Association Between Intensification of Metformin Treatment with Insulin Vs
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

A2) United States Patent (10) Patent No.: US 8,940,711 B2 Olsonet Al
US008940711B2 a2) United States Patent (10) Patent No.: US 8,940,711 B2 Olsonet al. (45) Date of Patent: Jan. 27, 2015 (54) MICRO-RNA FAMILY THAT MODULATES C1I2N 2310/321 (2013.01); C12N 2310/346 FIBROSIS AND USES THEREOF (2013.01); CI2N 2310/3515 (2013.01); C12N (71) Applicant: The Board of Regents, The University 2320/31 (2013.01); CI2N 2330/10 (2013.01) of Texas System, Austin, TX (US) USPC iececcsesseeseeesenensceesensessecsenesenscnseeee 514/444 (58) Field of Classification Search (72) Inventors: Erie N. Olson, Dallas, TX (US); Eva USPC iececcsesseeseeesenensceesensessecsenesenscnseeee 514/44A van Rooij, Utrecht (NL) See application file for complete search history. (56) References Cited (73) Assignee: The Board of Regents, The University of Texas System, Austin, TX (US) U.S. PATENT DOCUMENTS Notice: Subject to any disclaimer, the term of this 7,232,806 B2 6/2007 Tuschletal. (*) 7,674,617 B2 3/2010 Kim etal. patent is extended or adjusted under 35 2005/0059005 Al 3/2005 Tuschletal. U.S.C. 154(b) by 1 day. 2005/0222399 Al 10/2005 Bentwich 2005/0261218 Al 11/2005 Esau etal. (21) Appl. No.: 13/840,242 2006/0019286 Al 1/2006 Horvitz et al. 2006/0105360 Al 5/2006 Croceet al. Filed: Mar.15, 2013 2006/0185027 Al 8/2006 Bartel etal. (22) 2006/0247193 Al 11/2006 Taira etal. 2007/0087335 Al 4/2007 Brahmacharietal. (65) Prior Publication Data 2007/0092882 Al 4/2007 Wangetal. US 2013/0261169 Al Oct. 3, 2013 2008/0050722 Al 2/2008 Kim etal. 2008/0176766 Al 7/2008 Brown etal. -

(12) United States Patent (10) Patent N0.: US 8,343,962 B2 Kisak Et Al
US008343962B2 (12) United States Patent (10) Patent N0.: US 8,343,962 B2 Kisak et al. (45) Date of Patent: *Jan. 1, 2013 (54) TOPICAL FORMULATION (58) Field of Classi?cation Search ............. .. 514/226.5, 514/334, 420, 557, 567 (75) Inventors: Edward T. Kisak, San Diego, CA (US); See application ?le fOr Complete Search history. John M. NeWsam, La Jolla, CA (US); _ Dominic King-Smith, San Diego, CA (56) References C‘ted (US); Pankaj Karande, Troy, NY (US); Samir Mitragotri, Goleta, CA (US) US' PATENT DOCUMENTS 5,602,183 A 2/1997 Martin et al. (73) Assignee: NuvoResearchOntano (CA) Inc., Mississagua, 6,328,979 2B1 12/2001 Yamashita et a1. 7,001,592 B1 2/2006 Traynor et a1. ( * ) Notice: Subject to any disclaimer, the term of this 7,795,309 B2 9/2010 Kisak eta1~ patent is extended or adjusted under 35 2002/0064524 A1 5/2002 Cevc U.S.C. 154(b) by 212 days. FOREIGN PATENT DOCUMENTS This patent is subject to a terminal dis- W0 WO 2005/009510 2/2005 claimer- OTHER PUBLICATIONS (21) APPI' NO‘, 12/848,792 International Search Report issued on Aug. 8, 2008 in application No. PCT/lB2007/0l983 (corresponding to US 7,795,309). _ Notice ofAlloWance issued on Apr. 29, 2010 by the Examiner in US. (22) Med Aug- 2’ 2010 Appl. No. 12/281,561 (US 7,795,309). _ _ _ Of?ce Action issued on Dec. 30, 2009 by the Examiner in US. Appl. (65) Prior Publication Data No, 12/281,561 (Us 7,795,309), Us 2011/0028460 A1 Feb‘ 3’ 2011 Primary Examiner * Raymond Henley, 111 Related U 5 Application Data (74) Attorney, Agent, or Firm * Foley & Lardner LLP (63) Continuation-in-part of application No. -

