Complete Exon-Intron Structure of the RPGR-Interacting Protein (RPGRIP1) Gene Allows the Identification of Mutations Underlying Leber Congenital Amaurosis
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

A Multistep Bioinformatic Approach Detects Putative Regulatory
BMC Bioinformatics BioMed Central Research article Open Access A multistep bioinformatic approach detects putative regulatory elements in gene promoters Stefania Bortoluzzi1, Alessandro Coppe1, Andrea Bisognin1, Cinzia Pizzi2 and Gian Antonio Danieli*1 Address: 1Department of Biology, University of Padova – Via Bassi 58/B, 35131, Padova, Italy and 2Department of Information Engineering, University of Padova – Via Gradenigo 6/B, 35131, Padova, Italy Email: Stefania Bortoluzzi - [email protected]; Alessandro Coppe - [email protected]; Andrea Bisognin - [email protected]; Cinzia Pizzi - [email protected]; Gian Antonio Danieli* - [email protected] * Corresponding author Published: 18 May 2005 Received: 12 November 2004 Accepted: 18 May 2005 BMC Bioinformatics 2005, 6:121 doi:10.1186/1471-2105-6-121 This article is available from: http://www.biomedcentral.com/1471-2105/6/121 © 2005 Bortoluzzi et al; licensee BioMed Central Ltd. This is an Open Access article distributed under the terms of the Creative Commons Attribution License (http://creativecommons.org/licenses/by/2.0), which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited. Abstract Background: Searching for approximate patterns in large promoter sequences frequently produces an exceedingly high numbers of results. Our aim was to exploit biological knowledge for definition of a sheltered search space and of appropriate search parameters, in order to develop a method for identification of a tractable number of sequence motifs. Results: Novel software (COOP) was developed for extraction of sequence motifs, based on clustering of exact or approximate patterns according to the frequency of their overlapping occurrences. -

Functional Characterization of the Human RPGR Proximal Promoter
Biochemistry and Molecular Biology Functional Characterization of the Human RPGR Proximal Promoter Xinhua Shu,*,1,2 Julie R. Simpson,2 Alan W. Hart,2 Zhihong Zeng,3 Sarita Rani Patnaik,1 Philippe Gautier,2 Emma Murdoch,2 Brian Tulloch,2 and Alan F. Wright*,2 PURPOSE. Mutations in the retinitis pigmentosa (RP) GTPase promoter will facilitate understanding of the functional role regulator (RPGR) gene account for more than 70% of X-linked of RPGR in the retina and gene therapy of X-linked RP. (Invest RP cases. This study aims to characterize the proximal Ophthalmol Vis Sci. 2012;53:3951–3958) DOI:10.1167/ promoter region of the human RPGR gene. iovs.11-8811 0 METHODS. The 5 -flanking region (5 kb) of human RPGR was cloned and sequenced. A potential transcription start site and etinitis pigmentosa (RP) is a genetically heterogeneous transcription factor binding motifs were identified by bio- Rgroup of retinal degenerations that affect 1 in 4000 in the informatic analysis. Constructs containing the putative human general population.1,2 Most cases are inherited in an autosomal RPGR promoter region upstream of a luciferase reporter gene dominant, autosomal recessive, X-linked, or mitochondrial were generated and analyzed by transient transfection and manner, but oligogenic inheritance has been established in a luciferase assays. Transgenic mouse lines carrying a 3-kb small proportion of families.3 X-linked RP (XLRP) is one of the human RPGR promoter sequence fused to lacZ were generated most consistently severe forms of RP, with a reported average and RPGR proximal promoter activity was analyzed by X-gal age at onset of 7.2 6 1.7 years.4 XLRP affects 10% to 20% of all staining. -

