] (M.D.); [email protected] (M.A.); [email protected] (S.B.A.R.); [email protected] (P.D.) 2 Faculty of Pharmacy, Université de Montréal, Montreal, QC H3C 3J7, Canada * Correspondence: [email protected]; Tel.: +856-938-8793 Received: 11 June 2020; Accepted: 2 July 2020; Published: 3 July 2020 Abstract: Angiotensin converting enzyme 2 (ACE2) is the recognized host cell receptor responsible for mediating infection by severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2). ACE2 bound to tissue facilitates infectivity of SARS-CoV-2; thus, one could argue that decreasing ACE2 tissue expression would be beneficial. However, ACE2 catalytic activity towards angiotensin I (Ang I) and II (Ang II) mitigates deleterious effects associated with activation of the renin-angiotensin-aldosterone system (RAAS) on several organs, including a pro-inflammatory status. At the tissue level, SARS-CoV-2 (a) binds to ACE2, leading to its internalization, and (b) favors ACE2 cleavage to form soluble ACE2: these actions result in decreased ACE2 tissue levels. Preserving tissue ACE2 activity while preventing ACE2 shredding is expected to circumvent unrestrained inflammatory response. Concerns have been raised around RAAS modulators and their effects on ACE2 expression or catalytic activity. -

Fosinopril Winthrop 10 / 20Mg Tablets 01/2008
Product Information Page 1 of 19 Fosinopril Winthrop 10 / 20mg tablets 01/2008 PRODUCT INFORMATION ® FOSINOPRIL WINTHROPP 10 / 20 MG TABLETS NAME OF THE MEDICINE Fosinopril sodium Chemical name: (4S)-4-cyclohexyl-1-[[(R)-[(1S)-2-methyl-1-(1- oxopropoxy)propoxy](4-phenylbutyl)phosphinyl]acetyl]-L-proline sodium salt. Molecular formula: C30H45NNaO7P. MW: 585.65. CAS: 88889-14-9. DESCRIPTION Fosinopril sodium is the sodium salt of fosinopril, the ester prodrug of a long acting angiotensin converting enzyme (ACE) inhibitor, fosinopril diacid. Fosinopril is a subclass of ACE inhibitors. It contains a phosphinate group which makes it different from other marketed ACE inhibitors. Fosinopril sodium is a white to off white crystalline powder. It is soluble in water (100 mg/mL), methanol and ethanol, and slightly soluble in hexane. Inactive ingredients: lactose anhydrous, carnauba wax, crospovidone, microcrystalline cellulose, zinc stearate Actions ACE inhibitor Product Information Page 2 of 19 Fosinopril Winthrop 10 / 20mg tablets 01/2008 PHARMACOLOGY Pharmacodynamics Following absorption, in humans and animals, fosinopril sodium is hydrolysed to the pharmacologically active fosinopril diacid, a specific competitive inhibitor of angiotensin converting enzyme (ACE). ACE, a peptidyl dipeptidase, catalyses the conversion of the decapeptide angiotensin I to the octapeptide angiotensin II. Angiotensin II is a potent vasoconstrictor and it also stimulates aldosterone secretion by the adrenal cortex, thereby contributing to sodium and fluid retention. The effects of fosinopril in hypertension appear to result primarily from inhibition of angiotensin II formation and decreased aldosterone secretion. Inhibition of ACE activity leads to decreased levels of angiotensin II, thereby resulting in diminished vasoconstriction, aldosterone secretion, peripheral vascular resistance and sodium and fluid retention. -

Medicines for Cardiovascular Health
Appendix Appendix tables Table A1: Supply of cardiovascular medicines in selected OECD countries, 2004 Medicine class Country ARA CCB BBA DIU AHYP ART GLY CHOL DDD/1,000 population/day Australia 146.9 77.0 25.5 39.4 5.2 2.2 5.3 161.8 Belgium 91.3 40.4 66.6 43.7 5.4 8.0 5.0 104.1 Czech Republic 105.1 69.9 71.3 95.5 12.2 5.6 7.1 65.0 Denmark(a) 85.1 46.2 29.9 111.5 2.7 1.6 6.3 67.2 Finland(a) 125.3 48.3 68.1 61.9 1.7 1.8 6.6 81.7 Germany(b) 143.8 48.8 67.6 65.9 11.6 2.3 10.1 57.3 Greece 149.4 68.6 30.7 35.7 6.7 5.5 9.9 84.0 Iceland 80.9 31.5 46.4 67.3 1.4 3.3 3.2 75.2 Norway(a) 99.1 46.6 39.4 45.4 n/a 1.3 4.4 110.0 Portugal(c) 109.7 37.0 18.3 42.4 0.3 7.3 6.8 71.1 Slovak Republic 88.8 63.3 42.8 31.7 10.7 4.3 6.6 31.5 Sweden(a) 85.6 40.7 54.7 87.3 1.9 1.1 6.5 75.2 ARA agents acting on renin–angiotensin system CCB calcium-channel blockers BBA beta-blocking agents DIU diuretics AHYP antihypertensives ART antiarrhythmics GLY cardiac glycosides CHOL serum-lipid-reducing agents n/a not available (a) Data for Denmark, Finland, Norway and Sweden cover supply of medicines in the community and in hospitals. -

