2019 Icar Program & Abstracts Book
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Signature Redacted Signature of Author
Developing VHH-based tools to study Ebolavirus Infection By Jason V. M. H. Nguyen B.S., Biochemistry and Molecular Biology University of California, Santa Cruz, 2012 Submitted to the Microbiology Graduate Program in partial fulfillment of the requirements for the degree of Doctor of Philosophy At the Massachusetts Institute of Technology June 2019 2019 Massachusetts Institute of Technology. All rights reserved. Signature redacted Signature of Author...... .................................................................... V Y' Jason V. M. H. Nguyen Microbiology Graduate Program May 13, 2019 Signature redacted Certified by.............. ................................................................... Hidde L. Ploegh Senior Investigator Boston Children's Hospital It Signature redacted Accepted by....... ......................................................................... Jacquin C. Niles Associate Professor of Biological Engineering Chair of Microbiology Program MASSACHUSETTS INSTITUTE OF TECHNOLOGY JUN 2 12019 1 LIBRARIES ARCHIVES 2 Developing VHH-based tools to study Ebolavirus Infection By Jason V. M. H. Nguyen Submitted to the Microbiology Graduate Program on May 13th, 2019 in partial fulfillment of the requirements for the degree of Doctor of Philosophy ABSTRACT Variable domains of camelid-derived heavy chain-only antibodies, or VHHs, have emerged as a unique antigen binding moiety that holds promise in its versatility and utilization as a tool to study biological questions. This thesis focuses on two aspects on developing tools to study infectious disease, specifically Ebolavirus entry. In Chapter 1, I provide an overview about antibodies and how antibodies have transformed the biomedical field and how single domain antibody fragments, or VHHs, have entered this arena. I will also touch upon how VHHs have been used in various fields and certain aspects that remain underexplored. Chapter 2 focuses on the utilization of VHHs to study Ebolavirus entry using VHHs that were isolated from alpacas. -

Infectious Diseases
2013 MEDICINES IN DEVELOPMENT REPORT Infectious Diseases A Report on Diseases Caused by Bacteria, Viruses, Fungi and Parasites PRESENTED BY AMERICA’S BIOPHARMACEUTICAL RESEARCH COMPANIES Biopharmaceutical Research Evolves Against Infectious Diseases with Nearly 400 Medicines and Vaccines in Testing Throughout history, infectious diseases hepatitis C that inhibits the enzyme have taken a devastating toll on the lives essential for viral replication. and well-being of people around the • An anti-malarial drug that has shown Medicines in Development world. Caused when pathogens such activity against Plasmodium falci- For Infectious Diseases as bacteria or viruses enter a body and parum malaria which is resistant to multiply, infectious diseases were the current treatments. Application leading cause of death in the United Submitted States until the 1920s. Today, vaccines • A potential new antibiotic to treat methicillin-resistant Staphylococcus Phase III and infectious disease treatments have proven to be effective treatments in aureus (MRSA). Phase II many cases, but infectious diseases still • A novel treatment that works by Phase I pose a very serious threat to patients. blocking the ability of the smallpox Recently, some infectious pathogens, virus to spread to other cells, thus 226 such as pseudomonas bacteria, have preventing it from causing disease. become resistant to available treatments. Infectious diseases may never be fully Diseases once considered conquered, eradicated. However, new knowledge, such as tuberculosis, have reemerged new technologies, and the continuing as a growing health threat. commitment of America’s biopharma- America’s biopharmaceutical research ceutical research companies can help companies are developing 394 medicines meet the continuing—and ever-changing and vaccines to combat the many threats —threat from infectious diseases. -
