Polymorphisms of Cell Cycle Control Genes Influence the Development Of
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

CDKN2A and CDKN2B Methylation in Coronary Heart Disease Cases and Controls
EXPERIMENTAL AND THERAPEUTIC MEDICINE 14: 6093-6098, 2016 CDKN2A and CDKN2B methylation in coronary heart disease cases and controls JINYAN ZHONG1,2*, XIAOYING CHEN3*, HUADAN YE3, NAN WU1,3, XIAOMIN CHEN1 and SHIWEI DUAN3 1Cardiology Center, Ningbo First Hospital, Ningbo University; 2Department of Cardiology, Ningbo Second Hospital, Ningbo, Zhejiang 315010; 3Medical Genetics Center, School of Medicine, Ningbo University, Ningbo, Zhejiang 315211, P.R. China Received May 27, 2016; Accepted March 24, 2017 DOI: 10.3892/etm.2017.5310 Abstract. The aim of the present study was to investigate frequencies were significantly associated with age, and there the association between cyclin-dependent kinase inhibitor was a gender dimorphism in CDKN2B methylation. 2A (CDKN2A) and cyclin-dependent kinase inhibitor 2B (CDKN2B) methylation, and coronary heart disease (CHD), Introduction and to explore the interaction between methylation status and CHD clinical characteristics in Han Chinese patients. A total Coronary heart disease (CHD) is a complex chronic disease of 189 CHD (96 males, 93 females) and 190 well-matched that is caused by an imbalance between blood supply and non-CHD controls (96 males, 94 females) were recruited for demand in myocardium. Various environmental and genetic the study. Methylation-specific polymerase chain reaction factors are known to contribute to onset and development of technology was used to examine gene promoter methylation CHD (1). As of 2010, CHD was the leading cause of mortality status. Comparisons of methylation frequencies between CHD globally, resulting in over 7 million cases of mortality (2). and non-CHD patients were carried out using the Chi-square Therefore, association studies for CHD biomarkers have been test. -

Epigenetic Silencing of CDKN1A and CDKN2B by SNHG1 Promotes The
Li et al. Cell Death and Disease (2020) 11:823 https://doi.org/10.1038/s41419-020-03031-6 Cell Death & Disease ARTICLE Open Access Epigenetic silencing of CDKN1A and CDKN2B by SNHG1 promotes the cell cycle, migration and epithelial-mesenchymal transition progression of hepatocellular carcinoma Bei Li1,AngLi2, Zhen You 1,JingchangXu1 and Sha Zhu3 Abstract Enhanced SNHG1 (small nucleolar RNA host gene 1) expression has been found to play a critical role in the initiation and progression of hepatocellular carcinoma (HCC) with its detailed mechanism largely unknown. In this study, we show that SNHG1 promotes the HCC progression through epigenetically silencing CDKN1A and CDKN2B in the nucleus, and competing with CDK4 mRNA for binding miR-140-5p in the cytoplasm. Using bioinformatics analyses, we found hepatocarcinogenesis is particularly associated with dysregulated expression of SNHG1 and activation of the cell cycle pathway. SNHG1 was upregulated in HCC tissues and cells, and its knockdown significantly inhibited HCC cell cycle, growth, metastasis, and epithelial–mesenchymal transition (EMT) both in vitro and in vivo. Chromatin immunoprecipitation and RNA immunoprecipitation assays demonstrate that SNHG1 inhibit the transcription of CDKN1A and CDKN2B through enhancing EZH2 mediated-H3K27me3 in the promoter of CDKN1A and CDKN2B, thus resulting in the de-repression of the cell cycle. Dual-luciferase assay and RNA pulldown revealed that SNHG1 promotes 1234567890():,; 1234567890():,; 1234567890():,; 1234567890():,; the expression of CDK4 by competitively binding to miR-140-5p. In conclusion, we propose that SNHG1 formed a regulatory network to confer an oncogenic function in HCC and SNHG1 may serve as a potential target for HCC diagnosis and treatment. -

