Problems in Cladistic Classification: Higher-Level Relationships in Land Plants Peter R
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Diversity Patterns of the Vascular Plant Group Zosterophyllopsida in Relation to Devonian Paleogeography Borja Cascales-Miñana, Brigitte Meyer-Berthaud
Diversity patterns of the vascular plant group Zosterophyllopsida in relation to Devonian paleogeography Borja Cascales-Miñana, Brigitte Meyer-Berthaud To cite this version: Borja Cascales-Miñana, Brigitte Meyer-Berthaud. Diversity patterns of the vascular plant group Zosterophyllopsida in relation to Devonian paleogeography. Palaeogeography, Palaeoclimatology, Palaeoecology, Elsevier, 2015, 423, pp.53-61. 10.1016/j.palaeo.2015.01.024. hal-01140840 HAL Id: hal-01140840 https://hal-sde.archives-ouvertes.fr/hal-01140840 Submitted on 26 Nov 2020 HAL is a multi-disciplinary open access L’archive ouverte pluridisciplinaire HAL, est archive for the deposit and dissemination of sci- destinée au dépôt et à la diffusion de documents entific research documents, whether they are pub- scientifiques de niveau recherche, publiés ou non, lished or not. The documents may come from émanant des établissements d’enseignement et de teaching and research institutions in France or recherche français ou étrangers, des laboratoires abroad, or from public or private research centers. publics ou privés. Palaeogeography, Palaeoclimatology, Palaeoecology 423 (2015) 53–61 Contents lists available at ScienceDirect Palaeogeography, Palaeoclimatology, Palaeoecology journal homepage: www.elsevier.com/locate/palaeo Diversity patterns of the vascular plant group Zosterophyllopsida in relation to Devonian paleogeography Borja Cascales-Miñana a,b,⁎, Brigitte Meyer-Berthaud a a CNRS, Université de Montpellier, UMR Botanique et bioinformatique de l'architecture des plantes et des végétations (AMAP), F-34398 Montpellier Cedex 5, France b PPP, Département de Géologie, Université de Liège, Allée du 6 Août, B18 Sart Tilman, B-4000 Liège, Belgium article info abstract Article history: The Zosterophyllopsida originated in the Silurian and became prominent vascular components of Early Devonian Received 11 April 2014 floras worldwide. -

Fossils and Plant Phylogeny1
American Journal of Botany 91(10): 1683±1699. 2004. FOSSILS AND PLANT PHYLOGENY1 PETER R. CRANE,2,5 PATRICK HERENDEEN,3 AND ELSE MARIE FRIIS4 2Royal Botanic Gardens, Kew, Richmond, Surrey TW9 3AB, UK; 3Department of Biological Sciences, The George Washington University, Washington DC 20052 USA; 4Department of Palaeobotany, Swedish Museum of Natural History, Box 50007, S-104 05 Stockholm, Sweden Developing a detailed estimate of plant phylogeny is the key ®rst step toward a more sophisticated and particularized understanding of plant evolution. At many levels in the hierarchy of plant life, it will be impossible to develop an adequate understanding of plant phylogeny without taking into account the additional diversity provided by fossil plants. This is especially the case for relatively deep divergences among extant lineages that have a long evolutionary history and in which much of the relevant diversity has been lost by extinction. In such circumstances, attempts to integrate data and interpretations from extant and fossil plants stand the best chance of success. For this to be possible, what will be required is meticulous and thorough descriptions of fossil material, thoughtful and rigorous analysis of characters, and careful comparison of extant and fossil taxa, as a basis for determining their systematic relationships. Key words: angiosperms; fossils; paleobotany; phylogeny; spermatophytes; tracheophytes. Most biological processes, such as reproduction or growth distic context, neither fossils nor their stratigraphic position and development, can only be studied directly or manipulated have any special role in inferring phylogeny, and although experimentally using living organisms. Nevertheless, much of more complex models have been developed (see Fisher, 1994; what we have inferred about the large-scale processes of plant Huelsenbeck, 1994), these have not been widely adopted. -

