Photochemical & Photobiological Sciences
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-
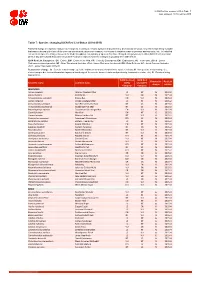
Table 7: Species Changing IUCN Red List Status (2014-2015)
IUCN Red List version 2015.4: Table 7 Last Updated: 19 November 2015 Table 7: Species changing IUCN Red List Status (2014-2015) Published listings of a species' status may change for a variety of reasons (genuine improvement or deterioration in status; new information being available that was not known at the time of the previous assessment; taxonomic changes; corrections to mistakes made in previous assessments, etc. To help Red List users interpret the changes between the Red List updates, a summary of species that have changed category between 2014 (IUCN Red List version 2014.3) and 2015 (IUCN Red List version 2015-4) and the reasons for these changes is provided in the table below. IUCN Red List Categories: EX - Extinct, EW - Extinct in the Wild, CR - Critically Endangered, EN - Endangered, VU - Vulnerable, LR/cd - Lower Risk/conservation dependent, NT - Near Threatened (includes LR/nt - Lower Risk/near threatened), DD - Data Deficient, LC - Least Concern (includes LR/lc - Lower Risk, least concern). Reasons for change: G - Genuine status change (genuine improvement or deterioration in the species' status); N - Non-genuine status change (i.e., status changes due to new information, improved knowledge of the criteria, incorrect data used previously, taxonomic revision, etc.); E - Previous listing was an Error. IUCN Red List IUCN Red Reason for Red List Scientific name Common name (2014) List (2015) change version Category Category MAMMALS Aonyx capensis African Clawless Otter LC NT N 2015-2 Ailurus fulgens Red Panda VU EN N 2015-4 -

Priority Contribution Quantifying the Illegal Parrot Trade in Santa Cruz De La Sierra, Bolivia, with Emphasis on Threatened Spec
Bird Conservation International (2007) 17:295–300. ß BirdLife International 2007 doi: 10.1017/S0959270907000858 Printed in the United Kingdom Priority contribution Quantifying the illegal parrot trade in Santa Cruz de la Sierra, Bolivia, with emphasis on threatened species MAURICIO HERRERA HURTADO and BENNETT HENNESSEY Summary We monitored the illegal pet trade in Los Pozos pet market from August 2004 to July 2005. As indicated in Bolivian law, all unauthorized trade in wild animal species is illegal, especially species considered threatened by IUCN. During this period, we recorded 7,279 individuals of 31 parrot species, including four threatened species, two of which were being transported from Brazil through Bolivia to markets in Peru. The most frequently sold species was the Blue-fronted Parrot Amazona aestiva with 1,468 individuals observed during our study, the majority of which (94%) were believed to have been captured in the wild. Most of the purchased birds remain within Bolivia, while the more expensive, threatened species frequently head to Peru; some individuals may even reach Europe. We believe our study describes only a small proportion of the Bolivian parrot trade, underscoring the potential extent of the illegal pet trade and the need for better Bolivian law enforcement. Resumen Monitoreamos el comercio ilegal de aves en el mercado de mascotas de Los Pozos, desde agosto de 2004 a julio de 2005. De acuerdo a lo que establece la ley boliviana, todo comercio no autorizado de animales salvajes es ilegal, especialmente de especies consideradas Amenazadas por la IUCN. Durante este periodo, grabamos 7.279 individuos de 31 especies de loros, incluyendo 4 especies amenazadas, de las cuales dos fueron transportadas desde Brasil a trave´s de Bolivia hacia mercados en Peru´ . -

