Antimony(V) Fluoride
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Seymur Cahangirov Hasan Sahin Guy Le Lay Angel Rubio Introduction to the Physics of Silicene and Other 2D Materials Lecture Notes in Physics
Lecture Notes in Physics 930 Seymur Cahangirov Hasan Sahin Guy Le Lay Angel Rubio Introduction to the Physics of Silicene and other 2D Materials Lecture Notes in Physics Volume 930 Founding Editors W. Beiglböck J. Ehlers K. Hepp H. Weidenmöller Editorial Board M. Bartelmann, Heidelberg, Germany B.-G. Englert, Singapore, Singapore P. Hanggi,R Augsburg, Germany M. Hjorth-Jensen, Oslo, Norway R.A.L. Jones, Sheffield, UK M. Lewenstein, Barcelona, Spain H. von Lohneysen,R Karlsruhe, Germany J.-M. Raimond, Paris, France A. Rubio, Hamburg, Germany M. Salmhofer, Heidelberg, Germany W. Schleich, Ulm, Germany S. Theisen, Potsdam, Germany D. Vollhardt, Augsburg, Germany J.D. Wells, Ann Arbor, USA G.P. Zank, Huntsville, USA [email protected] The Lecture Notes in Physics The series Lecture Notes in Physics (LNP), founded in 1969, reports new devel- opments in physics research and teaching-quickly and informally, but with a high quality and the explicit aim to summarize and communicate current knowledge in an accessible way. Books published in this series are conceived as bridging material between advanced graduate textbooks and the forefront of research and to serve three purposes: • to be a compact and modern up-to-date source of reference on a well-defined topic • to serve as an accessible introduction to the field to postgraduate students and nonspecialist researchers from related areas • to be a source of advanced teaching material for specialized seminars, courses and schools Both monographs and multi-author volumes will be considered for publication. Edited volumes should, however, consist of a very limited number of contributions only. -

UNITED STATES PATENT OFFICE 2,499,833 METEOD of MARING FLUORNATED ORGANIC COMPOUNDS Melvin A
Patented Mar. 7, 1950 2,499,833 UNITED STATES PATENT OFFICE 2,499,833 METEOD OF MARING FLUORNATED ORGANIC COMPOUNDS Melvin A. Perkins, Wilmington, Del, assignor to the United States of America as represented by the United States Atomic Energy Commission No Drawing. Application July 2, 1944, Serial No. 546,916 3 Claims. (C. 260-648) 2 This invention relates to new halogen com silver difluoride to replace hydrogen and saturate pounds and to methods for their production. any residual double bonds. It is an object of the invention to provide prod The products have densities in the neighbor lucts of high thermal stability and inertness to hood of 1.8 and refractive indices of about 1.3 chemical reagents, particularly oxidizing agents, s (sodium-D-line) at 23° C. They are usually ob acids, bases and metals. A further object is the tained in the crude state as mixtures of compounds provision of products suitable for use as heat of the molecular formulae CaF16, CaClF15, CaCl2F14 transfer media, lubricants and Sealants. Further and other chloro-fluorocarbons, from which the objects will appear from the following detailed individual constituents may be obtained by frac description of the invention. tional distillation. The products possess a high In my United States patent application Serial degree of stability to oxidizing agents, acids, bases Number 546,915 entitled "Halogenated organic and metals, and the stability increases with the compounds and method of making,' filed on the degree of fluorination. Thus the compound CaF16 Same date herewith, which became U. S. Patent is the most stable compound of the group and the No. -