The In¯Uence of Medication on Erectile Function
International Journal of Impotence Research (1997) 9, 17±26 ß 1997 Stockton Press All rights reserved 0955-9930/97 $12.00 The in¯uence of medication on erectile function W Meinhardt1, RF Kropman2, P Vermeij3, AAB Lycklama aÁ Nijeholt4 and J Zwartendijk4 1Department of Urology, Netherlands Cancer Institute/Antoni van Leeuwenhoek Hospital, Plesmanlaan 121, 1066 CX Amsterdam, The Netherlands; 2Department of Urology, Leyenburg Hospital, Leyweg 275, 2545 CH The Hague, The Netherlands; 3Pharmacy; and 4Department of Urology, Leiden University Hospital, P.O. Box 9600, 2300 RC Leiden, The Netherlands Keywords: impotence; side-effect; antipsychotic; antihypertensive; physiology; erectile function Introduction stopped their antihypertensive treatment over a ®ve year period, because of side-effects on sexual function.5 In the drug registration procedures sexual Several physiological mechanisms are involved in function is not a major issue. This means that erectile function. A negative in¯uence of prescrip- knowledge of the problem is mainly dependent on tion-drugs on these mechanisms will not always case reports and the lists from side effect registries.6±8 come to the attention of the clinician, whereas a Another way of looking at the problem is drug causing priapism will rarely escape the atten- combining available data on mechanisms of action tion. of drugs with the knowledge of the physiological When erectile function is in¯uenced in a negative mechanisms involved in erectile function. The way compensation may occur. For example, age- advantage of this approach is that remedies may related penile sensory disorders may be compen- evolve from it. sated for by extra stimulation.1 Diminished in¯ux of In this paper we will discuss the subject in the blood will lead to a slower onset of the erection, but following order: may be accepted. -

Questions in the Chemical Enzymology of MAO
Review Questions in the Chemical Enzymology of MAO Rona R. Ramsay 1,* and Alen Albreht 2 1 Biomedical Sciences Research Complex, School of Biology, University of St Andrews, St Andrews KY16 9ST, UK 2 Laboratory for Food Chemistry, Department of Analytical Chemistry, National Institute of Chemistry, Hajdrihova 19, SI-1000 Ljubljana, Slovenia; [email protected] * Correspondence: [email protected]; Tel.: +44-(0)-1334-474740 Abstract: We have structure, a wealth of kinetic data, thousands of chemical ligands and clinical information for the effects of a range of drugs on monoamine oxidase activity in vivo. We have comparative information from various species and mutations on kinetics and effects of inhibition. Nevertheless, there are what seem like simple questions still to be answered. This article presents a brief summary of existing experimental evidence the background and poses questions that remain intriguing for chemists and biochemists researching the chemical enzymology of and drug design for monoamine oxidases (FAD-containing EC 4.1.3.4). Keywords: chemical mechanism; kinetic mechanism; oxidation; protein flexibility; cysteine modifica- tion; reversible/irreversible inhibition; molecular dynamics; simulation 1. Introduction Monoamine oxidase (E.C. 1.4.3.4) enzymes MAO A and MAO B are FAD-containing Citation: Ramsay, R.R.; Albreht, A. proteins located on the outer face of the mitochondrial inner membrane, retained there Questions in the Chemical Enzymology of MAO. Chemistry 2021, by hydrophobic interactions and a transmembrane helix. The redox co-factor (FAD) is 3, 959–978. https://doi.org/10.3390/ covalently attached to a cysteine and buried deep inside the protein [1]. -

Azilect, INN-Rasagiline
SCIENTIFIC DISCUSSION 1. Introduction AZILECT is indicated for the treatment of idiopathic Parkinson’s disease (PD) as monotherapy (without levodopa) or as adjunct therapy (with levodopa) in patients with end of dose fluctuations. Rasagiline is administered orally, at a dose of 1 mg once daily with or without levodopa. Parkinson’s disease is a common neurodegenerative disorder typified by loss of dopaminergic neurones from the basal ganglia, and by a characteristic clinical syndrome with cardinal physical signs of resting tremor, bradikinesia and rigidity. The main treatment aims at alleviating symptoms through a balance of anti-cholinergic and dopaminergic drugs. Parkinson’s disease (PD) treatment is complex due to the progressive nature of the disease, and the array of motor and non-motor features combined with early and late side effects associated with therapeutic interventions. Rasagiline is a chemical inhibitor of the enzyme monoamine oxidase (MAO) type B which has a major role in the inactivation of biogenic and diet-derived amines in the central nervous system. MAO has two isozymes (types A and B) and type B is responsible for metabolising dopamine in the central nervous system; as dopamine deficiency is the main contributing factor to the clinical manifestations of Parkinson’s disease, inhibition of MAO-B should tend to restore dopamine levels towards normal values and this improve the condition. Rasagiline was developed for the symptomatic treatment of Parkinson’s disease both as monotherapy in early disease and as adjunct therapy to levodopa + aminoacids decarboxylase inhibitor (LD + ADI) in patients with motor fluctuations. 2. Quality Introduction Drug Substance • Composition AZILECT contains rasagiline mesylate as the active substance. -