Expression Profiling of KLF4
Expression Profiling of KLF4 AJCR0000006 Supplemental Data Figure S1. Snapshot of enriched gene sets identified by GSEA in Klf4-null MEFs. Figure S2. Snapshot of enriched gene sets identified by GSEA in wild type MEFs. 98 Am J Cancer Res 2011;1(1):85-97 Table S1: Functional Annotation Clustering of Genes Up-Regulated in Klf4 -Null MEFs ILLUMINA_ID Gene Symbol Gene Name (Description) P -value Fold-Change Cell Cycle 8.00E-03 ILMN_1217331 Mcm6 MINICHROMOSOME MAINTENANCE DEFICIENT 6 40.36 ILMN_2723931 E2f6 E2F TRANSCRIPTION FACTOR 6 26.8 ILMN_2724570 Mapk12 MITOGEN-ACTIVATED PROTEIN KINASE 12 22.19 ILMN_1218470 Cdk2 CYCLIN-DEPENDENT KINASE 2 9.32 ILMN_1234909 Tipin TIMELESS INTERACTING PROTEIN 5.3 ILMN_1212692 Mapk13 SAPK/ERK/KINASE 4 4.96 ILMN_2666690 Cul7 CULLIN 7 2.23 ILMN_2681776 Mapk6 MITOGEN ACTIVATED PROTEIN KINASE 4 2.11 ILMN_2652909 Ddit3 DNA-DAMAGE INDUCIBLE TRANSCRIPT 3 2.07 ILMN_2742152 Gadd45a GROWTH ARREST AND DNA-DAMAGE-INDUCIBLE 45 ALPHA 1.92 ILMN_1212787 Pttg1 PITUITARY TUMOR-TRANSFORMING 1 1.8 ILMN_1216721 Cdk5 CYCLIN-DEPENDENT KINASE 5 1.78 ILMN_1227009 Gas2l1 GROWTH ARREST-SPECIFIC 2 LIKE 1 1.74 ILMN_2663009 Rassf5 RAS ASSOCIATION (RALGDS/AF-6) DOMAIN FAMILY 5 1.64 ILMN_1220454 Anapc13 ANAPHASE PROMOTING COMPLEX SUBUNIT 13 1.61 ILMN_1216213 Incenp INNER CENTROMERE PROTEIN 1.56 ILMN_1256301 Rcc2 REGULATOR OF CHROMOSOME CONDENSATION 2 1.53 Extracellular Matrix 5.80E-06 ILMN_2735184 Col18a1 PROCOLLAGEN, TYPE XVIII, ALPHA 1 51.5 ILMN_1223997 Crtap CARTILAGE ASSOCIATED PROTEIN 32.74 ILMN_2753809 Mmp3 MATRIX METALLOPEPTIDASE -

Infrequent Methylation of CDKN2A(Mts1p16) and Rare Mutation of Both CDKN2A and CDKN2B(Mts2ip15) in Primary Astrocytic Tumours
British Joumal of Cancer (1997) 75(1), 2-8 © 1997 Cancer Research Campaign Infrequent methylation of CDKN2A(MTS1p16) and rare mutation of both CDKN2A and CDKN2B(MTS2Ip15) in primary astrocytic tumours EE Schmidt, K Ichimura, KR Messerle, HM Goike and VP Collins Institute for Oncology and Pathology, Division of Tumour Pathology, and Ludwig Institute for Cancer Research, Stockholm Branch, Karolinska Hospital, S-171 76 Stockholm, Sweden Summary In a series of 46 glioblastomas, 16 anaplastic astrocytomas and eight astrocytomas, all tumours retaining one or both alleles of CDKN2A (48 tumours) and CDKN2B (49 tumours) were subjected to sequence analysis (entire coding region and splice acceptor and donor sites). One glioblastoma with hemizygous deletion of CDKN2A showed a missense mutation in exon 2 (codon 83) that would result in the substitution of tyrosine for histidine in the protein. None of the tumours retaining alleles of CDKN2B showed mutations of this gene. Glioblastomas with retention of both alleles of CDKN2A (14 tumours) and CDKN2B (16 tumours) expressed transcripts for these genes. In contrast, 7/13 glioblastomas with hemizygous deletions of CDKN2A and 8/11 glioblastomas with hemizygous deletions of CDKN2B showed no or weak expression. Anaplastic astrocytomas and astrocytomas showed a considerable variation in the expression of both genes, regardless of whether they retained one or two copies of the genes. The methylation status of the 5' CpG island of the CDKN2A gene was studied in all 15 tumours retaining only one allele of CDKN2A as well as in the six tumours showing no significant expression of transcript despite their retaining both CDKN2A alleles. -