Seed Plant Models
Review Tansley insight Why we need more non-seed plant models Author for correspondence: Stefan A. Rensing1,2 Stefan A. Rensing 1 2 Tel: +49 6421 28 21940 Faculty of Biology, University of Marburg, Karl-von-Frisch-Str. 8, 35043 Marburg, Germany; BIOSS Biological Signalling Studies, Email: stefan.rensing@biologie. University of Freiburg, Sch€anzlestraße 18, 79104 Freiburg, Germany uni-marburg.de Received: 30 October 2016 Accepted: 18 December 2016 Contents Summary 1 V. What do we need? 4 I. Introduction 1 VI. Conclusions 5 II. Evo-devo: inference of how plants evolved 2 Acknowledgements 5 III. We need more diversity 2 References 5 IV. Genomes are necessary, but not sufficient 3 Summary New Phytologist (2017) Out of a hundred sequenced and published land plant genomes, four are not of flowering plants. doi: 10.1111/nph.14464 This severely skewed taxonomic sampling hinders our comprehension of land plant evolution at large. Moreover, most genetically accessible model species are flowering plants as well. If we are Key words: Charophyta, evolution, fern, to gain a deeper understanding of how plants evolved and still evolve, and which of their hornwort, liverwort, moss, Streptophyta. developmental patterns are ancestral or derived, we need to study a more diverse set of plants. Here, I thus argue that we need to sequence genomes of so far neglected lineages, and that we need to develop more non-seed plant model species. revealed much, the exact branching order and evolution of the I. Introduction nonbilaterian lineages is still disputed (Lanna, 2015). Research on animals has for a long time relied on a number of The first (small) plant genome to be sequenced was of THE traditional model organisms, such as mouse, fruit fly, zebrafish or model plant, the weed Arabidopsis thaliana (c. -

Chromosome Numbers in Gymnosperms - an Update
Rastogi and Ohri . Silvae Genetica (2020) 69, 13 - 19 13 Chromosome Numbers in Gymnosperms - An Update Shubhi Rastogi and Deepak Ohri Amity Institute of Biotechnology, Research Cell, Amity University Uttar Pradesh, Lucknow Campus, Malhaur (Near Railway Station), P.O. Chinhat, Luc know-226028 (U.P.) * Corresponding author: Deepak Ohri, E mail: [email protected], [email protected] Abstract still some controversy with regard to a monophyletic or para- phyletic origin of the gymnosperms (Hill 2005). Recently they The present report is based on a cytological data base on 614 have been classified into four subclasses Cycadidae, Ginkgoi- (56.0 %) of the total 1104 recognized species and 82 (90.0 %) of dae, Gnetidae and Pinidae under the class Equisetopsida the 88 recognized genera of gymnosperms. Family Cycada- (Chase and Reveal 2009) comprising 12 families and 83 genera ceae and many genera of Zamiaceae show intrageneric unifor- (Christenhusz et al. 2011) and 88 genera with 1104 recognized mity of somatic numbers, the genus Zamia is represented by a species according to the Plant List (www.theplantlist.org). The range of number from 2n=16-28. Ginkgo, Welwitschia and Gen- validity of accepted name of each taxa and the total number of tum show 2n=24, 2n=42, and 2n=44 respectively. Ephedra species in each genus has been checked from the Plant List shows a range of polyploidy from 2x-8x based on n=7. The (www.theplantlist.org). The chromosome numbers of 688 taxa family Pinaceae as a whole shows 2n=24except for Pseudolarix arranged according to the recent classification (Christenhusz and Pseudotsuga with 2n=44 and 2n=26 respectively. -