Deliverable 5.A Interim Report on the Study Results APPENDIX 2
Deliverable 5.a Interim report on the study results APPENDIX 2: Algorithms used to identify study variables for service contract EMA/2011/38/CN ‐ PIOGLITAZONE November 28th 2012 D5.a Interim report on the study results (Appendix 2) for Service Contract EMA/2011/38/CN PIOGLITAZONE Author(s): Vera Ehrenstein (AUH‐AS) APPENDIX 2. ALGORITHMS USED TO IDENTIFY STUDY VARIABLES Algorithms for AU Database DISEASE/CONDITION ICD-8 CODE (1977-1993) ICD-10 CODE (1994-) Diabetes type 2 250.00; 250.06; 250.07; 250.09 E11.0; E11.1; E11.9 Cancer of bladder 188 C67 Haematuria N/A R31 Haematuria, unspecified B18, K70.0–K70.3, K70.9, K71, K73, Mild hepatic impairment 571, 573.01, 573.04 K74, K76.0 Moderate to severe hepatic 070.00, 070.02, 070.04, 070.06, B15.0, B16.0, B16.2, B19.0, K70.4, impairment 070.08, 573.00, 456.00–456.09 K72, K76.6, I85 Acute myocardial infarction 410 I21-I23 Acute coronary syndrome 410, 413 I20-I24 Ischemic heart disease 410-414 I20-I25 427.09, 427.10, 427.11, 427.19, Congestive heart failure I50, I11.0, I13.0,I13.2 428.99, 782.49; Acute renal failure N/A N17 Diabetic coma N/A E10.0, E11.0, E12.0,E13.0, E14.0 Diabetic acidosis N/A E10.1, E11.1, E12.1,E13.1, E14.1 F10.1-F10.9, G31.2, G62.1, G72.1, Alcoholism 291, 303, 577.10, 571.09, 571.10 I42.6, K29.2, K86.0, Z72.1 Obesity 277.99 E65-E66 D5.a Interim report on the study results (Appendix 2) for Service Contract EMA/2011/38/CN PIOGLITAZONE Author(s): Vera Ehrenstein (AUH‐AS) Algorithms for defining acute events in Denmark, ICD-10 code Event ICD-10 code I21.x, I23.x http://apps.who.int/classifications/icd10/browse/2010/en#/I21 -

Pharmacy and Poisons (Third and Fourth Schedule Amendment) Order 2017
Q UO N T FA R U T A F E BERMUDA PHARMACY AND POISONS (THIRD AND FOURTH SCHEDULE AMENDMENT) ORDER 2017 BR 111 / 2017 The Minister responsible for health, in exercise of the power conferred by section 48A(1) of the Pharmacy and Poisons Act 1979, makes the following Order: Citation 1 This Order may be cited as the Pharmacy and Poisons (Third and Fourth Schedule Amendment) Order 2017. Repeals and replaces the Third and Fourth Schedule of the Pharmacy and Poisons Act 1979 2 The Third and Fourth Schedules to the Pharmacy and Poisons Act 1979 are repealed and replaced with— “THIRD SCHEDULE (Sections 25(6); 27(1))) DRUGS OBTAINABLE ONLY ON PRESCRIPTION EXCEPT WHERE SPECIFIED IN THE FOURTH SCHEDULE (PART I AND PART II) Note: The following annotations used in this Schedule have the following meanings: md (maximum dose) i.e. the maximum quantity of the substance contained in the amount of a medicinal product which is recommended to be taken or administered at any one time. 1 PHARMACY AND POISONS (THIRD AND FOURTH SCHEDULE AMENDMENT) ORDER 2017 mdd (maximum daily dose) i.e. the maximum quantity of the substance that is contained in the amount of a medicinal product which is recommended to be taken or administered in any period of 24 hours. mg milligram ms (maximum strength) i.e. either or, if so specified, both of the following: (a) the maximum quantity of the substance by weight or volume that is contained in the dosage unit of a medicinal product; or (b) the maximum percentage of the substance contained in a medicinal product calculated in terms of w/w, w/v, v/w, or v/v, as appropriate. -

