Supplemental Data Fig. S1- Related to Fig. 1
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

XPO1E571K Mutation Modifies Exportin 1 Localisation And
cancers Article XPO1E571K Mutation Modifies Exportin 1 Localisation and Interactome in B-Cell Lymphoma Hadjer Miloudi 1, Élodie Bohers 1,2, François Guillonneau 3 , Antoine Taly 4,5 , Vincent Cabaud Gibouin 6,7 , Pierre-Julien Viailly 1,2 , Gaëtan Jego 6,7 , Luca Grumolato 8 , Fabrice Jardin 1,2 and Brigitte Sola 1,* 1 INSERM U1245, Unicaen, Normandie University, F-14000 Caen, France; [email protected] (H.M.); [email protected] (E.B.); [email protected] (P.-J.V.); [email protected] (F.J.) 2 Centre de lutte contre le Cancer Henri Becquerel, F-76000 Rouen, France 3 Plateforme Protéomique 3P5, Université de Paris, Institut Cochin, INSERM, CNRS, F-75014 Paris, France; [email protected] 4 Laboratoire de Biochimie Théorique, CNRS UPR 9030, Université de Paris, F-75005 Paris, France; [email protected] 5 Institut de Biologie Physico-Chimique, Fondation Edmond de Rothschild, PSL Research University, F-75005 Paris, France 6 INSERM, LNC UMR1231, F-21000 Dijon, France; [email protected] (V.C.G.); [email protected] (G.J.) 7 Team HSP-Pathies, University of Burgundy and Franche-Comtée, F-21000 Dijon, France 8 INSERM U1239, Unirouen, Normandie University, F-76130 Mont-Saint-Aignan, France; [email protected] * Correspondence: [email protected]; Tel.: +33-2-3156-8210 Received: 11 September 2020; Accepted: 28 September 2020; Published: 30 September 2020 Simple Summary: Almost 25% of patients with either primary mediastinal B-cell lymphoma (PMBL) or classical Hodgkin lymphoma (cHL) possess a recurrent mutation of the XPO1 gene encoding the major nuclear export protein. -

Targeting the Nuclear Import Receptor Kpnb1 As an Anticancer Therapeutic
Published OnlineFirst February 1, 2016; DOI: 10.1158/1535-7163.MCT-15-0052 Small Molecule Therapeutics Molecular Cancer Therapeutics Targeting the Nuclear Import Receptor Kpnb1as an Anticancer Therapeutic Pauline J. van der Watt1, Alicia Chi1, Tamara Stelma1, Catherine Stowell1, Erin Strydom1, Sarah Carden1, Liselotte Angus1, Kate Hadley1, Dirk Lang2, Wei Wei3, Michael J. Birrer3, John O. Trent4, and Virna D. Leaner1 Abstract Karyopherin beta 1 (Kpnb1) is a nuclear transport receptor tissue origins. Minimum effect on the proliferation of non- that imports cargoes into the nucleus. Recently, elevated Kpnb1 cancer cells was observed at the concentration of INI-43 that expression was found in certain cancers and Kpnb1silencing showed a significant cytotoxic effect on various cervical and with siRNA was shown to induce cancer cell death. This study esophageal cancer cell lines. A rescue experiment confirmed aimed to identify novel small molecule inhibitors of Kpnb1, that INI-43 exerted its cell killing effects, in part, by targeting and determine their anticancer activity. An in silico screen Kpnb1. INI-43 treatment elicited a G2–M cell-cycle arrest in identified molecules that potentially bind Kpnb1 and Inhibitor cancer cells and induced the intrinsic apoptotic pathway. Intra- of Nuclear Import-43, INI-43 (3-(1H-benzimidazol-2-yl)-1-(3- peritoneal administration of INI-43 significantly inhibited the dimethylaminopropyl)pyrrolo[5,4-b]quinoxalin-2-amine) was growth of subcutaneously xenografted esophageal and cervical investigated further as it interfered with the nuclear localization tumor cells. We propose that Kpnb1 inhibitors could have of Kpnb1andknownKpnb1cargoesNFAT,NFkB, AP-1, and therapeutic potential for the treatment of cancer. -