Ptolemeba N. Gen., a Novel Genus of Hartmannellid Amoebae (Tubulinea, Amoebozoa); with an Emphasis on the Taxonomy of Saccamoeba
The Journal of Published by the International Society of Eukaryotic Microbiology Protistologists Journal of Eukaryotic Microbiology ISSN 1066-5234 ORIGINAL ARTICLE Ptolemeba n. gen., a Novel Genus of Hartmannellid Amoebae (Tubulinea, Amoebozoa); with an Emphasis on the Taxonomy of Saccamoeba Pamela M. Watsona, Stephanie C. Sorrella & Matthew W. Browna,b a Department of Biological Sciences, Mississippi State University, Mississippi State, Mississippi, 39762 b Institute for Genomics, Biocomputing & Biotechnology, Mississippi State University, Mississippi State, Mississippi, 39762 Keywords ABSTRACT 18S rRNA; amoeba; amoeboid; Cashia; cristae; freshwater amoebae; Hartmannella; Hartmannellid amoebae are an unnatural assemblage of amoeboid organisms mitochondrial morphology; SSU rDNA; SSU that are morphologically difficult to discern from one another. In molecular phy- rRNA; terrestrial amoebae; tubulinid. logenetic trees of the nuclear-encoded small subunit rDNA, they occupy at least five lineages within Tubulinea, a well-supported clade in Amoebozoa. The Correspondence polyphyletic nature of the hartmannellids has led to many taxonomic problems, M.W. Brown, Department of Biological in particular paraphyletic genera. Recent taxonomic revisions have alleviated Sciences, Mississippi State University, some of the problems. However, the genus Saccamoeba is paraphyletic and is Mississippi State, MS 39762, USA still in need of revision as it currently occupies two distinct lineages. Here, we Telephone number: +1 662-325-2406; report a new clade on the tree of Tubulinea, which we infer represents a novel FAX number: +1 662-325-7939; genus that we name Ptolemeba n. gen. This genus subsumes a clade of hart- e-mail: [email protected] mannellid amoebae that were previously considered in the genus Saccamoeba, but whose mitochondrial morphology is distinct from Saccamoeba. -

Heterospory: the Most Iterative Key Innovation in the Evolutionary History of the Plant Kingdom
Biol. Rej\ (1994). 69, l>p. 345-417 345 Printeii in GrenI Britain HETEROSPORY: THE MOST ITERATIVE KEY INNOVATION IN THE EVOLUTIONARY HISTORY OF THE PLANT KINGDOM BY RICHARD M. BATEMAN' AND WILLIAM A. DiMlCHELE' ' Departments of Earth and Plant Sciences, Oxford University, Parks Road, Oxford OXi 3P/?, U.K. {Present addresses: Royal Botanic Garden Edinburiih, Inverleith Rojv, Edinburgh, EIIT, SLR ; Department of Geology, Royal Museum of Scotland, Chambers Street, Edinburgh EHi ijfF) '" Department of Paleohiology, National Museum of Natural History, Smithsonian Institution, Washington, DC^zo^bo, U.S.A. CONTENTS I. Introduction: the nature of hf^terospon' ......... 345 U. Generalized life history of a homosporous polysporangiophyle: the basis for evolutionary excursions into hetcrospory ............ 348 III, Detection of hcterospory in fossils. .......... 352 (1) The need to extrapolate from sporophyte to gametophyte ..... 352 (2) Spatial criteria and the physiological control of heterospory ..... 351; IV. Iterative evolution of heterospory ........... ^dj V. Inter-cladc comparison of levels of heterospory 374 (1) Zosterophyllopsida 374 (2) Lycopsida 374 (3) Sphenopsida . 377 (4) PtiTopsida 378 (5) f^rogymnospermopsida ............ 380 (6) Gymnospermopsida (including Angiospermales) . 384 (7) Summary: patterns of character acquisition ....... 386 VI. Physiological control of hetcrosporic phenomena ........ 390 VII. How the sporophyte progressively gained control over the gametophyte: a 'just-so' story 391 (1) Introduction: evolutionary antagonism between sporophyte and gametophyte 391 (2) Homosporous systems ............ 394 (3) Heterosporous systems ............ 39(1 (4) Total sporophytic control: seed habit 401 VIII. Summary .... ... 404 IX. .•Acknowledgements 407 X. References 407 I. I.NIRODUCTION: THE NATURE OF HETEROSPORY 'Heterospory' sensu lato has long been one of the most popular re\ie\v topics in organismal botany. -