And Cone-Rod Dystrophy in Dogs
University of Pennsylvania ScholarlyCommons Departmental Papers (Vet) School of Veterinary Medicine 2012 RPGRIP1 and Cone-Rod Dystrophy in Dogs Tatyana N. Kuznetsova Barbara Zangerl University of Pennsylvania, [email protected] Gustavo D. Aguirre University of Pennsylvania, [email protected] Follow this and additional works at: https://repository.upenn.edu/vet_papers Part of the Eye Diseases Commons, Geriatrics Commons, Medical Biotechnology Commons, Medical Genetics Commons, Medical Immunology Commons, Ophthalmology Commons, and the Veterinary Medicine Commons Recommended Citation Kuznetsova, T. N., Zangerl, B., & Aguirre, G. D. (2012). RPGRIP1 and Cone-Rod Dystrophy in Dogs. Retinal Degenerative Diseases: Advances in Experimental Medicine and Biology, 723 321-328. http://dx.doi.org/ 10.1007/978-1-4614-0631-0_42 This paper is posted at ScholarlyCommons. https://repository.upenn.edu/vet_papers/85 For more information, please contact [email protected]. RPGRIP1 and Cone-Rod Dystrophy in Dogs Abstract Cone–rod dystrophies (crd) represent a group of progressive inherited blinding diseases characterized by primary dysfunction and loss of cone photoreceptors accompanying or preceding rod death. Recessive crd type 1 was described in dogs associated with an RPGRIP1 exon 2 mutation, but with lack of complete concordance between genotype and phenotype. This review highlights role of the RPGRIP1, a component of complex protein networks, and its function in the primary cilium, and discusses the potential mechanisms of genotype–phenotype discordance observed in dogs with the RPGRIP1 mutation. Keywords RPGRIP1, polymorphism, cone-rod dystrophy, protein network, photoreceptor cilia Disciplines Eye Diseases | Geriatrics | Medical Biotechnology | Medical Genetics | Medical Immunology | Ophthalmology | Veterinary Medicine This journal article is available at ScholarlyCommons: https://repository.upenn.edu/vet_papers/85 Published in final edited form as: Adv Exp Med Biol. -

Selective Loss of RPGRIP1-Dependent Ciliary Targeting of NPHP4, RPGR and SDCCAG8 Underlies the Degeneration of Photoreceptor Neurons
Citation: Cell Death and Disease (2012) 3, e355; doi:10.1038/cddis.2012.96 & 2012 Macmillan Publishers Limited All rights reserved 2041-4889/12 www.nature.com/cddis Selective loss of RPGRIP1-dependent ciliary targeting of NPHP4, RPGR and SDCCAG8 underlies the degeneration of photoreceptor neurons H Patil1,3, N Tserentsoodol1,3, A Saha1, Y Hao1, M Webb1 and PA Ferreira*,1,2 The retinitis pigmentosa GTPase regulator (RPGR) and nephrocystin-4 (NPHP4) comprise two key partners of the assembly complex of the RPGR-interacting protein 1 (RPGRIP1). Mutations in RPGR and NPHP4 are linked to severe multisystemic diseases with strong retinal involvement of photoreceptor neurons, whereas those in RPGRIP1 cause the fulminant photoreceptor dystrophy, Leber congenital amaurosis (LCA). Further, mutations in Rpgrip1 and Nphp4 suppress the elaboration of the outer segment compartment of photoreceptor neurons by elusive mechanisms, the understanding of which has critical implications in uncovering the pathogenesis of syndromic retinal dystrophies. Here we show RPGRIP1 localizes to the photoreceptor connecting cilium (CC) distally to the centriole/basal body marker, centrin-2 and the ciliary marker, acetylated-a- tubulin. NPHP4 abuts proximally RPGRIP1, RPGR and the serologically defined colon cancer antigen-8 (SDCCAG8), a protein thought to partake in the RPGRIP1 interactome and implicated also in retinal–renal ciliopathies. Ultrastructurally, RPGRIP1 localizes exclusively throughout the photoreceptor CC and Rpgrip1nmf247 photoreceptors present shorter cilia with a ruffled membrane. Strikingly, Rpgrip1nmf247 mice without RPGRIP1 expression lack NPHP4 and RPGR in photoreceptor cilia, whereas the SDCCAG8 and acetylated-a-tubulin ciliary localizations are strongly decreased, even though the NPHP4 and SDCCAG8 expression levels are unaffected and those of acetylated-a-tubulin and c-tubulin are upregulated. -