UNIVERSITY of CALIFORNIA, SAN DIEGO Functional Analysis of Sall4
UNIVERSITY OF CALIFORNIA, SAN DIEGO Functional analysis of Sall4 in modulating embryonic stem cell fate A dissertation submitted in partial satisfaction of the requirements for the degree Doctor of Philosophy in Molecular Pathology by Pei Jen A. Lee Committee in charge: Professor Steven Briggs, Chair Professor Geoff Rosenfeld, Co-Chair Professor Alexander Hoffmann Professor Randall Johnson Professor Mark Mercola 2009 Copyright Pei Jen A. Lee, 2009 All rights reserved. The dissertation of Pei Jen A. Lee is approved, and it is acceptable in quality and form for publication on microfilm and electronically: ______________________________________________________________ ______________________________________________________________ ______________________________________________________________ ______________________________________________________________ Co-Chair ______________________________________________________________ Chair University of California, San Diego 2009 iii Dedicated to my parents, my brother ,and my husband for their love and support iv Table of Contents Signature Page……………………………………………………………………….…iii Dedication…...…………………………………………………………………………..iv Table of Contents……………………………………………………………………….v List of Figures…………………………………………………………………………...vi List of Tables………………………………………………….………………………...ix Curriculum vitae…………………………………………………………………………x Acknowledgement………………………………………………….……….……..…...xi Abstract………………………………………………………………..…………….....xiii Chapter 1 Introduction ..…………………………………………………………………………….1 Chapter 2 Materials and Methods……………………………………………………………..…12 -

1 Supporting Information for a Microrna Network Regulates
Supporting Information for A microRNA Network Regulates Expression and Biosynthesis of CFTR and CFTR-ΔF508 Shyam Ramachandrana,b, Philip H. Karpc, Peng Jiangc, Lynda S. Ostedgaardc, Amy E. Walza, John T. Fishere, Shaf Keshavjeeh, Kim A. Lennoxi, Ashley M. Jacobii, Scott D. Rosei, Mark A. Behlkei, Michael J. Welshb,c,d,g, Yi Xingb,c,f, Paul B. McCray Jr.a,b,c Author Affiliations: Department of Pediatricsa, Interdisciplinary Program in Geneticsb, Departments of Internal Medicinec, Molecular Physiology and Biophysicsd, Anatomy and Cell Biologye, Biomedical Engineeringf, Howard Hughes Medical Instituteg, Carver College of Medicine, University of Iowa, Iowa City, IA-52242 Division of Thoracic Surgeryh, Toronto General Hospital, University Health Network, University of Toronto, Toronto, Canada-M5G 2C4 Integrated DNA Technologiesi, Coralville, IA-52241 To whom correspondence should be addressed: Email: [email protected] (M.J.W.); yi- [email protected] (Y.X.); Email: [email protected] (P.B.M.) This PDF file includes: Materials and Methods References Fig. S1. miR-138 regulates SIN3A in a dose-dependent and site-specific manner. Fig. S2. miR-138 regulates endogenous SIN3A protein expression. Fig. S3. miR-138 regulates endogenous CFTR protein expression in Calu-3 cells. Fig. S4. miR-138 regulates endogenous CFTR protein expression in primary human airway epithelia. Fig. S5. miR-138 regulates CFTR expression in HeLa cells. Fig. S6. miR-138 regulates CFTR expression in HEK293T cells. Fig. S7. HeLa cells exhibit CFTR channel activity. Fig. S8. miR-138 improves CFTR processing. Fig. S9. miR-138 improves CFTR-ΔF508 processing. Fig. S10. SIN3A inhibition yields partial rescue of Cl- transport in CF epithelia. -