Photoinhibition and Photoprotection in Symbiotic Dinoflagellates from Reef-Building Corals
MARINE ECOLOGY PROGRESS SERIES Vol. 183: 73-86.1999 Published July 6 Mar Ecol Prog Ser 1 Photoinhibition and photoprotection in symbiotic dinoflagellates from reef-building corals Ove Hoegh-Guldberg*, Ross J. Jones School of Biological Sciences, Building A08. The University of Sydney, New South Wales 2006. Australia ABSTRACT: Pulse-amplitude-modulation fluorometry and oxygen respirometry were used to investi- gate die1 photosynthetic responses by symbiotic dnoflagellates to light levels in summer and winter on a high latitude coral reef. The symbiotic dinoflagellates from 2 species of reef-building coral (Porites cylindnca and Stylophora pistillata) showed photoinhibitory decreases in the ratio of variable (F,) to maximal (F,) fluorescence (F,/F,,,) as early as 09:00 h on both summer and winter days on the reefs associated wlth One Tree Island (23" 30'S, 1.52" 06' E; Great Barrier Reef, Australia). This was due to decreases in maximum, F,, and to a smaller extent minimum, F,, chlorophyll fluorescence. Complete recovery took 4 to 6 h and began to occur as soon as light levels fell each day. Chlorophyll fluorescence quenching analysis of corals measured during the early afternoon revealed classic regulation of photo- system I1 (PSII) efficiency through non-photochemical quenching (NPQ). These results appear to be similar to data collected for other algae and higher plants, suggesting involvement of the xanthophyll cycle of symbiotic dinoflagellates in regulating the quantum efficiency of PSII. The ability of symbiotic dinoflagellates to develop significant NPQ, however, depended strongly on when the symbiotic dinoflagellates were studied. Whereas symbiotic dinoflagellates from corals in the early afternoon showed a significant capacity to regulate the efficiency of PSII using NPQ, those sampled before sun- rise had a slower and much reduced capacity, suggesting that elements of the xanthophyll cycle are suppressed prior to sunrise. -

TAG Operational Structure
PARROT TAXON ADVISORY GROUP (TAG) Regional Collection Plan 5th Edition 2020-2025 Sustainability of Parrot Populations in AZA Facilities ...................................................................... 1 Mission/Objectives/Strategies......................................................................................................... 2 TAG Operational Structure .............................................................................................................. 3 Steering Committee .................................................................................................................... 3 TAG Advisors ............................................................................................................................... 4 SSP Coordinators ......................................................................................................................... 5 Hot Topics: TAG Recommendations ................................................................................................ 8 Parrots as Ambassador Animals .................................................................................................. 9 Interactive Aviaries Housing Psittaciformes .............................................................................. 10 Private Aviculture ...................................................................................................................... 13 Communication ........................................................................................................................ -

Prevalence of Color Blindness Among Students of Four Basic Schools in Koya City
Journal of Advanced Laboratory Research in Biology E-ISSN: 0976-7614 Volume 10, Issue 2, April 2019 PP 31-34 https://e-journal.sospublication.co.in Research Article Prevalence of Color Blindness among Students of four basic schools in Koya City Harem Othman Smail*, Evan Mustafa Ismael and Sayran Anwer Ali Department of Biology, Koya University, University Park, Danielle Mitterrand Boulevard, Koysinjaq, Kurdistan Region –Iraq. Abstract: Color blindness or color vision deficiency is X-linked recessive disorder that affects males more frequently than females. Abnormality in any one or all three cone photoreceptors caused Congenital disorders. Protanopia, deuteranopia results when long wavelength (L), photopigments (red), middle wavelength (M) and photopigments (green) are missing. This cross-sectional study was done to find out the prevalence of color vision deficiency among basic school students in Koya city with different ages and genders. The study was conducted in four basic schools that were present in Koya city (Zheen, Zanst, Nawroz and Najibaxan). All students screened by using Ishihara 24 plates. For the study (n=400, male=206, female=194, age=8-14) were selected & examined. The result revealed that the prevalence rate of the deficiency in four primary schools 3.39% (7) in males and 0% females. The study concluded that color blindness is different between students in each school, cannot find the prevalence of Color blindness in females in each school and affects males more than females because color blindness is an X-linked recessive disorder. The aim of the study was to determine the prevalence of color blindness in some basic schools in Koya city, Kurdistan Region/ Iraq. -