An RPGRIP1 Mutation
Hindawi Publishing Corporation Stem Cells International Volume 2012, Article ID 685901, 12 pages doi:10.1155/2012/685901 Research Article Assessment of Hereditary Retinal Degeneration in the English Springer Spaniel Dog and Disease Relationship to an RPGRIP1 Mutation Kristina Narfstrom,¨ 1, 2 Manbok Jeong,3 Jennifer Hyman,4 Richard W. Madsen,5 and Tomas F. Bergstrom¨ 6 1 Department of Veterinary Medicine and Surgery, College of Veterinary Medicine, University of Missouri, Columbia, MO 65211, USA 2 Department of Ophthalmology, Mason Eye Institute, University of Missouri, Columbia, MO 65211, USA 3 Department of Veterinary Medicine and Surgery, College of Veterinary Medicine and BK21 Program for Veterinary Science, Seoul National University, Seoul 151-742, Republic of Korea 4 Eye Care For Animals, VA, 20176-3367, USA 5 Biostatistics, School of Medicine, University of Missouri, Columbia, MO 65211, USA 6 Department of Animal Breeding and Genetics, Swedish University of Agricultural Sciences, 750 07 Uppsala, Sweden Correspondence should be addressed to Kristina Narfstrom,¨ [email protected] Received 12 September 2011; Revised 9 November 2011; Accepted 15 November 2011 Academic Editor: Henry J. Klassen Copyright © 2012 Kristina Narfstrom¨ et al. This is an open access article distributed under the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited. Intensive breeding and selection on desired traits have produced high rates of inherited diseases in dogs. Hereditary retinal degeneration, often called progressive retinal atrophy (PRA), is prevalent in dogs with disease entities comparable to human retinitis pigmentosa (RP) and Leber’s congenital amaurosis (LCA). Recent molecular studies in the English Springer Spaniel (ESS) dog have shown that PRA cases are often homozygous for a mutation in the RPGRIP1 gene, the defect also causing human RP, LCA, and cone rod dystrophies. -

In Silico Approach to Calculate the Transcript Capacity
In silico approach to calculate the transcript capacity Young-Sup Lee, Kyung-Hye Won, Jae-Don Oh*, Donghyun Shin** Department of Animal Biotechnology, Chonbuk National University, Jeonju 54896, Korea Original article We sought the novel concept, transcript capacity (TC) and analyzed TC. Our approach to estimate TC was through an in silico method. TC refers to the capacity that a transcript ex- eISSN 2234-0742 erts in a cell as enzyme or protein function after translation. We used the genome-wide Genomics Inform 2019;17(3):e31 association study (GWAS) beta effect and transcription level in RNA-sequencing to esti- https://doi.org/10.5808/GI.2019.17.3.e31 mate TC. The trait was body fat percent and the transcript reads were obtained from the human protein atlas. The assumption was that the GWAS beta effect is the gene’s effect and TC was related to the corresponding gene effect and transcript reads. Further, we sur- Received: September 2, 2019 veyed gene ontology (GO) in the highest TC and the lowest TC genes. The most frequent Revised: September 18, 2019 GOs with the highest TC were neuronal-related and cell projection organization related. Accepted: September 19, 2019 The most frequent GOs with the lowest TC were wound-healing related and embryo devel- opment related. We expect that our analysis contributes to estimating TC in the diverse *Corresponding author: E-mail: [email protected] species and playing a benevolent role to the new bioinformatic analysis. **Corresponding author: Keywords: fat, genome-wide association study, in silico method, transcript capacity, RNA- E-mail: [email protected] seq Introduction There have been various experimental studies regarding enzyme activity [1,2]. -