T-Cell Receptor (TCR) Signaling Promotes the Assembly of Ranbp2
RESEARCH ARTICLE T-cell receptor (TCR) signaling promotes the assembly of RanBP2/RanGAP1- SUMO1/Ubc9 nuclear pore subcomplex via PKC--mediated phosphorylation of RanGAP1 Yujiao He1, Zhiguo Yang1†, Chen-si Zhao1†, Zhihui Xiao1†, Yu Gong1, Yun-Yi Li1, Yiqi Chen1, Yunting Du1, Dianying Feng1, Amnon Altman2, Yingqiu Li1* 1MOE Key Laboratory of Gene Function and Regulation, Guangdong Province Key Laboratory of Pharmaceutical Functional Genes, State Key Laboratory of Biocontrol, School of Life Sciences, Sun Yat-sen University, Guangzhou, China; 2Center for Cancer Immunotherapy, La Jolla Institute for Immunology, La Jolla, United States Abstract The nuclear pore complex (NPC) is the sole and selective gateway for nuclear transport, and its dysfunction has been associated with many diseases. The metazoan NPC subcomplex RanBP2, which consists of RanBP2 (Nup358), RanGAP1-SUMO1, and Ubc9, regulates the assembly and function of the NPC. The roles of immune signaling in regulation of NPC remain poorly understood. Here, we show that in human and murine T cells, following T-cell receptor (TCR) stimulation, protein kinase C-q (PKC-q) directly phosphorylates RanGAP1 to facilitate RanBP2 subcomplex assembly and nuclear import and, thus, the nuclear translocation of AP-1 transcription *For correspondence: factor. Mechanistically, TCR stimulation induces the translocation of activated PKC-q to the NPC, 504 506 [email protected] where it interacts with and phosphorylates RanGAP1 on Ser and Ser . RanGAP1 phosphorylation increases its binding affinity for Ubc9, thereby promoting sumoylation of RanGAP1 †These authors contributed and, finally, assembly of the RanBP2 subcomplex. Our findings reveal an unexpected role of PKC-q equally to this work as a direct regulator of nuclear import and uncover a phosphorylation-dependent sumoylation of Competing interests: The RanGAP1, delineating a novel link between TCR signaling and assembly of the RanBP2 NPC authors declare that no subcomplex. -
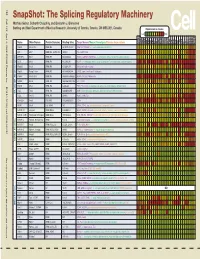
Snapshot: the Splicing Regulatory Machinery Mathieu Gabut, Sidharth Chaudhry, and Benjamin J
192 Cell SnapShot: The Splicing Regulatory Machinery Mathieu Gabut, Sidharth Chaudhry, and Benjamin J. Blencowe 133 Banting and Best Department of Medical Research, University of Toronto, Toronto, ON M5S 3E1, Canada Expression in mouse , April4, 2008©2008Elsevier Inc. Low High Name Other Names Protein Domains Binding Sites Target Genes/Mouse Phenotypes/Disease Associations Amy Ceb Hip Hyp OB Eye SC BM Bo Ht SM Epd Kd Liv Lu Pan Pla Pro Sto Spl Thy Thd Te Ut Ov E6.5 E8.5 E10.5 SRp20 Sfrs3, X16 RRM, RS GCUCCUCUUC SRp20, CT/CGRP; −/− early embryonic lethal E3.5 9G8 Sfrs7 RRM, RS, C2HC Znf (GAC)n Tau, GnRH, 9G8 ASF/SF2 Sfrs1 RRM, RS RGAAGAAC HipK3, CaMKIIδ, HIV RNAs; −/− embryonic lethal, cond. KO cardiomyopathy SC35 Sfrs2 RRM, RS UGCUGUU AChE; −/− embryonic lethal, cond. KO deficient T-cell maturation, cardiomyopathy; LS SRp30c Sfrs9 RRM, RS CUGGAUU Glucocorticoid receptor SRp38 Fusip1, Nssr RRM, RS ACAAAGACAA CREB, type II and type XI collagens SRp40 Sfrs5, HRS RRM, RS AGGAGAAGGGA HipK3, PKCβ-II, Fibronectin SRp55 Sfrs6 RRM, RS GGCAGCACCUG cTnT, CD44 DOI 10.1016/j.cell.2008.03.010 SRp75 Sfrs4 RRM, RS GAAGGA FN1, E1A, CD45; overexpression enhances chondrogenic differentiation Tra2α Tra2a RRM, RS GAAARGARR GnRH; overexpression promotes RA-induced neural differentiation SR and SR-Related Proteins Tra2β Sfrs10 RRM, RS (GAA)n HipK3, SMN, Tau SRm160 Srrm1 RS, PWI AUGAAGAGGA CD44 SWAP Sfrs8 RS, SWAP ND SWAP, CD45, Tau; possible asthma susceptibility gene hnRNP A1 Hnrnpa1 RRM, RGG UAGGGA/U HipK3, SMN2, c-H-ras; rheumatoid arthritis, systemic lupus -