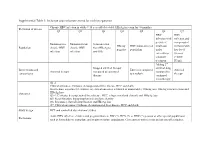
Inclusion and Exclusion Criteria for Each Key Question
Supplemental Table 1: Inclusion and exclusion criteria for each key question Chronic HBV infection in adults ≥ 18 year old (detectable HBsAg in serum for >6 months) Definition of disease Q1 Q2 Q3 Q4 Q5 Q6 Q7 HBV HBV infection with infection and persistent compensated Immunoactive Immunotolerant Seroconverted HBeAg HBV mono-infected viral load cirrhosis with Population chronic HBV chronic HBV from HBeAg to negative population under low level infection infection anti-HBe entecavir or viremia tenofovir (<2000 treatment IU/ml) Adding 2nd Stopped antiviral therapy antiviral drug Interventions and Entecavir compared Antiviral Antiviral therapy compared to continued compared to comparisons to tenofovir therapy therapy continued monotherapy Q1-2: Clinical outcomes: Cirrhosis, decompensated liver disease, HCC and death Intermediate outcomes (if evidence on clinical outcomes is limited or unavailable): HBsAg loss, HBeAg seroconversion and Outcomes HBeAg loss Q3-4: Cirrhosis, decompensated liver disease, HCC, relapse (viral and clinical) and HBsAg loss Q5: Renal function, hypophosphatemia and bone density Q6: Resistance, flare/decompensation and HBeAg loss Q7: Clinical outcomes: Cirrhosis, decompensated liver disease, HCC and death Study design RCT and controlled observational studies Acute HBV infection, children and pregnant women, HIV (+), HCV (+) or HDV (+) persons or other special populations Exclusions such as hemodialysis, transplant, and treatment failure populations. Co treatment with steroids and uncontrolled studies. Supplemental Table 2: Detailed Search Strategy: Ovid Database(s): Embase 1988 to 2014 Week 37, Ovid MEDLINE(R) In-Process & Other Non- Indexed Citations and Ovid MEDLINE(R) 1946 to Present, EBM Reviews - Cochrane Central Register of Controlled Trials August 2014, EBM Reviews - Cochrane Database of Systematic Reviews 2005 to July 2014 Search Strategy: # Searches Results 1 exp Hepatitis B/dt 26410 ("hepatitis B" or "serum hepatitis" or "hippie hepatitis" or "injection hepatitis" or 2 178548 "hepatitis type B").mp. -

Microorganisms-07-00521-V2.Pdf
microorganisms Review Are Community Acquired Respiratory Viral Infections an Underestimated Burden in Hematology Patients? Cristian-Marian Popescu 1,* , Aurora Livia Ursache 2, Gavriela Feketea 1 , Corina Bocsan 3, Laura Jimbu 1, Oana Mesaros 1, Michael Edwards 4,5, Hongwei Wang 6, Iulia Berceanu 7, Alexandra Neaga 1 and Mihnea Zdrenghea 1,7,* 1 Department of Hematology, Iuliu Hatieganu University of Medicine and Pharmacy, 8 Babes str., 400012 Cluj-Napoca, Romania; [email protected] (G.F.); [email protected] (L.J.); [email protected] (O.M.); [email protected] (A.N.) 2 The Faculty of Veterinary Medicine, University of Agricultural Sciences and Veterinary Medicine, Calea Mănăs, tur 3-5, 400372 Cluj-Napoca, Romania; [email protected] 3 Department of Pharmacology, Toxicology and Clinical Pharmacology, Iuliu Ha¸tieganuUniversity of Medicine and Pharmacy, 400337 Cluj-Napoca, Romania; [email protected] 4 National Heart and Lung Institute, St Mary’s Campus, Imperial College London, London W2 1PG, UK; [email protected] 5 Medical Research Council (MRC) and Asthma UK Centre in Allergic Mechanisms of Asthma, London W2 1PG, UK 6 School of Medicine, Nanjing University, 22 Hankou Road, Nanjing 210093, China; [email protected] 7 Department of Hematology, Ion Chiricuta Oncology Institute, 34-36 Republicii Street, 400015 Cluj-Napoca, Romania; [email protected] * Correspondence: [email protected] (C.