Contribution of Structural and Intronic Mutations to RPGRIP1
bioRxiv preprint doi: https://doi.org/10.1101/211292; this version posted October 30, 2017. The copyright holder for this preprint (which was not certified by peer review) is the author/funder. All rights reserved. No reuse allowed without permission. Contribution of structural and intronic mutations to RPGRIP1- mediated inherited retinal dystrophies. Farzad Jamshidi1, Emily M. Place1, Daniel Navarro-Gomez1, Mathew Maher1, Elise Valkanas3, Monkol Lek2,3, Daniel MacArthur2,3, Kinga M. Bujakowska1, Eric A. Pierce1 1Ocular Genomics Institute, Department of Ophthalmology, Massachusetts Eye and Ear Infirmary, Harvard Medical School, Boston, MA. 2Analytic and Translational Genetics Unit, Massachusetts General Hospital, Boston, Massachusetts, USA. 3Program in Medical and Population Genetics, Broad Institute of MIT and Harvard, Cambridge, Massachusetts, USA. Correspondence: Eric A. Pierce M.D., Ph.D. Solman and Libe Friedman Professor of Ophthalmology Massachusetts Eye and Ear Infirmary Harvard Medical School 243 Charles Street Boston, MA 02114 Phone: 617-573-6917 [email protected] bioRxiv preprint doi: https://doi.org/10.1101/211292; this version posted October 30, 2017. The copyright holder for this preprint (which was not certified by peer review) is the author/funder. All rights reserved. No reuse allowed without permission. 1. Abstract With the completion of the first phase 3 human gene therapy randomized clinical trial, in the form of voretigene neparvovec for RPE65-mediated inherited retinal dystrophy, as well as the advent of more than 10 other gene therapy trials for inherited retinal disorders, accurate genetic diagnostics will have an increasingly important role in clinical decision- making. Current genetic diagnostic testing panels primarily focus on coding sequences. -

(LCA9) for Leber's Congenital Amaurosis on Chromosome 1P36
European Journal of Human Genetics (2003) 11, 420–423 & 2003 Nature Publishing Group All rights reserved 1018-4813/03 $25.00 www.nature.com/ejhg SHORT REPORT Identification of a locus (LCA9) for Leber’s congenital amaurosis on chromosome 1p36 T Jeffrey Keen1, Moin D Mohamed1, Martin McKibbin2, Yasmin Rashid3, Hussain Jafri3, Irene H Maumenee4 and Chris F Inglehearn*,1 1Molecular Medicine Unit, University of Leeds, Leeds, UK; 2Department of Ophthalmology, St James’s University Hospital, Leeds, UK; 3Department of Obstetrics and Gynaecology, Fatima Jinnah Medical College, Lahore, Pakistan; 4Department of Ophthalmology, Johns Hopkins University School of Medicine, Baltimore, MD, USA Leber’s congenital amaurosis (LCA) is the most common cause of inherited childhood blindness and is characterised by severe retinal degeneration at or shortly after birth. We have identified a new locus, LCA9, on chromosome 1p36, at which the disease segregates in a single consanguineous Pakistani family. Following a whole genome linkage search, an autozygous region of 10 cM was identified between the markers D1S1612 and D1S228. Multipoint linkage analysis generated a lod score of 4.4, strongly supporting linkage to this region. The critical disease interval contains at least 5.7 Mb of DNA and around 50 distinct genes. One of these, retinoid binding protein 7 (RBP7), was screened for mutations in the family, but none was found. European Journal of Human Genetics (2003) 11, 420–423. doi:10.1038/sj.ejhg.5200981 Keywords: Leber’s congenital amaurosis; LCA; LCA9; retina; linkage; 1p36 Introduction guineous pedigrees, known as homozygosity or autozygos- Leber’s congenital amaurosis (LCA) is the name given to a ity mapping, is a powerful approach to identify recessively group of recessively inherited retinal dystrophies repre- inherited disease gene loci.10 Cultural precedents in some senting the most common genetic cause of blindness in Pakistani communities have led to a high frequency of infants and children. -