Aneuploidy: Using Genetic Instability to Preserve a Haploid Genome?
Health Science Campus FINAL APPROVAL OF DISSERTATION Doctor of Philosophy in Biomedical Science (Cancer Biology) Aneuploidy: Using genetic instability to preserve a haploid genome? Submitted by: Ramona Ramdath In partial fulfillment of the requirements for the degree of Doctor of Philosophy in Biomedical Science Examination Committee Signature/Date Major Advisor: David Allison, M.D., Ph.D. Academic James Trempe, Ph.D. Advisory Committee: David Giovanucci, Ph.D. Randall Ruch, Ph.D. Ronald Mellgren, Ph.D. Senior Associate Dean College of Graduate Studies Michael S. Bisesi, Ph.D. Date of Defense: April 10, 2009 Aneuploidy: Using genetic instability to preserve a haploid genome? Ramona Ramdath University of Toledo, Health Science Campus 2009 Dedication I dedicate this dissertation to my grandfather who died of lung cancer two years ago, but who always instilled in us the value and importance of education. And to my mom and sister, both of whom have been pillars of support and stimulating conversations. To my sister, Rehanna, especially- I hope this inspires you to achieve all that you want to in life, academically and otherwise. ii Acknowledgements As we go through these academic journeys, there are so many along the way that make an impact not only on our work, but on our lives as well, and I would like to say a heartfelt thank you to all of those people: My Committee members- Dr. James Trempe, Dr. David Giovanucchi, Dr. Ronald Mellgren and Dr. Randall Ruch for their guidance, suggestions, support and confidence in me. My major advisor- Dr. David Allison, for his constructive criticism and positive reinforcement. -

A Novel Resveratrol Analog: Its Cell Cycle Inhibitory, Pro-Apoptotic and Anti-Inflammatory Activities on Human Tumor Cells
A NOVEL RESVERATROL ANALOG : ITS CELL CYCLE INHIBITORY, PRO-APOPTOTIC AND ANTI-INFLAMMATORY ACTIVITIES ON HUMAN TUMOR CELLS A dissertation submitted to Kent State University in partial fulfillment of the requirements for the degree of Doctor of Philosophy by Boren Lin May 2006 Dissertation written by Boren Lin B.S., Tunghai University, 1996 M.S., Kent State University, 2003 Ph. D., Kent State University, 2006 Approved by Dr. Chun-che Tsai , Chair, Doctoral Dissertation Committee Dr. Bryan R. G. Williams , Co-chair, Doctoral Dissertation Committee Dr. Johnnie W. Baker , Members, Doctoral Dissertation Committee Dr. James L. Blank , Dr. Bansidhar Datta , Dr. Gail C. Fraizer , Accepted by Dr. Robert V. Dorman , Director, School of Biomedical Sciences Dr. John R. Stalvey , Dean, College of Arts and Sciences ii TABLE OF CONTENTS LIST OF FIGURES……………………………………………………………….………v LIST OF TABLES……………………………………………………………………….vii ACKNOWLEDGEMENTS….………………………………………………………….viii I INTRODUCTION….………………………………………………….1 Background and Significance……………………………………………………..1 Specific Aims………………………………………………………………………12 II MATERIALS AND METHODS.…………………………………………….16 Cell Culture and Compounds…….……………….…………………………….….16 MTT Cell Viability Assay………………………………………………………….16 Trypan Blue Exclusive Assay……………………………………………………...18 Flow Cytometry for Cell Cycle Analysis……………..……………....……………19 DNA Fragmentation Assay……………………………………………...…………23 Caspase-3 Activity Assay………………………………...……….….…….………24 Annexin V-FITC Staining Assay…………………………………..…...….………28 NF-kappa B p65 Activity Assay……………………………………..………….…29 -