Comparative Proteinuria Management of Different Angiotensin-Converting Enzyme Inhibitors Or Angiotensin Receptor Blockers for No
Comparative proteinuria management of different angiotensin-converting enzyme inhibitors or angiotensin receptor blockers for normotensive patients with CKD: a Bayesian network meta-analysis Huizhen Ye*, Zhihao Huo*, Peiyi Ye, Guanqing Xiao, Zhe Zhang, Chao Xie and Yaozhong Kong Nephrology Department, The First People's Foshan Hospital, Foshan, Guangdong, China * These authors contributed equally to this work. ABSTRACT Background. Both angiotensin-converting enzyme inhibitors (ACEIs) and angiotensin receptor blockers (ARBs) are blood pressure-lowering agents, but they are also being used to control proteinuria in early chronic kidney disease (CKD) patients. However, clinically, some patients present merely proteinuria without hypertension. No guidelines pointed out how to select treatments for proteinuria in normotensive patients. Thus, we conducted a Bayesian network analysis to evaluate the relative effects of different kinds of ACEI or ARB or their combination on proteinuria and blood pressure reduction. Methods. The protocol was registered in PROSPERO with ID CRD42017073721. A comprehensive literature database query was carried out systematically according to PICOS strategies. The primary outcome was reduction in proteinuria, and the secondary outcomes were eGFR reduction and blood pressure reduction. Random- effects pairwise and Bayesian network meta-analyses were used to estimate the effect of different regimens. Results. A total of 14 RCTs with 1,098 patients were included in the analysis. All Submitted 2 September 2019 treatment strategies of ACEI, ARB or their combination had significantly greater efficacy Accepted 16 January 2020 Published 12 March 2020 in reducing proteinuria than placebo in normotensive CKD patients. The combination therapy of olmesartan+temocapril had the highest probability (22%) of being the most Corresponding author Yaozhong Kong, [email protected] effective treatment to reduce proteinuria in normotensive CKD patients. -

Candesartan Cilexetil – Atacand , Astra Merck Development And
Candesartan cilexetil – AtacandÒ, Astra Merck Development and Pharmacology:1-3 The renin-angiotensin-aldosterone system (RAAS) plays a critical role in cardiovascular and renal function and therefore in the regulation of blood pressure. Renin, a proteinase enzyme, is secreted by the kidney in response to a reduction in renal blood flow, blood pressure or sodium concentration. Renin converts angiotensinogen, which is secreted by the liver, to the decapeptide angiotensin I (AI). AI is cleaved by angiotensin converting enzyme (ACE) to the octapeptide angiotensin II (AII). AII produces potent vasoconstriction via interaction with vascular angiotensin receptors (AT1 receptors). AII also promotes aldosterone secretion and therefore sodium retention by stimulation of AT1 receptors present on the adrenal cortex. These actions result in elevated blood pressure secondary to the vasoconstriction and enhanced cardiac output secondary to sodium retention. In addition to its normal regulatory role, the RAAS also contributes to pathological conditions such as renovascular hypertension, essential hypertension and congestive heart failure. Over the past several decades research efforts have been directed toward developing drugs capable of suppressing of RAAS by inhibiting renin release, blocking the formation of AII via inhibition of ACE, or antagonism of AII at its physiologic receptors. These efforts led first to the introduction of clinically effective ACE inhibitors (ACEIs), beginning with captopril in 1981 and followed by enalapril (Vasotec), lisinopril (Prinivil, Zestril), benazepril (Lotensin), fosinopril (Monopril), quinapril (Accupril), ramipril (Altace), moexipril (Univasc) and trandolapril (Mavik). ACEIs have been successfully employed in the management of various forms of hypertension as well as congestive heart failure. However, ACE has other physiologic actions not related to the regulation of RAAS, including the degradation of bradykinin and other peptides including substance P. -

Fosinopril Sodium 10 and 20 Mg Tablets Module 1.3 Product
Fosinopril sodium 10 and 20 mg tablets Module 1.3 Product Information Version: 2014-10-14 Module 1.3.1 Summary of Product Characteristics Replaces : 2013-12-05 SUMMARY OF PRODUCT CHARACTERISTICS 1. NAME OF THE MEDICINAL PRODUCT [Fosinopril sodium 10 mg, tablets] [Fosinopril sodium 20 mg, tablets] 2. QUALITATIVE AND QUANTITATIVE COMPOSITION Each tablet contains 10 or 20 mg fosinopril sodium. Excipient with known effect: Each tablet fosinopril sodium 10 mg contains 87 mg of lactose, anhydrous. Each tablet fosinopril sodium 20 mg contains 174 mg of lactose, anhydrous. For the full list of excipients, see section 6.1. 3. PHARMACEUTICAL FORM Tablet. The 10 mg tablets are white and shaped like a capsule with indents. On one side they are engraved with the letters “APO” and on the other side with “FOS-10”. The 20 mg tablets are white and their shape is oval. On one side they are engraved with the letters “APO” and on the other side with “FOS-20”. 4. CLINICAL PARTICULARS 4.1 Therapeutic indications - Treatment of hypertension. - Treatment of symptomatic heart failure. 4.2 Posology and method of administration Posology Fosinopril sodium should be administered orally in a single daily dose. As with all other medicinal products taken once daily, it should be taken at approximately the same time each day. The absorption of fosinopril sodium is not affected by food. The dose should be individualised according to patient profile and blood pressure response (see section 4.4). Hypertension: Fosinopril sodium may be used as a monotherapy or in combination with other classes of antihypertensive medicinal products (see section 4.3, 4.4, 4.5 and 5.1).