Chemical Name Federal P Code CAS Registry Number Acutely
Acutely / Extremely Hazardous Waste List Federal P CAS Registry Acutely / Extremely Chemical Name Code Number Hazardous 4,7-Methano-1H-indene, 1,4,5,6,7,8,8-heptachloro-3a,4,7,7a-tetrahydro- P059 76-44-8 Acutely Hazardous 6,9-Methano-2,4,3-benzodioxathiepin, 6,7,8,9,10,10- hexachloro-1,5,5a,6,9,9a-hexahydro-, 3-oxide P050 115-29-7 Acutely Hazardous Methanimidamide, N,N-dimethyl-N'-[2-methyl-4-[[(methylamino)carbonyl]oxy]phenyl]- P197 17702-57-7 Acutely Hazardous 1-(o-Chlorophenyl)thiourea P026 5344-82-1 Acutely Hazardous 1-(o-Chlorophenyl)thiourea 5344-82-1 Extremely Hazardous 1,1,1-Trichloro-2, -bis(p-methoxyphenyl)ethane Extremely Hazardous 1,1a,2,2,3,3a,4,5,5,5a,5b,6-Dodecachlorooctahydro-1,3,4-metheno-1H-cyclobuta (cd) pentalene, Dechlorane Extremely Hazardous 1,1a,3,3a,4,5,5,5a,5b,6-Decachloro--octahydro-1,2,4-metheno-2H-cyclobuta (cd) pentalen-2- one, chlorecone Extremely Hazardous 1,1-Dimethylhydrazine 57-14-7 Extremely Hazardous 1,2,3,4,10,10-Hexachloro-6,7-epoxy-1,4,4,4a,5,6,7,8,8a-octahydro-1,4-endo-endo-5,8- dimethanonaph-thalene Extremely Hazardous 1,2,3-Propanetriol, trinitrate P081 55-63-0 Acutely Hazardous 1,2,3-Propanetriol, trinitrate 55-63-0 Extremely Hazardous 1,2,4,5,6,7,8,8-Octachloro-4,7-methano-3a,4,7,7a-tetra- hydro- indane Extremely Hazardous 1,2-Benzenediol, 4-[1-hydroxy-2-(methylamino)ethyl]- 51-43-4 Extremely Hazardous 1,2-Benzenediol, 4-[1-hydroxy-2-(methylamino)ethyl]-, P042 51-43-4 Acutely Hazardous 1,2-Dibromo-3-chloropropane 96-12-8 Extremely Hazardous 1,2-Propylenimine P067 75-55-8 Acutely Hazardous 1,2-Propylenimine 75-55-8 Extremely Hazardous 1,3,4,5,6,7,8,8-Octachloro-1,3,3a,4,7,7a-hexahydro-4,7-methanoisobenzofuran Extremely Hazardous 1,3-Dithiolane-2-carboxaldehyde, 2,4-dimethyl-, O- [(methylamino)-carbonyl]oxime 26419-73-8 Extremely Hazardous 1,3-Dithiolane-2-carboxaldehyde, 2,4-dimethyl-, O- [(methylamino)-carbonyl]oxime. -
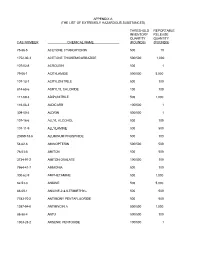
The List of Extremely Hazardous Substances)
APPENDIX A (THE LIST OF EXTREMELY HAZARDOUS SUBSTANCES) THRESHOLD REPORTABLE INVENTORY RELEASE QUANTITY QUANTITY CAS NUMBER CHEMICAL NAME (POUNDS) (POUNDS) 75-86-5 ACETONE CYANOHYDRIN 500 10 1752-30-3 ACETONE THIOSEMICARBAZIDE 500/500 1,000 107-02-8 ACROLEIN 500 1 79-06-1 ACRYLAMIDE 500/500 5,000 107-13-1 ACRYLONITRILE 500 100 814-68-6 ACRYLYL CHLORIDE 100 100 111-69-3 ADIPONITRILE 500 1,000 116-06-3 ALDICARB 100/500 1 309-00-2 ALDRIN 500/500 1 107-18-6 ALLYL ALCOHOL 500 100 107-11-9 ALLYLAMINE 500 500 20859-73-8 ALUMINUM PHOSPHIDE 500 100 54-62-6 AMINOPTERIN 500/500 500 78-53-5 AMITON 500 500 3734-97-2 AMITON OXALATE 100/500 100 7664-41-7 AMMONIA 500 100 300-62-9 AMPHETAMINE 500 1,000 62-53-3 ANILINE 500 5,000 88-05-1 ANILINE,2,4,6-TRIMETHYL- 500 500 7783-70-2 ANTIMONY PENTAFLUORIDE 500 500 1397-94-0 ANTIMYCIN A 500/500 1,000 86-88-4 ANTU 500/500 100 1303-28-2 ARSENIC PENTOXIDE 100/500 1 THRESHOLD REPORTABLE INVENTORY RELEASE QUANTITY QUANTITY CAS NUMBER CHEMICAL NAME (POUNDS) (POUNDS) 1327-53-3 ARSENOUS OXIDE 100/500 1 7784-34-1 ARSENOUS TRICHLORIDE 500 1 7784-42-1 ARSINE 100 100 2642-71-9 AZINPHOS-ETHYL 100/500 100 86-50-0 AZINPHOS-METHYL 10/500 1 98-87-3 BENZAL CHLORIDE 500 5,000 98-16-8 BENZENAMINE, 3-(TRIFLUOROMETHYL)- 500 500 100-14-1 BENZENE, 1-(CHLOROMETHYL)-4-NITRO- 500/500 500 98-05-5 BENZENEARSONIC ACID 10/500 10 3615-21-2 BENZIMIDAZOLE, 4,5-DICHLORO-2-(TRI- 500/500 500 FLUOROMETHYL)- 98-07-7 BENZOTRICHLORIDE 100 10 100-44-7 BENZYL CHLORIDE 500 100 140-29-4 BENZYL CYANIDE 500 500 15271-41-7 BICYCLO[2.2.1]HEPTANE-2-CARBONITRILE,5- -