Platform Abstracts
American Society of Human Genetics 65th Annual Meeting October 6–10, 2015 Baltimore, MD PLATFORM ABSTRACTS Wednesday, October 7, 9:50-10:30am Abstract #’s Friday, October 9, 2:15-4:15 pm: Concurrent Platform Session D: 4. Featured Plenary Abstract Session I Hall F #1-#2 46. Hen’s Teeth? Rare Variants and Common Disease Ballroom I #195-#202 Wednesday, October 7, 2:30-4:30pm Concurrent Platform Session A: 47. The Zen of Gene and Variant 15. Update on Breast and Prostate Assessment Ballroom III #203-#210 Cancer Genetics Ballroom I #3-#10 48. New Genes and Mechanisms in 16. Switching on to Regulatory Variation Ballroom III #11-#18 Developmental Disorders and 17. Shedding Light into the Dark: From Intellectual Disabilities Room 307 #211-#218 Lung Disease to Autoimmune Disease Room 307 #19-#26 49. Statistical Genetics: Networks, 18. Addressing the Difficult Regions of Pathways, and Expression Room 309 #219-#226 the Genome Room 309 #27-#34 50. Going Platinum: Building a Better 19. Statistical Genetics: Complex Genome Room 316 #227-#234 Phenotypes, Complex Solutions Room 316 #35-#42 51. Cancer Genetic Mechanisms Room 318/321 #235-#242 20. Think Globally, Act Locally: Copy 52. Target Practice: Therapy for Genetic Hilton Hotel Number Variation Room 318/321 #43-#50 Diseases Ballroom 1 #243-#250 21. Recent Advances in the Genetic Basis 53. The Real World: Translating Hilton Hotel of Neuromuscular and Other Hilton Hotel Sequencing into the Clinic Ballroom 4 #251-#258 Neurodegenerative Phenotypes Ballroom 1 #51-#58 22. Neuropsychiatric Diseases of Hilton Hotel Friday, October 9, 4:30-6:30pm Concurrent Platform Session E: Childhood Ballroom 4 #59-#66 54. -

Photoreceptor Cilia and Retinal Ciliopathies
Downloaded from http://cshperspectives.cshlp.org/ on September 26, 2021 - Published by Cold Spring Harbor Laboratory Press Photoreceptor Cilia and Retinal Ciliopathies Kinga M. Bujakowska, Qin Liu, and Eric A. Pierce Ocular Genomics Institute, Massachusetts Eye and Ear Infirmary, Department of Ophthalmology, Harvard Medical School, Boston, Massachusetts 02114 Correspondence: [email protected] Photoreceptors are sensory neurons designed to convert light stimuli into neurological re- sponses. This process, called phototransduction, takes place in the outer segments (OS) of rod and cone photoreceptors. OS are specialized sensory cilia, with analogous structures to those present in other nonmotile cilia. Deficient morphogenesis and/or dysfunction of pho- toreceptor sensory cilia (PSC) caused by mutations in a variety of photoreceptor-specific and common cilia genes can lead to inherited retinal degenerations (IRDs). IRDs can manifest as isolated retinal diseases or syndromic diseases. In this review, we describe the structure and composition of PSC and different forms of ciliopathies with retinal involvement. We review the genetics of the IRDs, which are monogenic disorders but genetically diverse with regard to causality. hotoreceptors are sensory neurons designed morphogenesis and/or dysfunction of photore- Pto convert light stimuli into electrical re- ceptor sensory cilia (PSC) caused by mutations sponses, a process called phototransduction. in a variety of photoreceptor-specific and com- Phototransduction takes place in the highly spe- mon cilia genes can lead to a group of clinical cialized compartment of photoreceptors, the manifestations, called inherited retinal degener- outer segment (OS) (Pearring et al. 2013; Mol- ations (IRDs). In this review, we will discuss the day and Moritz 2015). -