Charles M. Perou, Phd Associate Professor Departments of Genetics
Charles M. Perou, PhD Associate Professor Departments of Genetics and Pathology Carolina Center for Genome Sciences Lineberger Comprehensive Cancer Center University of North Carolina at Chapel Hill Identification of GBM Subtypes using Gene Expression Profiling Agilent Custom Affymetrix Human Affymetrix HT-HG- 244k Exon 1.0 Array U133A Array Microarray Identify Samples and Genes represented on all 3 platforms Factor Analysis Single unified gene expression measurement for each gene 202 patients 11,681 genes Identification of GBM Subtypes Census Clustering of the 202 samples X 1740 Unsupervised clustering of 1740 genes suggests that 4 subtypes of GBM exist variably expressed genes selected using a unified gene expression measure across 3 expression platforms Core TCGA Samples (173) Gene Ontology/Pathway: with Subtype-defining genes • ProNeural: 1. nervous system development ProNeural Normal-like EGFR Mesenchymal 2. neuron differentiation (SOXs) 3. cell cycle = proliferation 4. cell adhesion molecules 5. ErbB signaling pathway FBXO3 GABRB2 • Normal‐like: SNCG NTSR2 1. nucleotide metabolic process MBP 2. neurological system process DLL3 3. axon NKX2-2, NRXN1, NRXN2 TOP2B, CDC7, MYB 4. neuron projection SOX2, SOX4, SOX10,SOX11 5. synaptic transmission ERBB3, PAK3, PAK7 NCAM1 OLIG2 • EGFR: 1. regulation of transcription 2. cell migration 3. nervous system development FGFR3 4. cell proliferation PDGFRA 5. metal ion binding EGFR AKT2 GLI2 TGFB3 • Mesenchymal: CASP8, CASP5, CASP4, CASP1 1. immune response COL8A2, COL5A1, COL1A1,COL1A2 2. receptor activity ILR4 CHI3L1 3. wound healing TRADD TLR2, TLR4, 4. cytokine and chemokine IGFBI mediated signaling pathway RELB 5. NF‐B Signaling Pathway Correlations between gene expression subtypes and clinical parameters Survival Analysis of Subtypes n=196 Subtypes are correlated with: 1. -