United States Patent Office Patented Sept
2,904,588 United States Patent Office Patented Sept. 15, 1959 - 2 m Three grams of the product prepared as described above was placed in a polyethylene bottle and 6 g. of water 2,904,588 was added. After the initial exothermic reaction had FLUOROPHOSPHORANES AND THEIR subsided and the solution had cooled, a white crystalline PREPARATION solid separated. This solid was recrystallized twice from water. After drying, the product melted at 159 to 161 William C. Smith, Wilmington, Del, assignor to E. I. du C. The melting point of benzenephosphonic acid is 159 Pont de Nemours and Company, Wilmington, Del., a to 161° C., and this is the product expected from the com corporation of Delaware plete hydrolysis of phenyltetrafluorophosphorane. No Drawing. Application March 26, 1956 O Nuclear magnetic resonance examination of the phenyl Serial No. 573,659 tetrafluorophosphorane showed it to have four equiv alent fluorine atoms bound to phosphorus, which indi 15 Claims. (C. 260-543) cated a square pyramidal structure. This invention relates to new compositions of matter 5 EXAMPLE II and to their preparation. Example I was repeated, using a charge consisting Organic fluorine compounds have attained considerable of 53.7 g (0.3 mole) of phenylphosphonous dichloride importance in recent years and simple and economic in the reaction flask and 65.1 g (0.3 mole) of antimony methods for their preparation are greatly desired. pentafluoride in the dropping funnel. The antimony This invention has as an object the preparation of new 20 pentafluoride was added to the phenylphosphonous di fluorophosphoranes. A further object is the provision of chloride at such a rate that the temperature of the reac a new process for their preparation. -

Acutely / Extremely Hazardous Waste List
Acutely / Extremely Hazardous Waste List Federal P CAS Registry Acutely / Extremely Chemical Name Code Number Hazardous 4,7-Methano-1H-indene, 1,4,5,6,7,8,8-heptachloro-3a,4,7,7a-tetrahydro- P059 76-44-8 Acutely Hazardous 6,9-Methano-2,4,3-benzodioxathiepin, 6,7,8,9,10,10- hexachloro-1,5,5a,6,9,9a-hexahydro-, 3-oxide P050 115-29-7 Acutely Hazardous Methanimidamide, N,N-dimethyl-N'-[2-methyl-4-[[(methylamino)carbonyl]oxy]phenyl]- P197 17702-57-7 Acutely Hazardous 1-(o-Chlorophenyl)thiourea P026 5344-82-1 Acutely Hazardous 1-(o-Chlorophenyl)thiourea 5344-82-1 Extemely Hazardous 1,1,1-Trichloro-2, -bis(p-methoxyphenyl)ethane Extemely Hazardous 1,1a,2,2,3,3a,4,5,5,5a,5b,6-Dodecachlorooctahydro-1,3,4-metheno-1H-cyclobuta (cd) pentalene, Dechlorane Extemely Hazardous 1,1a,3,3a,4,5,5,5a,5b,6-Decachloro--octahydro-1,2,4-metheno-2H-cyclobuta (cd) pentalen-2- one, chlorecone Extemely Hazardous 1,1-Dimethylhydrazine 57-14-7 Extemely Hazardous 1,2,3,4,10,10-Hexachloro-6,7-epoxy-1,4,4,4a,5,6,7,8,8a-octahydro-1,4-endo-endo-5,8- dimethanonaph-thalene Extemely Hazardous 1,2,3-Propanetriol, trinitrate P081 55-63-0 Acutely Hazardous 1,2,3-Propanetriol, trinitrate 55-63-0 Extemely Hazardous 1,2,4,5,6,7,8,8-Octachloro-4,7-methano-3a,4,7,7a-tetra- hydro- indane Extemely Hazardous 1,2-Benzenediol, 4-[1-hydroxy-2-(methylamino)ethyl]- 51-43-4 Extemely Hazardous 1,2-Benzenediol, 4-[1-hydroxy-2-(methylamino)ethyl]-, P042 51-43-4 Acutely Hazardous 1,2-Dibromo-3-chloropropane 96-12-8 Extemely Hazardous 1,2-Propylenimine P067 75-55-8 Acutely Hazardous 1,2-Propylenimine 75-55-8 Extemely Hazardous 1,3,4,5,6,7,8,8-Octachloro-1,3,3a,4,7,7a-hexahydro-4,7-methanoisobenzofuran Extemely Hazardous 1,3-Dithiolane-2-carboxaldehyde, 2,4-dimethyl-, O- [(methylamino)-carbonyl]oxime 26419-73-8 Extemely Hazardous 1,3-Dithiolane-2-carboxaldehyde, 2,4-dimethyl-, O- [(methylamino)-carbonyl]oxime. -