Consequences of Color Vision Variation on Performance and Fitness in Capuchin Monkeys
University of Montana ScholarWorks at University of Montana Graduate Student Theses, Dissertations, & Professional Papers Graduate School 2014 Consequences of color vision variation on performance and fitness in capuchin monkeys Andrea Theresa Green Follow this and additional works at: https://scholarworks.umt.edu/etd Let us know how access to this document benefits ou.y Recommended Citation Green, Andrea Theresa, "Consequences of color vision variation on performance and fitness in capuchin monkeys" (2014). Graduate Student Theses, Dissertations, & Professional Papers. 10766. https://scholarworks.umt.edu/etd/10766 This Dissertation is brought to you for free and open access by the Graduate School at ScholarWorks at University of Montana. It has been accepted for inclusion in Graduate Student Theses, Dissertations, & Professional Papers by an authorized administrator of ScholarWorks at University of Montana. For more information, please contact [email protected]. CONSEQUENCES OF COLOR VISION VARIATION ON PERFORMANCE AND FITNESS IN CAPUCHIN MONKEYS By ANDREA THERESA GREEN Masters of Arts, Stony Brook University, Stony Brook, NY, 2007 Bachelors of Science, Warren Wilson College, Asheville, NC, 1997 Dissertation Paper presented in partial fulfillment of the requirements for the degree of Doctor of Philosophy in Organismal Biology and Ecology The University of Montana Missoula, MT May 2014 Approved by: Sandy Ross, Dean of The Graduate School Graduate School Charles H. Janson, Chair Division of Biological Sciences Erick Greene Division of Biological Sciences Doug J. Emlen Division of Biological Sciences Scott R. Miller Division of Biological Sciences Gerald H. Jacobs Psychological & Brain Sciences-UCSB UMI Number: 3628945 All rights reserved INFORMATION TO ALL USERS The quality of this reproduction is dependent upon the quality of the copy submitted. -

The Distribution of Mycosporine-Like Amino Acids (Maas) and the Phylogenetic Identity of Symbiotic Dinoflagellates in Cnidarian
Journal of Experimental Marine Biology and Ecology 337 (2006) 131–146 www.elsevier.com/locate/jembe The distribution of mycosporine-like amino acids (MAAs) and the phylogenetic identity of symbiotic dinoflagellates in cnidarian hosts from the Mexican Caribbean ⁎ Anastazia T. Banaszak a, , Maria Guadalupe Barba Santos a, Todd C. LaJeunesse b, Michael P. Lesser c a Unidad Académica Puerto Morelos, Instituto de Ciencias del Mar y Limnología, Universidad Nacional Autónoma de México, Apartado Postal 1152, Cancún, Quintana Roo, 77500, Mexico b Department of Biology, Florida International University, University Park Campus, Miami, Florida, 33199, USA c Department of Zoology and Center for Marine Biology, University of New Hampshire, Durham, New Hampshire, 03824, USA Received 22 May 2006; accepted 10 June 2006 Abstract A survey of 54 species of symbiotic cnidarians that included hydrozoan corals, anemones, gorgonians and scleractinian corals was conducted in the Mexican Caribbean for the presence of mycosporine-like amino acids (MAAs) in the host as well as the Symbio- dinium fractions. The host fractions contained relatively simple MAA profiles, all harbouring between one and three MAAs, principally mycosporine-glycine followed by shinorine and porphyra-334 in smaller amounts. Symbiodinium populations were identified to sub-generic levels using PCR-DGGE analysis of the Internal Transcribed Spacer 2 (ITS2) region. Regardless of clade identity, all Symbiodinium extracts contained MAAs, in contrast to the pattern that has been found in cultures of Symbiodinium, where clade A symbionts produced MAAs whereas clade B, C, D, and E symbionts did not. Under natural conditions between one and four MAAs were identified in the symbiont fractions, mycosporine-glycine (λmax =310 nm), shinorine (λmax =334 nm), porphyra-334 (λmax =334 nm) and palythine (λmax =320 nm). -