In Vivo Loss-Of-Function Screens Identify KPNB1 As a New Druggable
In vivo loss-of-function screens identify KPNB1 as a PNAS PLUS new druggable oncogene in epithelial ovarian cancer Michiko Kodamaa,b,1, Takahiro Kodamaa,c,1,2, Justin Y. Newberga,d, Hiroyuki Katayamae, Makoto Kobayashie, Samir M. Hanashe, Kosuke Yoshiharaf, Zhubo Weia, Jean C. Tiena,g, Roberto Rangela,h, Kae Hashimotob, Seiji Mabuchib, Kenjiro Sawadab, Tadashi Kimurab, Neal G. Copelanda,i, and Nancy A. Jenkinsa,i,2 aCancer Research Program, Houston Methodist Research Institute, Houston, TX 77030; bDepartment of Obstetrics and Gynecology, Osaka University Graduate School of Medicine, Osaka 5650871, Japan; cDepartment of Gastroenterology and Hepatology, Osaka University Graduate School of Medicine, Osaka 5650871, Japan; dDepartment of Molecular Oncology, Moffitt Cancer Center, Tampa, FL 33612; eDepartment of Clinical Cancer Prevention, University of Texas MD Anderson Cancer Center, Houston, TX 77030; fDepartment of Obstetrics and Gynecology, Niigata University Graduate School of Medical and Dental Sciences, Niigata 9518510, Japan; gDepartment of Pathology, Michigan Center for Translational Pathology, University of Michigan, Ann Arbor, MI 48109; hDepartment of Immunology, University of Texas MD Anderson Cancer Center, Houston, TX 77030; and iDepartment of Genetics, University of Texas MD Anderson Cancer Center, Houston, TX 77030 Contributed by Nancy A. Jenkins, July 23, 2017 (sent for review April 3, 2017; reviewed by Roland Rad and Kosuke Yusa) Epithelial ovarian cancer (EOC) is a deadly cancer, and its prognosis has To overcome this problem, several alternative methods have re- not been changed significantly during several decades. To seek new cently been used for cancer gene discovery, including insertional therapeutic targets for EOC, we performed an in vivo dropout screen mutagenesis and RNAi/shRNA/CRISPR/Cas9-based screens. -

RANBP1 (NM 002882) Human Recombinant Protein – TP323305 | Origene
OriGene Technologies, Inc. 9620 Medical Center Drive, Ste 200 Rockville, MD 20850, US Phone: +1-888-267-4436 [email protected] EU: [email protected] CN: [email protected] Product datasheet for TP323305 RANBP1 (NM_002882) Human Recombinant Protein Product data: Product Type: Recombinant Proteins Description: Recombinant protein of human RAN binding protein 1 (RANBP1) Species: Human Expression Host: HEK293T Tag: C-Myc/DDK Predicted MW: 23.1 kDa Concentration: >50 ug/mL as determined by microplate BCA method Purity: > 80% as determined by SDS-PAGE and Coomassie blue staining Buffer: 25 mM Tris.HCl, pH 7.3, 100 mM glycine, 10% glycerol Preparation: Recombinant protein was captured through anti-DDK affinity column followed by conventional chromatography steps. Storage: Store at -80°C. Stability: Stable for 12 months from the date of receipt of the product under proper storage and handling conditions. Avoid repeated freeze-thaw cycles. RefSeq: NP_002873 Locus ID: 5902 UniProt ID: P04049, P43487, L7RRS6, A0A140VK94 RefSeq Size: 884 Cytogenetics: 22q11.21 RefSeq ORF: 603 Synonyms: HTF9A Summary: This gene encodes a protein that forms a complex with Ras-related nuclear protein (Ran) and metabolizes guanoside triphosphate (GTP). This complex participates in the regulation of the cell cycle by controlling transport of proteins and nucleic acids into the nucleus. There are multiple pseudogenes for this gene on chromosomes 9, 12, 17, and X. Alternative splicing results in multiple transcript variants. [provided by RefSeq, Jul 2013] This product is to be used for laboratory only. Not for diagnostic or therapeutic use. View online » ©2021 OriGene Technologies, Inc., 9620 Medical Center Drive, Ste 200, Rockville, MD 20850, US 1 / 2 RANBP1 (NM_002882) Human Recombinant Protein – TP323305 Product images: Coomassie blue staining of purified RANBP1 protein (Cat# TP323305). -