A Variant at 9P21.3 Functionally Implicates CDKN2B in Paediatric B-Cell Precursor Acute Lymphoblastic Leukaemia Aetiology
UCSF UC San Francisco Previously Published Works Title A variant at 9p21.3 functionally implicates CDKN2B in paediatric B-cell precursor acute lymphoblastic leukaemia aetiology. Permalink https://escholarship.org/uc/item/3bz9m1pp Journal Nature communications, 7(1) ISSN 2041-1723 Authors Hungate, Eric A Vora, Sapana R Gamazon, Eric R et al. Publication Date 2016-02-12 DOI 10.1038/ncomms10635 Peer reviewed eScholarship.org Powered by the California Digital Library University of California ARTICLE Received 23 Apr 2015 | Accepted 7 Jan 2016 | Published 12 Feb 2016 DOI: 10.1038/ncomms10635 OPEN A variant at 9p21.3 functionally implicates CDKN2B in paediatric B-cell precursor acute lymphoblastic leukaemia aetiology Eric A. Hungate1,*, Sapana R. Vora2,*, Eric R. Gamazon3,4, Takaya Moriyama5, Timothy Best2, Imge Hulur6, Younghee Lee3, Tiffany-Jane Evans7, Eva Ellinghaus8, Martin Stanulla9,Je´remie Rudant10,11, Laurent Orsi10,11, Jacqueline Clavel10,11,12, Elizabeth Milne13, Rodney J. Scott7,14, Ching-Hon Pui15, Nancy J. Cox3, Mignon L. Loh16, Jun J. Yang5, Andrew D. Skol1 & Kenan Onel1 Paediatric B-cell precursor acute lymphoblastic leukaemia (BCP-ALL) is the most common cancer of childhood, yet little is known about BCP-ALL predisposition. In this study, in 2,187 cases of European ancestry and 5,543 controls, we discover and replicate a locus indexed by À 15 rs77728904 at 9p21.3 associated with BCP-ALL susceptibility (Pcombined ¼ 3.32 Â 10 , OR ¼ 1.72) and independent from rs3731217, the previously reported ALL-associated variant in this region. Of correlated SNPs tagged by this locus, only rs662463 is significant in African Americans, suggesting it is a plausible causative variant. -
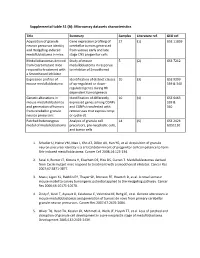
Supplemental Table S1 (A): Microarray Datasets Characteristics
Supplemental table S1 (A): Microarray datasets characteristics Title Summary Samples Literature ref. GEO ref. Acquisition of granule Gene expression profiling of 27 (1) GSE 11859 neuron precursor identity cerebellar tumors generated and Hedgehog‐induced from various early and late medulloblastoma in mice. stage CNS progenitor cells Medulloblastomas derived Study of mouse 5 (2) GSE 7212 from Cxcr6 mutant mice medulloblastoma in response respond to treatment with to inhibitor of Smoothened a Smoothened inhibitor Expression profiles of Identification of distinct classes 10 (3) GSE 9299 mouse medulloblastoma of up‐regulated or down‐ 339 & 340 regulated genes during Hh dependent tumorigenesis Genetic alterations in Identification of differently 10 (4) GSE 6463 mouse medulloblastomas expressed genes among CGNPs 339 & and generation of tumors and CGNPs transfected with 340 from cerebellar granule retroviruses that express nmyc neuron precursors or cyclin‐d1 Patched heterozygous Analysis of granule cell 14 (5) GSE 2426 model of medulloblastoma precursors, pre‐neoplastic cells, GDS1110 and tumor cells 1. Schuller U, Heine VM, Mao J, Kho AT, Dillon AK, Han YG, et al. Acquisition of granule neuron precursor identity is a critical determinant of progenitor cell competence to form Shh‐induced medulloblastoma. Cancer Cell 2008;14:123‐134. 2. Sasai K, Romer JT, Kimura H, Eberhart DE, Rice DS, Curran T. Medulloblastomas derived from Cxcr6 mutant mice respond to treatment with a smoothened inhibitor. Cancer Res 2007;67:3871‐3877. 3. Mao J, Ligon KL, Rakhlin EY, Thayer SP, Bronson RT, Rowitch D, et al. A novel somatic mouse model to survey tumorigenic potential applied to the Hedgehog pathway. Cancer Res 2006;66:10171‐10178. -

Deregulation of the P14arf/MDM2/P53 Pathway Is A
[CANCER RESEARCH 60, 417–424, January 15, 2000] Deregulation of the p14ARF/MDM2/p53 Pathway Is a Prerequisite for Human 1 Astrocytic Gliomas with G1-S Transition Control Gene Abnormalities Koichi Ichimura, Maria Bondesson Bolin,2 Helena M. Goike, Esther E. Schmidt, Ahmad Moshref, and V. Peter Collins3 Department of Pathology, Division of Molecular Histopathology, University of Cambridge, Addenbrooke’s Hospital, Cambridge CB2 2QQ, United Kingdom [K. I., E. E. S., V. P. C], and Ludwig Institute for Cancer Research and Unit of Tumorpathology, Department of Oncology and Pathology, Karolinska Hospital, 171 76 Stockholm, Sweden [K. I., M. B. B., H. M. G., E. E. S., A. M., V. P. C.] ABSTRACT There are several lines of evidence to indicate that deregulation of G1-S transition control alone may be detrimental for cell survival: (a) Deregulation of G -S transition control in cell cycle is one of the 1 transfection of rodent cells with the adenoviral E1A and E1B genes important mechanisms in the development of human tumors including resulted in transformation due to the viral proteins binding to and astrocytic gliomas. We have previously reported that approximately two- inactivating pRB and p53. Transfection with E1A alone induced p53 thirds of glioblastomas (GBs) had abnormalities of G1-S transition control either by mutation/homozygous deletion of RB1 or CDKN2A (p16INK4A), dependent apoptosis (10); (b) when E2F1 was ectopically expressed in or amplification of CDK4 (K. Ichimura et al., Oncogene, 13: 1065–1072, rodent embryo fibroblasts, although the cell indeed entered S phase 1996). However, abnormalities of G1-S transition control genes may in- (11), apoptosis was induced in a p53-dependent manner (12); (c) loss duce p53-dependent apoptosis in cells. -