Rauwolfia in the Treatment of Hypertension Douglas Lobay, Bsc, ND
REVIEW ARTICLE Rauwolfia in the Treatment of Hypertension Douglas Lobay, BSc, ND Abstract Rauwolfia serpentina is a safe and effective treatment for on the plant’s role in treating high blood pressure, the hypertension. The plant was used by many physicians author looks at medical uses of the plant, critically throughout India in the 1940s and then was used examining its adverse side effects, toxicology, and throughout the world in the 1950s, including in the carcinogenicity. The author refutes the association United States and Canada. It fell out of popularity when between the plant and carcinogenicity and discusses the adverse side effects, including depression and cancer, importance of correct dosing and of screening patients became associated with it. This author reviews the to minimize the occurrence of depression. He concludes scientific literature with regard to the use of Rauwolfia with the recommendation of use of low dose Rauwolfia and the treatment of hypertension. The author reviews (LDR) for suitable patients with hypertension. The the plant’s botany, chemistry, and pharmacology and plant provides clinicians with a safe and effective provides a researched and documented method of adjunct to pharmaceuticals in the treatment of high action for the active ingredients. With special emphasis blood pressure. Douglas Lobay, BSc, ND, is a naturopathic physician in History and Folk Use Kelowna, British Columbia, Canada. R serpentina was used in folk medicine in India for centuries to treat a wide variety of maladies, including snake and insect bites, febrile conditions, malaria, Corresponding author: Douglas Lobay, BSc, ND abdominal pain, and dysentery. It was also used as a E-mail address: [email protected] uterine stimulant, febrifuge, and cure for insanity. -

Evidence That Formulations of the Selective MAO-B Inhibitor, Selegiline, Which Bypass First-Pass Metabolism, Also Inhibit MAO-A in the Human Brain
Neuropsychopharmacology (2015) 40, 650–657 OPEN & 2015 American College of Neuropsychopharmacology. All rights reserved 0893-133X/15 www.neuropsychopharmacology.org Evidence that Formulations of the Selective MAO-B Inhibitor, Selegiline, which Bypass First-Pass Metabolism, also Inhibit MAO-A in the Human Brain Joanna S Fowler*,1, Jean Logan2, Nora D Volkow3,4, Elena Shumay4, Fred McCall-Perez5, Millard Jayne4, Gene-Jack Wang4, David L Alexoff1, Karen Apelskog-Torres4, Barbara Hubbard1, Pauline Carter1, 1 6 7 4 1 1 4 Payton King , Stanley Fahn , Michelle Gilmor , Frank Telang , Colleen Shea , Youwen Xu and Lisa Muench 1Biological, Environmental and Climate Sciences Department, Brookhaven National Laboratory, Upton, NY, USA; 2New York University Langone Medical Center, Department of Radiology, New York, NY, USA; 3National Institute on Drug Abuse, National Institutes of Health, Bethesda, MD, 4 5 USA; National Institute on Alcohol Abuse and Alcoholism, National Institutes of Health, Bethesda, MD, USA; Targeted Medical Pharma Inc, 6 7 Los Angeles, CA, USA; Department of Neurology, Columbia University Medical Center, New York, NY, USA; Novartis Pharmaceuticals, East Hanover, NJ, USA Selegiline (L-deprenyl) is a selective, irreversible inhibitor of monoamine oxidase B (MAO-B) at the conventional dose (10 mg/day oral) that is used in the treatment of Parkinson’s disease. However, controlled studies have demonstrated antidepressant activity for high doses of oral selegiline and for transdermal selegiline suggesting that when plasma levels of selegiline are elevated, brain MAO-A might also be inhibited. Zydis selegiline (Zelapar) is an orally disintegrating formulation of selegiline, which is absorbed through the buccal mucosa producing higher plasma levels of selegiline and reduced amphetamine metabolites compared with equal doses of conventional selegiline. -