-M.P.); [email protected] (M.Z.) Received: 8 October 2019; Accepted: 31 October 2019; Published: 2 November 2019 Abstract: Despite a plethora of studies demonstrating significant morbidity and mortality due to community-acquired respiratory viral (CRV) infections in intensively treated hematology patients, and despite the availability of evidence-based guidelines for the diagnosis and management of respiratory viral infections in this setting, there is no uniform inclusion of respiratory viral infection management in the clinical hematology routine. -

AHFS Pharmacologic-Therapeutic Classification System
AHFS Pharmacologic-Therapeutic Classification System Abacavir 48:24 - Mucolytic Agents - 382638 8:18.08.20 - HIV Nucleoside and Nucleotide Reverse Acitretin 84:92 - Skin and Mucous Membrane Agents, Abaloparatide 68:24.08 - Parathyroid Agents - 317036 Aclidinium Abatacept 12:08.08 - Antimuscarinics/Antispasmodics - 313022 92:36 - Disease-modifying Antirheumatic Drugs - Acrivastine 92:20 - Immunomodulatory Agents - 306003 4:08 - Second Generation Antihistamines - 394040 Abciximab 48:04.08 - Second Generation Antihistamines - 394040 20:12.18 - Platelet-aggregation Inhibitors - 395014 Acyclovir Abemaciclib 8:18.32 - Nucleosides and Nucleotides - 381045 10:00 - Antineoplastic Agents - 317058 84:04.06 - Antivirals - 381036 Abiraterone Adalimumab; -adaz 10:00 - Antineoplastic Agents - 311027 92:36 - Disease-modifying Antirheumatic Drugs - AbobotulinumtoxinA 56:92 - GI Drugs, Miscellaneous - 302046 92:20 - Immunomodulatory Agents - 302046 92:92 - Other Miscellaneous Therapeutic Agents - 12:20.92 - Skeletal Muscle Relaxants, Miscellaneous - Adapalene 84:92 - Skin and Mucous Membrane Agents, Acalabrutinib 10:00 - Antineoplastic Agents - 317059 Adefovir Acamprosate 8:18.32 - Nucleosides and Nucleotides - 302036 28:92 - Central Nervous System Agents, Adenosine 24:04.04.24 - Class IV Antiarrhythmics - 304010 Acarbose Adenovirus Vaccine Live Oral 68:20.02 - alpha-Glucosidase Inhibitors - 396015 80:12 - Vaccines - 315016 Acebutolol Ado-Trastuzumab 24:24 - beta-Adrenergic Blocking Agents - 387003 10:00 - Antineoplastic Agents - 313041 12:16.08.08 - Selective -

Interferon-Based and Interferon-Free New Treatment Options
Interferon-based and interferon-free new treatment options White Nights of Hepatology St. Petersburg, 7. June 2013 Christoph Sarrazin Klinikum der J. W. Goethe-Universität Medizinische Klinik I Frankfurt am Main Mode of action of Interferons natural immunomodulatory effects IFN-stimulated gene activation Antiviral Apoptotic Immunomodulatory activity activity activity B-cell proliferation Reduced transcription Elevates apoptosis by CTL proliferation Reduced translation multiple mechanisms MHC upregulation Reduced RNA stability Augments NK activity Host-mediated effects are important for DAA combination therapy • Potency and additive effects • Prevention of resistance and viral breakthrough Different types of Interferons Type I Interferons Type III Interferons Broad receptor Receptors distribution distributed throughout various primarily in body tissues epithelial cells and hepatocytes Antiviral effects Antiviral effects Adverse events of Type I IFNs treatment Peg‐Intron • Flu-like symptoms PegIFN‐2a Type III IFNs Potentially fewer • Haematologic IFN omega Peg‐IFN‐ adverse events disorders IFN‐alfa‐2b XL lambda than with type I • Psychiatric Belerofon (Peg‐rIL‐29) interferons symptoms Albuferon Locteron Adapted from 1. Marcello T et al. Gastroenterology 2006;131:1887–98; 2. Muir AJ et al. 2009 AASLD. Abstract 1591; 3. O'Brien TR. Nat Genet. 2009;41:1048–50. PEG-Interferon alfa / Ribavirin Approval studies: efficacy Approval study (n=1530) Approval study (n=1121) Therapy: IFN vs. PEG-IFN alfa 2b Therapy: IFN vs. PEG-IFN alfa 2a 1,0/1,5µg/kgKG -