Level Biology. Basic and Simplified Revision Notes
Systematic “A” level Biology. Basic and simplified Revision notes. SYSTEMATIC “A” LEVEL BIOLOGY. Basic and simplified revision notes. STANDARD TEACHING SYLABUS: 1. Cell biology or cytology………………………………………..………………………….2 Definition of cytology/cell biology, definition of the cell. Microscopy. light and electron microscopes their structure, mode of operation and comparison between them, microscope practical techniques. Cell theory, types of organism’s i.e prokaryotes and eukaryotes, comparison between Prokaryotes and eukaryotes, why cells are small? Structures of the cell, cell diversity.. Cell division. Types of cell division, events that occur during each type, comparison between them and the importance of each type. 2.Histology………………………………………………………………………………………..4 Definition, types of tissues, their structures and functions,adaptations of some tissues to suit their function. Levels of organization. ie unicellular level; tissue level; organ level; system level and organism level; advantages and disadvantages of being unicellular and multicelar organism; 3. Classification of living organisms…………………………………………………….65 Common terms used: classification, taxonomy, systematics, binomial nomenclature, dichotomous keys, taxonomic hierarchy, and five kingdom system: Animalia, Plantae, fungi, Protista and monera general characteristic of organismin each kingdom and the examples. 4. Transport of materials in living organisms………………………………………107 5. Chemicals of life………………………………………………………………………….150 DNA structure, RNA structure, DNA replication and protein -

Environmental Impact Assessment
Environmental Impact Assessment December 2013 IND: SASEC Road Connectivity Investment Program (formerly SASEC Road Connectivity Sector Project) Asian Highway 2 (India /Nepal Border to India/Bangladesh Border) Asian Highway 48 (India/Bhutan Border to India/Bangladesh Border) Prepared by Ministry of Roads Transport and Highways, Government of India and Public Works Department, Government of West Bengal for the Asian Development Bank. This is a revised version of the draft originally posted in July 2013 available on http://www.adb.org/projects/47341- 001/documents/. CURRENCY EQUIVALENTS (As of 30 April 2013) Currency unit – Indian rupee (INR) INR1.00 = $ 0.01818 $1.00 = INR 55.00 ABBREVIATION AADT Annual Average Daily Traffic AAQ Ambient air quality AAQM Ambient air quality monitoring ADB Asian Development Bank AH Asian Highway ASI Archaeological Survey of India BDL Below detectable limit BGL Below ground level BOD Biochemical oxygen demand BOQ Bill of quantity CCE Chief Controller of Explosives CGWA Central Ground Water Authority CITES Convention on International Trade in Endangered Species CO Carbon monoxide COD Chemical oxygen demand CPCB Central Pollution Control Board CSC Construction Supervision Consultant DFO Divisional Forest Officer DG Diesel generating set DO Dissolved oxygen DPR Detailed project report E&S Environment and social EA Executing agency EAC Expert Appraisal Committee EFP Environmental Focal Person EHS Environment Health and Safety EIA Environmental impact assessment EMOP Environmental monitoring plan EMP Environmental -

Color Dichromatism in Female American Redstarts.-Male American Redstarts (Setophaga Ruticilla) Are Easily Categorized by Plumage Into Yearlings (Subadults) and Adults
SHORT COMMUNICATIONS 257 HUDSON, R. H., R. K. TUCKER, AND M. A. HAEGELE. 1984. Handbook of toxicity of pesticides to wildlife. 3rd ed. U.S.D.I. Fish Wildlife Serv. Resource Publ. 153. MANNAN, R. W., M. L. MORRISON, AND E. C. MESLOW. 1984. Comment: The use of guilds in forest bird management. Wildl. Sot. Bull. 12:426-430. MCCLELLAND, B. R. AND S. S. FRISSEL. 1975. Identifying forest snags useful for hole-nesting birds. J. For. 73:404-417. MCCOMB, W. C. AND R. N. MULLER. 1983. Snag densities in old-growth and second- growth Appalachian forests. J. Wildl. Manage. 47:376-382. MCPEEK, G. A. 1985. Decay patterns and bird use of snags created with herbicide injections and tree topping. M.S. thesis. Univ. Kentucky, Lexington, Kentucky. MORIARTY, J. J. 1982. Long-term effects of timber stand improvement on snag and natural cavity characteristics and cavity use by vertebrates in a mixed mesophytic forest. M.S. thesis. Univ. Kentucky, Lexington, Kentucky. SAS INSTITUTE. 1982. SAS users’ guide: statistics, 1982 ed. SAS Institute, Cary, North Carolina. SEDGWICK, J. A. AND F. L. KNOPF. 1986. Cavity-nesting birds and the cavity-tree resource in plains cottonwood bottomlands. J. Wildl. Manage. 50:247-252. THOMAS, J. W., R. G. ANDERSON, C. MASER, AND E. L. BULL. 1979. Snags. Pp. 60-77 in Wildlife habitats in managed forests, the Blue Mountains of Oregon and Washington (J. W. Thomas, ed.). U.S.D.A. For. Serv. Agric. Handb. 553. U.S. FORESTSERVICE. 1984. Pesticide background statements. Vol. IV, Herbicides. U.S.D.A. -