Tagging Single Nucleotide Polymorphisms in Cell Cycle Control Genes and Susceptibility to Invasive Epithelial Ovarian Cancer
Research Article Tagging Single Nucleotide Polymorphisms in Cell Cycle Control Genes and Susceptibility to Invasive Epithelial Ovarian Cancer Simon A. Gayther,1 Honglin Song,2 Susan J. Ramus,1 Susan Kru¨ger Kjaer,4 Alice S. Whittemore,5 Lydia Quaye,1 Jonathan Tyrer,2 Danielle Shadforth,2 Estrid Hogdall,4 Claus Hogdall,6 Jan Blaeker,7 Richard DiCioccio,8 Valerie McGuire,5 Penelope M. Webb,9 Jonathan Beesley,9 Adele C. Green,9 David C. Whiteman,9 The Australian Ovarian Cancer Study Group,9 The Australian Cancer Study (Ovarian Cancer),20 Marc T. Goodman,10 Galina Lurie,10 Michael E. Carney,10 Francesmary Modugno,10 Roberta B. Ness,11 Robert P. Edwards,12 Kirsten B. Moysich,7 Ellen L. Goode,13 Fergus J. Couch,13 Julie M. Cunningham,13 Thomas A. Sellers,14 Anna H. Wu,15 Malcolm C. Pike,15 Edwin S. Iversen,16 Jeffrey R. Marks,16 Montserrat Garcia-Closas,17 Louise Brinton,17 Jolanta Lissowska,18 Beata Peplonska,19 Douglas F. Easton,3 Ian Jacobs,1 Bruce A.J. Ponder,2 Joellen Schildkraut,16 C. Leigh Pearce,15 Georgia Chenevix-Trench,9 Andrew Berchuck,16 Paul D.P. Pharoah,2 and on behalf of the Ovarian Cancer Association Consortium 1Translational Research Laboratories, University College London, London, United Kingdom; 2Cancer Research United Kingdom Department of Oncology and 3Cancer Research United Kingdom Genetic Epidemiology Unit, University of Cambridge, Strangeways Research Laboratory, Cambridge, United Kingdom; 4Danish Cancer Society, Copenhagen, Denmark; 5Stanford University School of Medicine, Stanford, California; 6University of Copenhagen, -

CDKN2C-Null Leiomyosarcoma: a Novel
original reports CDKN2C-Null Leiomyosarcoma: A Novel, Genomically Distinct Class of TP53/ RB1–Wild-Type Tumor With Frequent CIC Genomic Alterations and 1p/19q-Codeletion Erik A. Williams, MD1; Radwa Sharaf, PhD1; Brennan Decker, MD, PhD2; Adrienne J. Werth, MD3; Helen Toma, MD3; Meagan Montesion, PhD1; Ethan S. Sokol, PhD1; Dean C. Pavlick, BS1; Nikunj Shah, BS1; Kevin Jon Williams, MD4; Jeffrey M. Venstrom, MD1; Brian M. Alexander, MD, MPH1; Jeffrey S. Ross, MD1,5; Lee A. Albacker, PhD1; Douglas I. Lin, MD, PhD1; Shakti H. Ramkissoon, MD, PhD1,6; and Julia A. Elvin, MD, PhD1 abstract PURPOSE Leiomyosarcoma (LMS) harbors frequent mutations in TP53 and RB1 but few actionable genomic alterations. Here, we searched for recurrent actionable genomic alterations in LMS that occur in the absence of common untreatable oncogenic drivers. METHODS Tissues from 276,645 unique advanced cancers, including 2,570 uterine and soft tissue LMS, were sequenced by hybrid-capture–based next-generation DNA and RNA sequencing/comprehensive genomic profiling of up to 406 genes. We characterized clinicopathologic features of relevant patient cases. RESULTS Overall, 77 LMS exhibited homozygous copy loss of CDKN2C at chromosome 1p32.3 (3.0% of LMS). Genomic alterations (GAs) in TP53, RB1, and ATRX were rare compared with the remainder of the LMS cohort (11.7% v 73.4%, 0% v 54.5%, 2.6% v 24.5%, respectively; all P , .0001). CDKN2C-null LMS patient cases were significantly enriched for GAs in CIC (40.3% v 1.4%) at 19q13.2, CDKN2A (46.8% v 7.0%), and RAD51B (16.9% v 1.7%; all P , .0001). -