'Spanish' Influenza
1918 ‘Spanish’ Influenza: Lessons from the Past Jeffery Taubenberger, M.D., Ph.D. May 1, 2012 Influenza A virus (IAV) Family: Orthomyxoviridae •Negative sense, segmented, single-stranded RNA genome •8 segments, 12-13 ORF’s NA NA (9 NA subtypes) Nomenclature: SA ααα2-6Gal e.g., H5N1, H3N2 HA SA ααα2-3Gal (17 HA subtypes) Modified from: Horimoto & Kawaoka (2005) Nat Rev Micro 3:591-600 “Shift and Drift” Influenza A virus host range is quite diverse ? Influenza A viruses in animals Influenza A viruses in humans Annual epidemics with >200,000 hospitalizations; & up to 49,000 deaths/yr. in U.S. Occasional and unpredictable pandemic strains with increase in illness and death Pandemics are derived zoonotically: 1918, 1957, 1968, 2009 Morens, et al . 2009 NEJM . 361:225-229 The influenza problem: Influenza New strains pandemics emerge continually emerge; unpredictably and host range is very have variable extensive characteristics Human influenza A timeline H1N1 H3N2 H3N? H2N2 H1N1 H1N1 1889 1918 1957 1977 2009 1968 N = 4 Mortality impacts of past influenza pandemics Influenza pandemics throughout history • At least 14 pandemics in last 500 years (since 1510) • Average inter-pandemic interval = 36 years • Severity variable Morens & Taubenberger 2011. Rev Med Virol 21:262 1918 ‘Spanish’ influenza mortality • Total deaths in the 9 months of the pandemic in 1918-1919 estimated to be 50 million • U.S. Deaths = 675,000 • U.S. Military deaths to flu = 43,000 (out of ~100,000 U.S. Troop casualties in W.W.I.) • Case fatality rate ~1-2%; >98% had self-limited -

Drug Repurposing for Identification of Potential Inhibitors Against SARS-Cov-2 Spike Receptor-Binding Domain: an in Silico Approach
Indian J Med Res 153, January & February 2021, pp 132-143 Quick Response Code: DOI: 10.4103/ijmr.IJMR_1132_20 Drug repurposing for identification of potential inhibitors against SARS-CoV-2 spike receptor-binding domain: An in silico approach Santosh Kumar Behera1, Namita Mahapatra1, Chandra Sekhar Tripathy1 & Sanghamitra Pati2 1Health Informatics Centre, 2ICMR-Regional Medical Research Centre, Bhubaneswar, Odisha, India Received April 10, 2020 Background & objectives: The world is currently under the threat of coronavirus disease 2019 (COVID-19) infection, caused by SARS-CoV-2. The objective of the present investigation was to repurpose the drugs with potential antiviral activity against receptor-binding domain (RBD) of SARS-CoV-2 spike (S) protein among 56 commercially available drugs. Therefore, an integrative computational approach, using molecular docking, quantum chemical calculation and molecular dynamics, was performed to unzip the effective drug-target interactions between RBD and 56 commercially available drugs. Methods: The present in silico approach was based on information of drugs and experimentally derived crystal structure of RBD of SARS-CoV-2 S protein. Molecular docking analysis was performed for RBD against all 56 reported drugs using AutoDock 4.2 tool to screen the drugs with better potential antiviral activity which were further analysed by other computational tools for repurposing potential drug or drugs for COVID-19 therapeutics. Results: Drugs such as chalcone, grazoprevir, enzaplatovir, dolutegravir, daclatasvir, tideglusib, presatovir, remdesivir and simeprevir were predicted to be potentially effective antiviral drugs against RBD and could have good COVID-19 therapeutic efficacy. Simeprevir displayed the highest binding affinity and reactivity against RBD with the values of −8.52 kcal/mol (binding energy) and 9.254 kcal/mol (band energy gap) among all the 56 drugs under investigation. -