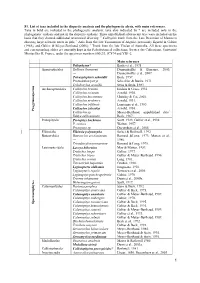
S1. List of Taxa Included in the Disparity Analysis and the Phylogenetic Alysis, with Main References
S1. List of taxa included in the disparity analysis and the phylogenetic alysis, with main references. Taxa in bold are included in the phylogenetic analysis; taxa also indicated by * are included only in the phylogenetic analysis and not in the disparity analysis. Three unpublished arborescent taxa were included on the basis that they showed additional anatomical diversity. 1 Callixylon trunk from the Late Devonian of Marrocco showing large sclerotic nests in pith; 2 Axis from the late Tournaisian of Algeria, previously figured in Galtier (1988), and Galtier & Meyer-Berthaud (2006); 3 Trunk from the late Viséan of Australia. All these specimens and corresponding slides are currently kept in the Paleobotanical collections, Service des Collections, Université Montpellier II, France, under the specimen numbers 600/2/3, JC874 and YB1-2. Main reference Psilophyton* Banks et al., 1975 Aneurophytales Rellimia thomsonii Dannenhoffer & Bonamo, 2003; --- Dannenhoffer et al., 2007. Tetraxylopteris schmidtii Beck, 1957. Proteokalon petryi Scheckler & Banks, 1971. Triloboxylon arnoldii Stein & Beck, 1983. s m Archaeopteridales Callixylon brownii Hoskin & Cross, 1951. r e Callixylon erianum Arnold, 1930. p s o Callixylon huronensis Chitaley & Cai, 2001. n Callixylon newberry Arnold, 1931. m y g Callixylon trifilievii Lemoigne et al., 1983. o r Callixylon zalesskyi Arnold, 1930. P Callixylon sp. Meyer-Berthaud, unpublished data1. Eddya sullivanensis Beck, 1967. Protopityales Protopitys buchiana Scott, 1923; Galtier et al., 1998. P. scotica Walton, 1957. Protopitys sp. Decombeix et al., 2005. Elkinsiales Elkinsia polymorpha Serbet & Rothwell, 1992. Buteoxylales Buteoxylon gordonianum Barnard &Long, 1973; Matten et al., --- 1980. Triradioxylon primaevum Barnard & Long, 1975. Lyginopteridales Laceya hibernica May & Matten, 1983. Tristichia longii Galtier, 1977. -

SYSTÉMATIQUE DES EMBRYOPHYTES Document Illustrant Plus Particulièrement Les Taxons Européens Version 4 Octobre 2007
Université Pierre et Marie Curie (PARIS 6) Préparation à l’agrégation SVSTU, secteur B SYSTÉMATIQUE DES EMBRYOPHYTES Document illustrant plus particulièrement les taxons européens version 4 octobre 2007 Catherine Reeb, Préparation agrégation SVSTU, UPMC Jean-Yves Dubuisson, Laboratoire Paléobotanique et Paléoécologie, équipe Paléodiversité, systématique et évolution des Embryophytes, UPMC Dessin de couverture : extrait d’un traité botanique tibétain Pour Garance Remerciements: merci à Jean et Monique Duperon pour les clés de détermination des familles d’Angiospermes, à A.M pour la reproduction de couverture, à Michaël Manuel et Eric Queinnec pour la partie I de l’introduction. Egalement à tous les illustrateurs qui ont autorisé l’utilisation de leurs documents mis en ligne sur Internet : Françoise Gantet, Prof. I. Foissner, Association Endemia de Nouvelle Calédonie. 1 Table des matières Introduction 3 Les données essentielles pour l’agrégation . 5 I Systématique générale des Embryophytes 6 I.1 Caractères des Embryophytes . 6 I.2 La place des Embryophytes au sein de la lignée verte . 7 I.3 Le groupe frère des Embryophytes . 7 I.4 Relations au sein des Embryophytes :les points acquis aujourd’hui . 8 I.4.1 Marchantiophytes, Bryophytes, Anthocérotophytes à la base des Embryophytes . 8 I.4.2 Les Trachéophytes ou plantes vasculaires, un groupe monophylétique . 9 I.4.3 Les Spermatophytes ou plantes à ovules, un groupe monophylétique . 9 I.5 Quelques points encore en discussion . 11 I.5.1 Position et relations des trois clades basaux (Bryophytes, Marchantiophytes et Anthocérotophytes) 11 I.5.2 Relations au sein des Spermatophytes :l’hypothèse "Gnepine" . 13 II Les principaux clades d’Embryophytes 15 II.1 MARCHANTIOPHYTA:Les Marchantiophytes ou Hépatiques . -