Co-Deletion of P18 and P16 /P14 /P15 in Glioblastoma Multiforme
Review Conspirators in a Capital Crime: Co-deletion of p18INK4c and p16INK4a/p14ARF/p15INK4b in Glioblastoma Multiforme David A. Solomon,1,2 Jung-Sik Kim,2 Walter Jean,3 and Todd Waldman2 1Tumor Biology Training Program, 2Department of Oncology, Lombardi Comprehensive Cancer Center, and 3Department of Neurosurgery, Georgetown University School of Medicine, Washington, District of Columbia Abstract and showing that p18INK4c-deficient mice are tumor prone, these INK4c Glioblastoma multiforme (GBM) is one of the most dreaded recent studies have suggested that inactivation of p18 may cancer diagnoses due to its poor prognosis and the limited play a perhaps underappreciated role in human cancer patho- INK4a genesis. As such, this review will provide a brief history of the treatment options. Homozygous deletion of the p16 / INK4c p14ARF/p15INK4b locus is among the most common genetic discovery of p18 , summarize the studies that link it to cancers in mice and humans, and provide a forward-looking alterations in GBM. Two recent studies have shown that INK4c deletion and mutation of another INK4 family member, assessment of the role of p18 inactivation in the pathogenesis p18INK4c, also drives the pathogenesis of GBM. This minireview of GBM (Fig. 1). INK4c will discuss the known roles for p18 in the initiation and INK4c progression of cancer and suggest opportunities for future Discovery of p18 studies. [Cancer Res 2008;68(21):8657–60] The human p18INK4c gene was initially discovered in 1994 by Guan and colleagues (8) in a yeast two-hybrid screen to identify Introduction proteins that interacted with human cdk6. The mouse homologue was reported the following year by Hirai and colleagues (9). -

Deletions of CDKN2C in Multiple Myeloma: Biological and Clinical Implications Paola E
Human Cancer Biology Deletions of CDKN2C in Multiple Myeloma: Biological and Clinical Implications Paola E. Leone,1Brian A. Walker,1Matthew W. Jenner,1Laura Chiecchio,2 GianPaolo Dagrada,2 Rebecca K.M. Protheroe,2 David C. Johnson,1Nicholas J. Dickens,1Jose Luis Brito,1Monica Else,1 David Gonzalez,1Fiona M. Ross,2 Selina Chen-Kiang,3 Faith E. Davies,1and Gareth J. Morgan1 Abstract Purpose: Deletions of chromosome 1have been described in 7% to 40% of cases of myeloma with inconsistent clinical consequences. CDKN2C at 1p32.3 has been identified in myeloma cell lines as the potential target of the deletion.We tested the clinical impact of 1p deletion and used high-resolution techniques to define the role of CDKN2C in primary patient material. Experimental Design: We analyzed 515 cases of monoclonal gammopathy of undetermined significance (MGUS), smoldering multiple myeloma (SMM), and newly diagnosed multiple mye- loma using fluorescence in situ hybridization (FISH) for deletions of CDKN2C.In78myeloma cases, we carried out Affymetrix single nucleotide polymorphism mapping and U133 Plus 2.0 ex- pression arrays. In addition, we did mutation, methylation, andWestern blotting analysis. Results: By FISH we identified deletion of 1p32.3 (CDKN2C)in3of66MGUS(4.5%),4of39 SMM (10.3%), and 55 of 369 multiple myeloma cases (15%).We examined the impact of copy number change at CDKN2C on overall survival (OS), and found that the cases with either hemi- zygous or homozygous deletion of CDKN2C had a worse OS compared with cases that were intact at this region (22 months versus 38 months; P = 0.003). Using gene mapping we identified three homozygous deletions at 1p32.3, containing CDKN2C,allofwhichlackedexpressionof CDKN2C.