Development of Novel Antiviral Therapies for Hepatitis C Virus Kai Lin** (Novartis Institutes for Biomedical Research, Inc
VIROLOGICA SINICA, August 2010, 25 (4):246-266 DOI 10.1007/s12250-010-3140-2 © Wuhan Institute of Virology, CAS and Springer-Verlag Berlin Heidelberg 2010 Development of Novel Antiviral Therapies for Hepatitis C Virus Kai Lin** (Novartis Institutes for BioMedical Research, Inc. Cambridge, Massachusetts 02139, USA) Abstract: Over 170 million people worldwide are infected with hepatitis C virus (HCV), a major cause of liver diseases. Current interferon-based therapy is of limited efficacy and has significant side effects and more effective and better tolerated therapies are urgently needed. HCV is a positive, single-stranded RNA virus with a 9.6 kb genome that encodes ten viral proteins. Among them, the NS3 protease and the NS5B polymerase are essential for viral replication and have been the main focus of drug discovery efforts. Aided by structure-based drug design, potent and specific inhibitors of NS3 and NS5B have been identified, some of which are in late stage clinical trials and may significantly improve current HCV treatment. Inhibitors of other viral targets such as NS5A are also being pursued. However, HCV is an RNA virus characterized by high replication and mutation rates and consequently, resistance emerges quickly in patients treated with specific antivirals as monotherapy. A complementary approach is to target host factors such as cyclophilins that are also essential for viral replication and may present a higher genetic barrier to resistance. Combinations of these inhibitors of different mechanism are likely to become the essential components of future HCV therapies in order to maximize antiviral efficacy and prevent the emergence of resistance. -

Development of Novel Anti-Respiratory Syncytial Virus Therapies
Development of Novel Anti-Respiratory Syncytial Virus Therapies by Michael J Norris A thesis submitted in conformity with the requirements for the degree of Doctor of Philosophy Laboratory Medicine and Pathobiology University of Toronto © Copyright by Michael J Norris 2017 Development of Novel Anti-Respiratory Syncytial Virus Therapies Michael J Norris Doctor of Philosophy Laboratory Medicine and Pathobiology University of Toronto 2017 Abstract Respiratory syncytial virus (RSV) is a leading cause of mortality in infants and young children. Despite the RSV disease burden, no vaccine is available and treatment remains non-specific. New drug candidates are needed to combat RSV. Towards this goal, we investigated two broad strategies to control RSV infection. First, we examined the utility of delivering an anti-4-1BB agonist antibody to enhance CD8 T cell costimulation in a mouse model of RSV infection. This strategy enhanced CD8 T cell numbers, however failed to reduce RSV titers in the lung. We found that this was likely due to reduced RSV specific CD8 T cells in the lung. Overall, this strategy did not attenuate RSV associated disease outcomes and was associated with increased morbidity. Second, we screened over 2000 FDA approved compounds to identify approved drugs with novel anti-RSV activity. Cardiac glycosides, inhibitors of the membrane bound Na+/K+-ATPase, were identified to have anti-RSV activity. Cardiac glycosides diminished RSV infection in HEp-2 cells and in primary human airway epithelial cells grown at an air liquid interface. Digoxin, an FDA approved cardiac glycoside, was further able to inhibit RSV infection of community isolates of RSV in primary nasal epithelial cells. -