Earliest Record of Megaphylls and Leafy Structures, and Their Initial Diversification
Review Geology August 2013 Vol.58 No.23: 27842793 doi: 10.1007/s11434-013-5799-x Earliest record of megaphylls and leafy structures, and their initial diversification HAO ShouGang* & XUE JinZhuang Key Laboratory of Orogenic Belts and Crustal Evolution, School of Earth and Space Sciences, Peking University, Beijing 100871, China Received January 14, 2013; accepted February 26, 2013; published online April 10, 2013 Evolutionary changes in the structure of leaves have had far-reaching effects on the anatomy and physiology of vascular plants, resulting in morphological diversity and species expansion. People have long been interested in the question of the nature of the morphology of early leaves and how they were attained. At least five lineages of euphyllophytes can be recognized among the Early Devonian fossil plants (Pragian age, ca. 410 Ma ago) of South China. Their different leaf precursors or “branch-leaf com- plexes” are believed to foreshadow true megaphylls with different venation patterns and configurations, indicating that multiple origins of megaphylls had occurred by the Early Devonian, much earlier than has previously been recognized. In addition to megaphylls in euphyllophytes, the laminate leaf-like appendages (sporophylls or bracts) occurred independently in several dis- tantly related Early Devonian plant lineages, probably as a response to ecological factors such as high atmospheric CO2 concen- trations. This is a typical example of convergent evolution in early plants. Early Devonian, euphyllophyte, megaphyll, leaf-like appendage, branch-leaf complex Citation: Hao S G, Xue J Z. Earliest record of megaphylls and leafy structures, and their initial diversification. Chin Sci Bull, 2013, 58: 27842793, doi: 10.1007/s11434- 013-5799-x The origin and evolution of leaves in vascular plants was phology and evolutionary diversification of early leaves of one of the most important evolutionary events affecting the basal euphyllophytes remain enigmatic. -

Embryophytic Sporophytes in the Rhynie and Windyfield Cherts
Transactions of the Royal Society of Edinburgh: Earth Sciences http://journals.cambridge.org/TRE Additional services for Transactions of the Royal Society of Edinburgh: Earth Sciences: Email alerts: Click here Subscriptions: Click here Commercial reprints: Click here Terms of use : Click here Embryophytic sporophytes in the Rhynie and Windyeld cherts Dianne Edwards Transactions of the Royal Society of Edinburgh: Earth Sciences / Volume 94 / Issue 04 / December 2003, pp 397 - 410 DOI: 10.1017/S0263593300000778, Published online: 26 July 2007 Link to this article: http://journals.cambridge.org/abstract_S0263593300000778 How to cite this article: Dianne Edwards (2003). Embryophytic sporophytes in the Rhynie and Windyeld cherts. Transactions of the Royal Society of Edinburgh: Earth Sciences, 94, pp 397-410 doi:10.1017/S0263593300000778 Request Permissions : Click here Downloaded from http://journals.cambridge.org/TRE, IP address: 131.251.254.13 on 25 Feb 2014 Transactions of the Royal Society of Edinburgh: Earth Sciences, 94, 397–410, 2004 (for 2003) Embryophytic sporophytes in the Rhynie and Windyfield cherts Dianne Edwards ABSTRACT: Brief descriptions and comments on relationships are given for the seven embryo- phytic sporophytes in the cherts at Rhynie, Aberdeenshire, Scotland. They are Rhynia gwynne- vaughanii Kidston & Lang, Aglaophyton major D. S. Edwards, Horneophyton lignieri Barghoorn & Darrah, Asteroxylon mackiei Kidston & Lang, Nothia aphylla Lyon ex Høeg, Trichopherophyton teuchansii Lyon & Edwards and Ventarura lyonii Powell, Edwards & Trewin. The superb preserva- tion of the silica permineralisations produced in the hot spring environment provides remarkable insights into the anatomy of early land plants which are not available from compression fossils and other modes of permineralisation. -