0 2 0 Material Safety Data Sheet
He a lt h 2 0 Fire 0 2 0 Re a c t iv it y 0 Pe rs o n a l Pro t e c t io n E Material Safety Data Sheet Potassium nitrate MSDS Section 1: Chemical Product and Company Identification Product Name: Potassium nitrate Contact Information: Catalog Codes: SLP4009, SLP3001, SLP4816, SLP1987 Sciencelab.com, Inc. 14025 Smith Rd. CAS#: 7757-79-1 Houston, Texas 77396 RTECS: TT370000 US Sales: 1-800-901-7247 International Sales: 1-281-441-4400 TSCA: TSCA 8(b) inventory: Potassium nitrate Order Online: ScienceLab.com CI#: Not available. CHEMTREC (24HR Emergency Telephone), call: Synonym: Nitric Acid, potassium salt; saltpeter 1-800-424-9300 Chemical Name: Potassium Nitrate International CHEMTREC, call: 1-703-527-3887 Chemical Formula: KNO3 For non-emergency assistance, call: 1-281-441-4400 Section 2: Composition and Information on Ingredients Composition: Name CAS # % by Weight Potassium nitrate 7757-79-1 100 Toxicological Data on Ingredients: Potassium nitrate: ORAL (LD50): Acute: 3750 mg/kg [Rat]. 1901 mg/kg [Rabbit]. Section 3: Hazards Identification Potential Acute Health Effects: Hazardous in case of skin contact (irritant), of eye contact (irritant), of ingestion, of inhalation (lung irritant). Prolonged exposure may result in skin burns and ulcerations. Over-exposure by inhalation may cause respiratory irritation. Potential Chronic Health Effects: CARCINOGENIC EFFECTS: Not available. MUTAGENIC EFFECTS: Not available. TERATOGENIC EFFECTS: Not available. DEVELOPMENTAL TOXICITY: Not available. The substance may be toxic to blood, kidneys, central nervous system (CNS). Repeated or prolonged exposure to the substance can produce target organs damage. Section 4: First Aid Measures Eye Contact: Check for and remove any contact lenses. -

List of Lists
United States Office of Solid Waste EPA 550-B-10-001 Environmental Protection and Emergency Response May 2010 Agency www.epa.gov/emergencies LIST OF LISTS Consolidated List of Chemicals Subject to the Emergency Planning and Community Right- To-Know Act (EPCRA), Comprehensive Environmental Response, Compensation and Liability Act (CERCLA) and Section 112(r) of the Clean Air Act • EPCRA Section 302 Extremely Hazardous Substances • CERCLA Hazardous Substances • EPCRA Section 313 Toxic Chemicals • CAA 112(r) Regulated Chemicals For Accidental Release Prevention Office of Emergency Management This page intentionally left blank. TABLE OF CONTENTS Page Introduction................................................................................................................................................ i List of Lists – Conslidated List of Chemicals (by CAS #) Subject to the Emergency Planning and Community Right-to-Know Act (EPCRA), Comprehensive Environmental Response, Compensation and Liability Act (CERCLA) and Section 112(r) of the Clean Air Act ................................................. 1 Appendix A: Alphabetical Listing of Consolidated List ..................................................................... A-1 Appendix B: Radionuclides Listed Under CERCLA .......................................................................... B-1 Appendix C: RCRA Waste Streams and Unlisted Hazardous Wastes................................................ C-1 This page intentionally left blank. LIST OF LISTS Consolidated List of Chemicals -