Ciliary Genes in Renal Cystic Diseases
cells Review Ciliary Genes in Renal Cystic Diseases Anna Adamiok-Ostrowska * and Agnieszka Piekiełko-Witkowska * Department of Biochemistry and Molecular Biology, Centre of Postgraduate Medical Education, 01-813 Warsaw, Poland * Correspondence: [email protected] (A.A.-O.); [email protected] (A.P.-W.); Tel.: +48-22-569-3810 (A.P.-W.) Received: 3 March 2020; Accepted: 5 April 2020; Published: 8 April 2020 Abstract: Cilia are microtubule-based organelles, protruding from the apical cell surface and anchoring to the cytoskeleton. Primary (nonmotile) cilia of the kidney act as mechanosensors of nephron cells, responding to fluid movements by triggering signal transduction. The impaired functioning of primary cilia leads to formation of cysts which in turn contribute to development of diverse renal diseases, including kidney ciliopathies and renal cancer. Here, we review current knowledge on the role of ciliary genes in kidney ciliopathies and renal cell carcinoma (RCC). Special focus is given on the impact of mutations and altered expression of ciliary genes (e.g., encoding polycystins, nephrocystins, Bardet-Biedl syndrome (BBS) proteins, ALS1, Oral-facial-digital syndrome 1 (OFD1) and others) in polycystic kidney disease and nephronophthisis, as well as rare genetic disorders, including syndromes of Joubert, Meckel-Gruber, Bardet-Biedl, Senior-Loken, Alström, Orofaciodigital syndrome type I and cranioectodermal dysplasia. We also show that RCC and classic kidney ciliopathies share commonly disturbed genes affecting cilia function, including VHL (von Hippel-Lindau tumor suppressor), PKD1 (polycystin 1, transient receptor potential channel interacting) and PKD2 (polycystin 2, transient receptor potential cation channel). Finally, we discuss the significance of ciliary genes as diagnostic and prognostic markers, as well as therapeutic targets in ciliopathies and cancer. -
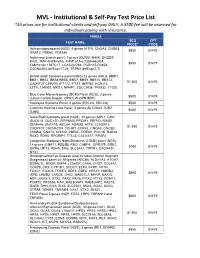
MVL - Institutional & Self-Pay Test Price List *All Prices Are for Institutional Clients and Self-Pay ONLY
MVL - Institutional & Self-Pay Test Price List *All prices are for institutional clients and self-pay ONLY. A $500 fee will be assessed for individuals paying with insurance. PANELS SEQ CPT TEST NAME PRICE* CODE Achromatopsiapanel (NGS), 6 genes (ATF6, CNGA3, CNGB3, $950 81479 GNAT2, PDE6C, PDE6H) Ashkenazi Jewish panel, 7 genes (CLRN1-N48K, DHDDS- K42E, MAK-K429insAlu, FAM161A-c.1355-6delCA, $550 81407 FAM161Ac.1567C>T, LCA5-Q279X, PCDH15-R245X, CACNA2D4-delExon17-26, TRPM1-delExon2-7) Bardet-Biedl Syndrome panel (NGS) 23 genes (ARL6, BBIP1, BBS1, BBS2, BBS4,BBS5, BBS7, BBS9, BBS10, BBS12, $1,500 81479 C8ORF37,CEP290, IFT172, IFT27, INPP5E, KCNJ13, LZTFL1,MKKS, MKS1, NPHP1, SDCCAG8, TRIM32, TTC8) Blue Cone Monochromacy (BCM) Panel (NGS), 2 genes $500 81479 (Locus Control Region, OPN1LW-OPN1MW) Cockayne Sydrome Panel, 2 genes (ERCC6, ERCC8) $500 81479 Common Hearing Loss Panel, 3 genes (SLC26A4, GJB2, $650 81479 GJB6) Cone-Rod Dystrophy panel (NGS), 33 genes (AIPL1, CRX, GUCA1A, GUCY2D, PITPNM3, PROM1, PRPH2, RIMS1, SEMA4A, UNC119, ABCA4, ADAM9, ATF6, C21ORF2, $1,950 81479 C8ORF37,CACNA2D4, CDHR1, CERKL, CNGA3, CNGB3, CNNM4, GNAT2, KCNV2, PDE6C, PDE6H, POC1B, RAB28, RAX2, RDH5, RPGRIP1, TTLL5, CACNA1F, RPGR) Congenital Stationary Night Blindness (CSNB) panel (NGS), 14 genes (GNAT1, PDE6B, RHO, CABP4, GPR179, GRK1, $950 81479 GRM6,LRIT3, RDH5, SAG, SLC24A1, TRPM1, CACNA1F, NYX) Developmental Eye Disease (also includes Anterior Segment Dysgenesis) panel v4: 59 genes (ABCB6, ALDH1A3, ATOH7, B3GALTL, BCOR, BMP4, c12orf57, CASK, CHD7, COL4A1,