And Interspecific Contex
Light matters: testing the “Light Environment Hypothesis” under intra- and interspecific contexts Angelica Hernandez-Palma School of Renewable Natural Resources, Louisiana State University Agricultural Center, Louisiana State University, Baton Rouge, Louisiana Keywords Abstract Dichromatism, Furnariides, model of color discrimination, species recognition, The “Light Environment Hypothesis” (LEH) proposes that evolution of inter- tetrahedral color space model, visual specific variation in plumage color is driven by variation in light environments signaling. across habitats. If ambient light has the potential to drive interspecific variation, a similar influence should be expected for intraspecific recognition, as color sig- Correspondence nals are an adaptive response to the change in ambient light levels in different Angelica Hernandez-Palma, School of habitats. Using spectrometry, avian-appropriate models of vision, and phyloge- Renewable Natural Resources, Louisiana State netic comparative methods, I quantified dichromatism and tested the LEH in University Agricultural Center, Louisiana State University, Room 227 RNR Building, Baton both intra- and interspecific contexts in 33 Amazonian species from the infraor- Rouge, Louisiana 70803. der Furnariides living in environments with different light levels. Although Tel: 225 578 4228; these birds are sexually monochromatic to humans, 81.8% of the species had at Fax: 225-578-4227; least one dichromatic patch in their plumage, mostly from dorsal areas, which E-mail: [email protected] provides evidence for a role for dichromatism in sex recognition. Furthermore, birds from habitats with high levels of ambient light had higher dichromatism Funding Information levels, as well as brighter, more saturated, and more diverse plumages, suggest- National Science Foundation (Grant/Award Number: ‘0545491’, ‘1257340’) ing that visual communication is less constrained in these habitats. -

International Journal of Research Volume VIII, Issue VI, JUNE/2019
International Journal of Research ISSN NO:2236-6124 A Study on the Congregation of Adult Butterflies on Non-floral Resources at Different Locations in Jalpaiguri district of West Bengal, India Panchali Sengupta1*, Narayan Ghorai2 1Department of Zoology, West Bengal State University, Berunanpukaria, Malikapur, Barasat, District-24 Parganas (North), Kolkata-700126.West Bengal, India Email id: [email protected] 2Department of Zoology, West Bengal State University, Berunanpukaria, Malikapur, Barasat, District-24 Parganas (North), Kolkata-700126.West Bengal, India email id: [email protected] Abstract Several instances of puddling, as reported among different herbivore arthropods, appears quite interesting. Significantly, congregation of adult butterflies at several non-floral resources (wet soil/mud, animal dung, bird droppings, carrion, rotten/fermenting fruits) were examined at different locations in Jalpaiguri district adjacent to the tea estates, villages and agricultural tracts. Different species of papilionids and pierids congregate on wet soil patch and puddle collectively. However other species of nymphalid, lycaenid and hesperid are found to puddle individually, without associating with others on resources like excrements and carrion. Irrespective of any species newly emerged males, and aged females are found to puddle. Interestingly, each species belonging to a particular family have a specific range of puddling duration. Such specificity in puddling among species of a family could probably be associated with their need for a common nutrient. Keywords:, congregation, hesperid, lycaenid, nymphalid, papilionid, pierid *corresponding author Volume VIII, Issue VI, JUNE/2019 Page No:5877 International Journal of Research ISSN NO:2236-6124 Introduction Puddling is a widely recognised fascinating event in the life history of any herbivore arthropods except beetles targeted towards accumulation of specific micronutrient (Mollemann, 2010).