2020 Emergency Response Guidebook
2020 A guidebook intended for use by first responders A guidebook intended for use by first responders during the initial phase of a transportation incident during the initial phase of a transportation incident involving hazardous materials/dangerous goods involving hazardous materials/dangerous goods EMERGENCY RESPONSE GUIDEBOOK THIS DOCUMENT SHOULD NOT BE USED TO DETERMINE COMPLIANCE WITH THE HAZARDOUS MATERIALS/ DANGEROUS GOODS REGULATIONS OR 2020 TO CREATE WORKER SAFETY DOCUMENTS EMERGENCY RESPONSE FOR SPECIFIC CHEMICALS GUIDEBOOK NOT FOR SALE This document is intended for distribution free of charge to Public Safety Organizations by the US Department of Transportation and Transport Canada. This copy may not be resold by commercial distributors. https://www.phmsa.dot.gov/hazmat https://www.tc.gc.ca/TDG http://www.sct.gob.mx SHIPPING PAPERS (DOCUMENTS) 24-HOUR EMERGENCY RESPONSE TELEPHONE NUMBERS For the purpose of this guidebook, shipping documents and shipping papers are synonymous. CANADA Shipping papers provide vital information regarding the hazardous materials/dangerous goods to 1. CANUTEC initiate protective actions. A consolidated version of the information found on shipping papers may 1-888-CANUTEC (226-8832) or 613-996-6666 * be found as follows: *666 (STAR 666) cellular (in Canada only) • Road – kept in the cab of a motor vehicle • Rail – kept in possession of a crew member UNITED STATES • Aviation – kept in possession of the pilot or aircraft employees • Marine – kept in a holder on the bridge of a vessel 1. CHEMTREC 1-800-424-9300 Information provided: (in the U.S., Canada and the U.S. Virgin Islands) • 4-digit identification number, UN or NA (go to yellow pages) For calls originating elsewhere: 703-527-3887 * • Proper shipping name (go to blue pages) • Hazard class or division number of material 2. -

Antimony Pentafluoride Apf
ANTIMONY PENTAFLUORIDE APF CAUTIONARY RESPONSE INFORMATION 4. FIRE HAZARDS 7. SHIPPING INFORMATION 4.1 Flash Point: 7.1 Grades of Purity: Commercial Common Synonyms Liquid Colorless Sharp odor Not flammable 7.2 Storage Temperature: Ambient 4.2 Flammable Limits in Air: Not flammable 7.3 Inert Atmosphere: No requirement 4.3 Fire Extinguishing Agents: Not pertinent Reacts violently with water. Poisonous gas is produced on contact 7.4 Venting: Pressure-vacuum with water. Freezing point is 45°F. 4.4 Fire Extinguishing Agents Not to Be Used: Do not use water or foam on 7.5 IMO Pollution Category: Currently not available AVOID CONTACT WITH LIQUID AND VAPOR. KEEP PEOPLE AWAY. adjacent fires. 7.6 Ship Type: Currently not available Wear goggles and self-contained breathing apparatus. 4.5 Special Hazards of Combustion 7.7 Barge Hull Type: Currently not available Stop discharge if possible. Products: Not pertinent Call fire department. 4.6 Behavior in Fire: Gives off toxic hydrogen Evacuate. 8. HAZARD CLASSIFICATIONS fluoride fumes when water is used to Isolate and remove discharged material. extinguish adjacent fire. 8.1 49 CFR Category: Corrosive material Notify local health and pollution control agencies. Protect water intakes. 4.7 Auto Ignition Temperature: Not pertinent 8.2 49 CFR Class: 8 4.8 Electrical Hazards: Not pertinent 8.3 49 CFR Package Group: II Not flammable. 4.9 Burning Rate: Not pertinent 8.4 Marine Pollutant: No Fire May cause fire on contact with combustibles. 4.10 Adiabatic Flame Temperature: Not 8.5 NFPA Hazard Classification: POISONOUS GASES ARE PRODUCED WHEN HEATED. -

Jeffrey E. Lucius U.S. Geological Survey This Report Is Preliminary