Gene Expression During Normal and FSHD Myogenesis Tsumagari Et Al
Gene expression during normal and FSHD myogenesis Tsumagari et al. Tsumagari et al. BMC Medical Genomics 2011, 4:67 http://www.biomedcentral.com/1755-8794/4/67 (27 September 2011) Tsumagari et al. BMC Medical Genomics 2011, 4:67 http://www.biomedcentral.com/1755-8794/4/67 RESEARCHARTICLE Open Access Gene expression during normal and FSHD myogenesis Koji Tsumagari1, Shao-Chi Chang1, Michelle Lacey2,3, Carl Baribault2,3, Sridar V Chittur4, Janet Sowden5, Rabi Tawil5, Gregory E Crawford6 and Melanie Ehrlich1,3* Abstract Background: Facioscapulohumeral muscular dystrophy (FSHD) is a dominant disease linked to contraction of an array of tandem 3.3-kb repeats (D4Z4) at 4q35. Within each repeat unit is a gene, DUX4, that can encode a protein containing two homeodomains. A DUX4 transcript derived from the last repeat unit in a contracted array is associated with pathogenesis but it is unclear how. Methods: Using exon-based microarrays, the expression profiles of myogenic precursor cells were determined. Both undifferentiated myoblasts and myoblasts differentiated to myotubes derived from FSHD patients and controls were studied after immunocytochemical verification of the quality of the cultures. To further our understanding of FSHD and normal myogenesis, the expression profiles obtained were compared to those of 19 non-muscle cell types analyzed by identical methods. Results: Many of the ~17,000 examined genes were differentially expressed (> 2-fold, p < 0.01) in control myoblasts or myotubes vs. non-muscle cells (2185 and 3006, respectively) or in FSHD vs. control myoblasts or myotubes (295 and 797, respectively). Surprisingly, despite the morphologically normal differentiation of FSHD myoblasts to myotubes, most of the disease-related dysregulation was seen as dampening of normal myogenesis- specific expression changes, including in genes for muscle structure, mitochondrial function, stress responses, and signal transduction. -

Identification of Differentially Expressed Genes in Human Bladder Cancer Through Genome-Wide Gene Expression Profiling
521-531 24/7/06 18:28 Page 521 ONCOLOGY REPORTS 16: 521-531, 2006 521 Identification of differentially expressed genes in human bladder cancer through genome-wide gene expression profiling KAZUMORI KAWAKAMI1,3, HIDEKI ENOKIDA1, TOKUSHI TACHIWADA1, TAKENARI GOTANDA1, KENGO TSUNEYOSHI1, HIROYUKI KUBO1, KENRYU NISHIYAMA1, MASAKI TAKIGUCHI2, MASAYUKI NAKAGAWA1 and NAOHIKO SEKI3 1Department of Urology, Graduate School of Medical and Dental Sciences, Kagoshima University, 8-35-1 Sakuragaoka, Kagoshima 890-8520; Departments of 2Biochemistry and Genetics, and 3Functional Genomics, Graduate School of Medicine, Chiba University, 1-8-1 Inohana, Chuo-ku, Chiba 260-8670, Japan Received February 15, 2006; Accepted April 27, 2006 Abstract. Large-scale gene expression profiling is an effective CKS2 gene not only as a potential biomarker for diagnosing, strategy for understanding the progression of bladder cancer but also for staging human BC. This is the first report (BC). The aim of this study was to identify genes that are demonstrating that CKS2 expression is strongly correlated expressed differently in the course of BC progression and to with the progression of human BC. establish new biomarkers for BC. Specimens from 21 patients with pathologically confirmed superficial (n=10) or Introduction invasive (n=11) BC and 4 normal bladder samples were studied; samples from 14 of the 21 BC samples were subjected Bladder cancer (BC) is among the 5 most common to microarray analysis. The validity of the microarray results malignancies worldwide, and the 2nd most common tumor of was verified by real-time RT-PCR. Of the 136 up-regulated the genitourinary tract and the 2nd most common cause of genes we detected, 21 were present in all 14 BCs examined death in patients with cancer of the urinary tract (1-7).