Appendix 1. Systematic Arrangement of the Native Vascular Plants of Mexico
570 J.L. Villase˜nor / Revista Mexicana de Biodiversidad 87 (2016) 559–902 Appendix 1. Systematic arrangement of the native vascular plants of Mexico. The number of the families corresponds to the linear arrangement proposed by APG III (2009), Chase and Reveal (2009), Christenhusz, Chun, et al. (2011), Christenhusz, Reveal, et al. (2011), Haston et al. (2009) and Wearn et al. (2013). In parentheses, the first number indicates the number of genera and the second the number of species recorded for the family in Mexico Ferns and Lycophytes Order Cyatheales 12. Taxaceae (1/1) 17. Culcitaceae (1/1) Angiosperms Lycophytes 18. Plagiogyriaceae (1/1) 19. Cibotiaceae (1/2) Superorder Nymphaeanae Subclass Lycopodiidae 20. Cyatheaceae (3/14) 21. Dicksoniaceae (2/2) Orden Nymphaeales Order Lycopodiales 22. Metaxyaceae (1/1) 3. Cabombaceae (2/2) 1. Lycopodiaceae (4/21) 4. Nymphaeaceae (2/12) Order Polypodiales Order Isoetales 23. Lonchitidaceae (1/1) Superorder Austrobaileyanae 2. Isoetaceae (1/7) 24. Saccolomataceae (1/2) 26. Lindsaeaceae (3/8) Order Selaginellalles Orden Austrobaileyales 27. Dennstaedtiaceae (4/23) 3. Selaginellaceae (1/79) 7. Schisandraceae (2/2) 28. Pteridaceae (33/214) 29. Cystopteridaceae (1/4) Pteridophytes Superorder Chloranthanae 30. Aspleniaceae (4/89) 31. Diplaziopsidaceae (1/1) Subclass Equisetidae Orden Chloranthales 32. Thelypteridaceae (1/70) 8. Chloranthaceae (1/1) 33. Woodsiaceae (1/8) Order Equisetales 35. Onocleaceae (1/1) 1. Equisetaceae (1/6) Superorder Magnolianae 36. Blechnaceae (2/20) 37. Athyriaceae (2/31) Subclass Ophioglossidae Orden Canellales 38. Hypodematiaceae (1/1) 9. Canellaceae (1/1) Order Ophioglossales 39. Dryopteridaceae 10. Winteraceae (1/1) 2. Ophioglossaceae (2/16) (14/159) 40. -

THE EVOLUTION of XYLEM ANATOMY in EARLY TRACHEOPHYTES by ELISABETH ANNE BERGMAN
Conquering the terrestrial environment: the evolution of xylem anatomy in early tracheophytes Item Type text; Electronic Thesis Authors Bergman, Elisabeth Anne Publisher The University of Arizona. Rights Copyright © is held by the author. Digital access to this material is made possible by the University Libraries, University of Arizona. Further transmission, reproduction or presentation (such as public display or performance) of protected items is prohibited except with permission of the author. Download date 27/09/2021 03:01:29 Item License http://rightsstatements.org/vocab/InC/1.0/ Link to Item http://hdl.handle.net/10150/626731 CONQUERING THE TERRESTRIAL ENVIRONMENT: THE EVOLUTION OF XYLEM ANATOMY IN EARLY TRACHEOPHYTES By ELISABETH ANNE BERGMAN ____________________ A Thesis Submitted to The Honors College In Partial Fulfillment of the Bachelors Degree With Honors in Biology with an Emphasis in Biomedical Sciences THE UNIVERSITY OF ARIZONA D E C E M B E R 2 0 1 7 Approved by: ____________________________ Dr. Brian Enquist Department of Ecology and Evolutionary Biology Acknowledgements Many thanks go to all of those who made contributions, big and small, to my honors thesis, and more notably, my education. Foremost, I thank Dr. Brian Enquist for accepting me into his lab and serving as my mentor for two years. I appreciate all of the time he put in to meet with me and help me to develop my honors thesis. Additional thanks go to Dr. Sean Michaletz who first introduced me to the work that would eventually become my honors thesis. From the University of Santa Cruz, California, I thank Dr.