Communication DOI: 10.5582/Ddt.2020.01012
58 Drug Discoveries & Therapeutics. 2020; 14(1):58-60. Communication DOI: 10.5582/ddt.2020.01012 Discovering drugs to treat coronavirus disease 2019 (COVID-19) Liying Dong1, Shasha Hu2, Jianjun Gao1,* 1 Department of Pharmacology, School of Pharmacy, Qingdao University, Qingdao, Shandong, China; 2 Department of Pathology, the Affiliated Hospital of Qingdao University, Qingdao, Shandong, China. SUMMARY The SARS-CoV-2 virus emerged in December 2019 and then spread rapidly worldwide, particularly to China, Japan, and South Korea. Scientists are endeavoring to find antivirals specific to the virus. Several drugs such as chloroquine, arbidol, remdesivir, and favipiravir are currently undergoing clinical studies to test their efficacy and safety in the treatment of coronavirus disease 2019 (COVID-19) in China; some promising results have been achieved thus far. This article summarizes agents with potential efficacy against SARS-CoV-2. Keywords novel coronavirus, pneumonia, COVID-19, 2019-nCoV, SARS-CoV-2 The virus SARS-CoV-2 (formerly designated 2019- day. Ribavirin should be administered via intravenous nCoV) emerged in December 2019 and then spread infusion at a dose of 500 mg for adults, 2 to 3 times/ rapidly worldwide, particularly to China, Japan, and day in combination with IFN-α or lopinavir/ritonavir. South Korea. As of February 21, 2020, a total of Chloroquine phosphate is orally administered at a dose 76,288 confirmed cases of coronavirus disease 2019 of 500 mg (300 mg for chloroquine) for adults, 2 times/ (COVID-19) and 2,345 deaths have been reported in day. Arbidol is orally administered at a dose of 200 mg mainland China (1). -

Reviews in Basic and Clinical Gastroenterology
GASTROENTEROLOGY 2010;138:447–462 REVIEWS IN BASIC AND CLINICAL GASTROENTEROLOGY REVIEWS IN BASIC AND CLINICAL John P. Lynch and David C. Metz, Section Editors GASTROENTEROLOGY Resistance to Direct Antiviral Agents in Patients With Hepatitis C Virus Infection CHRISTOPH SARRAZIN and STEFAN ZEUZEM J. W. Goethe-University Hospital, Medizinische Klinik 1, Frankfurt am Main, Germany Chronic hepatitis C virus (HCV) infection is one of agents that are also named specific, targeted antiviral the major causes of cirrhosis, hepatocellular carci- therapies (STAT-C) for HCV infection are in phase 1–3 noma, and liver failure that leads to transplantation. trials. We review resistance to DAA agents, focusing on The current standard treatment, a combination of compounds in development such as reagents that target pegylated interferon alfa and ribavirin, eradicates the the HCV nonstructural (NS)3 protease, the NS5A pro- virus in only about 50% of patients. Directly acting tein, and the RNA-dependent RNA polymerase NS5B. We antiviral (DAA) agents, which inhibit HCV replica- also discuss indirect inhibitors or compounds that in- tion, are in phase 1, 2, and 3 trials; these include hibit HCV replication by not yet completely resolved reagents that target the nonstructural (NS)3 protease, mechanisms, such as cyclophilin inhibitors, nitazox- the NS5A protein, the RNA-dependent RNA-polymer- anide, and silibinin (Table 1, Figure 1). ase NS5B, as well as compounds that directly inhibit HCV replication through interaction with host cell Parameters That Affect Resistance proteins. Because of the high genetic heterogeneity of Heterogeneity of HCV HCV and its rapid replication, monotherapy with HCV has a high rate of turnover; its half-life was DAA agents poses a high risk for selection of resistant estimated to be only 2–5 hours, with the production and variants.