(12) United States Patent (10) Patent No.: US 9,642,912 B2 Kisak Et Al
USOO9642912B2 (12) United States Patent (10) Patent No.: US 9,642,912 B2 Kisak et al. (45) Date of Patent: *May 9, 2017 (54) TOPICAL FORMULATIONS FOR TREATING (58) Field of Classification Search SKIN CONDITIONS CPC ...................................................... A61K 31f S7 (71) Applicant: Crescita Therapeutics Inc., USPC .......................................................... 514/171 Mississauga (CA) See application file for complete search history. (72) Inventors: Edward T. Kisak, San Diego, CA (56) References Cited (US); John M. Newsam, La Jolla, CA (US); Dominic King-Smith, San Diego, U.S. PATENT DOCUMENTS CA (US); Pankaj Karande, Troy, NY (US); Samir Mitragotri, Santa Barbara, 5,602,183 A 2f1997 Martin et al. CA (US); Wade A. Hull, Kaysville, UT 5,648,380 A 7, 1997 Martin 5,874.479 A 2, 1999 Martin (US); Ngoc Truc-ChiVo, Longueuil 6,328,979 B1 12/2001 Yamashita et al. (CA) 7,001,592 B1 2/2006 Traynor et al. 7,795,309 B2 9/2010 Kisak et al. (73) Assignee: Crescita Therapeutics Inc., 8,343,962 B2 1/2013 Kisak et al. Mississauga (CA) 8,513,304 B2 8, 2013 Kisak et al. 8,535,692 B2 9/2013 Pongpeerapat et al. (*) Notice: Subject to any disclaimer, the term of this 9,308,181 B2* 4/2016 Kisak ..................... A61K 47/12 patent is extended or adjusted under 35 2002fOOO6435 A1 1/2002 Samuels et al. 2002fOO64524 A1 5, 2002 Cevc U.S.C. 154(b) by 204 days. 2005, OO 14823 A1 1/2005 Soderlund et al. This patent is Subject to a terminal dis 2005.00754O7 A1 4/2005 Tamarkin et al. -

Title 16. Crimes and Offenses Chapter 13. Controlled Substances Article 1
TITLE 16. CRIMES AND OFFENSES CHAPTER 13. CONTROLLED SUBSTANCES ARTICLE 1. GENERAL PROVISIONS § 16-13-1. Drug related objects (a) As used in this Code section, the term: (1) "Controlled substance" shall have the same meaning as defined in Article 2 of this chapter, relating to controlled substances. For the purposes of this Code section, the term "controlled substance" shall include marijuana as defined by paragraph (16) of Code Section 16-13-21. (2) "Dangerous drug" shall have the same meaning as defined in Article 3 of this chapter, relating to dangerous drugs. (3) "Drug related object" means any machine, instrument, tool, equipment, contrivance, or device which an average person would reasonably conclude is intended to be used for one or more of the following purposes: (A) To introduce into the human body any dangerous drug or controlled substance under circumstances in violation of the laws of this state; (B) To enhance the effect on the human body of any dangerous drug or controlled substance under circumstances in violation of the laws of this state; (C) To conceal any quantity of any dangerous drug or controlled substance under circumstances in violation of the laws of this state; or (D) To test the strength, effectiveness, or purity of any dangerous drug or controlled substance under circumstances in violation of the laws of this state. (4) "Knowingly" means having general knowledge that a machine, instrument, tool, item of equipment, contrivance, or device is a drug related object or having reasonable grounds to believe that any such object is or may, to an average person, appear to be a drug related object. -
Guidance for the Format and Content of the Protocol of Non-Interventional
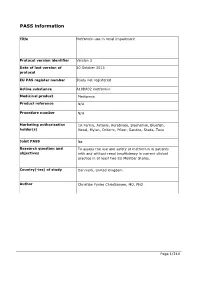
PASS information Title Metformin use in renal impairment Protocol version identifier Version 2 Date of last version of 30 October 2013 protocol EU PAS register number Study not registered Active substance A10BA02 metformin Medicinal product Metformin Product reference N/A Procedure number N/A Marketing authorisation 1A Farma, Actavis, Aurobindo, Biochemie, Bluefish, holder(s) Hexal, Mylan, Orifarm, Pfizer, Sandoz, Stada, Teva Joint PASS No Research question and To assess the use and safety of metformin in patients objectives with and without renal insufficiency in current clinical practice in at least two EU Member States. Country(-ies) of study Denmark, United Kingdom Author Christian Fynbo Christiansen, MD, PhD Page 1/214 Marketing authorisation holder(s) Marketing authorisation N/A holder(s) MAH contact person N/A Page 2/214 1. Table of Contents PASS information .......................................................................................................... 1 Marketing authorisation holder(s) .................................................................................... 2 1. Table of Contents ...................................................................................................... 3 2. List of abbreviations ................................................................................................... 4 3. Responsible parties .................................................................................................... 5 4. Abstract ..................................................................................................................