U. S. DEPARTMENT OF THE INTERIOR GEOLOGICAL SURVEY PHYSICAL AND CHEMICAL PROPERTIES AND HEALTH EFFECTS OF THIRTY-THREE TOXIC ORGANIC CHEMICALS Jeffrey E. Lucius U.S. Geological Survey Open-File Report 87-428 August 1987 This report is preliminary and has not been reviewed for conformity with U.S. Geological Survey editorial standards. Any use of trade names is for descriptive purposes only and does not imply endorsement by the U.S. Geological Survey. CONTENTS Page Introduction 4 The Properties 6 Abbreviations 13 Conversion Factors 16 Summary Tables 17 Acetic acid 27 Acetone 31 Benzene 34 Bis(2-ethylhexyl)phthalate 37 Bromoform 39 Carbon tetrachloride 42 Chlorobenzene 46 Chloroethane 49 Chloroform 52 Cyclohexane 55 Di-n-butyl phthalate 58 1.1-Dichloroethane 60 1.2-Dichloroethane 63 1,1-Dichloroethene 66 trans-1,2-Dichloroethene 69 Dimethyl sulfoxide 71 1,4-Dioxane 74 Ethanol 77 Ethylbenzene 81 Ethylene dibromide 84 Methanol 87 Methylene chloride 91 Naphthalene 94 Phenol 97 Quinoline 101 Tetrachloroethene 103 Toluene 106 1,1,1-Trichloroethane 109 Trichloroethene 112 Vinyl chloride 115 Water 118 m-Xylene 121 o-Xylene 124 p-Xylene 127 References and Bibliography 130 SUMMARY TABLES page 1. Ranking of top 20 organic ground water contaminants based on number of sites at which each contaminant was detected. 17 2. Selected toxic organic chemicals ordered by Chemical Abstract Service Registry Number (CAS RN). Those chemicals on the U.S. EPA top 100 hazardous substances list are also noted. 18 3. Selected toxic organic chemicals ordered by number of carbon and hydrogen atoms. 19 4. Ranking of selected toxic organic chemicals by specific gravity at room temperature. -

Hazardous Materials Descriptions and Codes
2012 Commodity Flow Survey Hazardous Materials Descriptions and Codes Hazardous Materials Descriptions and Proper Shipping Names UN or NA Code Accellerene, see p-Nitrosodimethylaniline Accumulators, electric, see Batteries, wet etc Accumulators, pressurized, pneumatic or hydraulic (containing non-flammable gas), see Articles pressurized, pneumatic or hydraulic (containing non-flammable gas) Acetal 1088 Acetaldehyde 1089 Acetaldehyde ammonia 1841 Acetaldehyde oxime 2332 Acetic acid, glacial or Acetic acid solution, with more than 80 percent acid, by mass 2789 Acetic acid solution, not less than 50 percent but not more than 80 percent acid, by 2790 mass Acetic acid solution, with more than 10 percent and less than 50 percent acid, by mass 2790 Acetic anhydride 1715 Acetone 1090 Acetone cyanohydrin, stabilized 1541 Acetone oils 1091 Acetonitrile 1648 Acetyl bromide 1716 Acetyl chloride 1717 Acetyl iodide 1898 Acetyl methyl carbinol 2621 Acetylene, dissolved 1001 Acetylene tetrabromide, see Tetrabromoethane Acid butyl phosphate, see Butyl acid phosphate Acid, sludge, see Sludge acid Acridine 2713 Acrolein dimer, stabilized 2607 Acrolein, stabilized 1092 Acrylamide, solid 2074 Acrylamide solution 3426 Acrylic acid, stabilized 2218 Acrylonitrile, stabilized 1093 Actuating cartridge, explosive, see Cartridges, power device Adhesives, containing a flammable liquid 1133 Adiponitrile 2205 Aerosols, poison, Packing Group III (each not exceeding 1 L capacity) 1950 Aerosols, flammable, (each not exceeding 1 L capacity) 1950 Source: Electronic Code of Federal Regulations http://ecfr.gpoaccess.gov/cgi/t/text/text- idx?c=ecfr&sid=dfec99df9f21ef29b59a8565433a56cd&rgn=div6&view=text&node=49:2.1.1.3.8.2&idno=49 1 Hazardous Materials Descriptions and Proper Shipping Names UN or NA Code Aerosols, flammable, n.o.s.