Luiza Waechter Severo
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

RNA Detection Technology for Applications in Marine Science: Microbes to Fish Robert Michael Ulrich University of South Florida, [email protected]
University of South Florida Scholar Commons Graduate Theses and Dissertations Graduate School 6-25-2014 RNA Detection Technology for Applications in Marine Science: Microbes to Fish Robert Michael Ulrich University of South Florida, [email protected] Follow this and additional works at: https://scholarcommons.usf.edu/etd Part of the Biology Commons, and the Molecular Biology Commons Scholar Commons Citation Ulrich, Robert Michael, "RNA Detection Technology for Applications in Marine Science: Microbes to Fish" (2014). Graduate Theses and Dissertations. https://scholarcommons.usf.edu/etd/5321 This Dissertation is brought to you for free and open access by the Graduate School at Scholar Commons. It has been accepted for inclusion in Graduate Theses and Dissertations by an authorized administrator of Scholar Commons. For more information, please contact [email protected]. RNA Detection Technology for Applications in Marine Science: Microbes to Fish by Robert M. Ulrich A dissertation submitted in partial fulfillment of the requirements for the degree of Doctor of Philosophy College of Marine Science University of South Florida Major Professor: John H. Paul, Ph.D. Valerie J. Harwood, Ph.D. Mya Breitbart, Ph.D. Christopher D. Stallings, Ph.D. David E. John, Ph.D. Date of Approval June 25, 2014 Keywords: NASBA, grouper, Karenia mikimotoi, Enterococcus Copyright © 2014, Robert M. Ulrich DEDICATION This dissertation is dedicated to my fiancée, Dr. Shannon McQuaig for inspiring my return to graduate school and her continued support over the last four years. On no other porch in our little town have there been more impactful scientific discussions, nor more words of encouragement. ACKNOWLEDGMENTS I gratefully acknowledge the many people who have encouraged and advised me throughout my graduate studies. -

Mediterranean Marine Science
Mediterranean Marine Science Vol. 20, 2019 “New Mediterranean Biodiversity Records” 2019 STERN NIR BADREDDINE ALI BITAR GHAZI CROCETTA FABIO DEIDUN ALAN DRAGIČEVIĆ BRANCO DULČIĆ JAKOV DURGHAM HANI GALIL BELLA GALIYA MOHAMMAD IKHTIYAR SAMAR IZQUIREDO-MUÑOZ ANDREAS KASSAR ABDERRAHMANE LOMBARDO ANDREA LUBINEVSKY HADAS MASALLES DAVID OTHMAN RANIM OUSSELLAM MARIAM PEŠIĆ VLADIMIR PIPITONE CARLO RAMOS-ESPLÁ ALFONSO RILOV GIL ROTHMAN SHEVY SELFATI MOHAMED TIRALONGO FRANCESCO TÜRKER ALI UGARKOVIĆ PERO YAPICI SERCAN ZAVA BRUNO http://dx.doi.org/10.12681/mms.20602 Copyright © 2019 Mediterranean Marine Science To cite this article: STERN, N., BADREDDINE, A., BITAR, G., CROCETTA, F., DEIDUN, A., DRAGIČEVIĆ, B., DULČIĆ, J., DURGHAM, http://epublishing.ekt.gr | e-Publisher: EKT | Downloaded at 03/10/2019 11:35:16 | H., GALIL, B., GALIYA, M., IKHTIYAR, S., IZQUIREDO-MUÑOZ, A., KASSAR, A., LOMBARDO, A., LUBINEVSKY, H., MASALLES, D., OTHMAN, R., OUSSELLAM, M., PEŠIĆ, V., PIPITONE, C., RAMOS-ESPLÁ, A., RILOV, G., ROTHMAN, S., SELFATI, M., TIRALONGO, F., TÜRKER, A., UGARKOVIĆ, P., YAPICI, S., & ZAVA, B. (2019). “New Mediterranean Biodiversity Records” 2019. Mediterranean Marine Science, 20(2), 409-426. doi:http://dx.doi.org/10.12681/mms.20602 http://epublishing.ekt.gr | e-Publisher: EKT | Downloaded at 03/10/2019 11:35:16 | Collective Article Mediterranean Marine Science Indexed in WoS (Web of Science, ISI Thomson) and SCOPUS The journal is available on line at http://www.medit-mar-sc.net DOI: http://dx.doi.org/10.12681/mms.20602 New Mediterranean Biodiversity Records (July 2019) Nir STERN1, Ali BADREDDINE2, Ghazi BITAR3, Fabio CROCETTA4, Alan DEIDUN5, Branko DRAGIČEVIĆ6, Jakov DULČIĆ6, Hani DURGHAM7,8, Bella S. -

Islnad Grouper Status Review 2015
Island Grouper (Mycteroperca fusca) Status Review Report Photo Credit: Philippe Burnel © July 2015 Ronald J. Salz Fishery Biologist National Marine Fisheries Service National Oceanic and Atmospheric Administration Silver Spring, MD Acknowledgements I thank João Pedro Barreiros, Claudia Ribeiro, and Fernando Tuya for providing their time, expertise, and insights as external peer reviewers of this report. I also thank the following NMFS co-workers who provided valuable information and feedback as internal reviewers: Dwayne Meadows, Maggie Miller, Marta Nammack, Angela Somma, and Chelsea Young. Other helpful sources of information were George Sedberry, Carlos Ferreira Santos, Albertino Martins, Rui Freitas, and the Portugal National Institute of Statistics. I also thank NOAA librarian Caroline Woods for quickly responding to article requests, and Philippe Burnel for allowing me to use his island grouper photograph on the cover page. This document should be cited as: Salz, R. J. 2015. Island Grouper (Mycteroperca fusca) Draft Status Review Report. Report to National Marine Fisheries Service, Office of Protected Resources. July 2015, 69 pp. 2 Executive Summary On July 15, 2013, NMFS received a petition to list 81 species of marine organisms as endangered or threatened species under the Endangered Species Act (ESA). If an ESA petition is found to present substantial scientific or commercial information that the petitioned action may be warranted, a status review shall be promptly commenced (16 U.S.C. 1533(b)(3)(A)). NMFS determined that for 27 of the 81 species, including the island grouper (Mycteroperca fusca), the petition had sufficient merit for consideration and that a status review was warranted (79 FR 10104, February 24, 2014). -

Ensuring Seafood Identity: Grouper Identification by Real-Time Nucleic
Food Control 31 (2013) 337e344 Contents lists available at SciVerse ScienceDirect Food Control journal homepage: www.elsevier.com/locate/foodcont Ensuring seafood identity: Grouper identification by real-time nucleic acid sequence-based amplification (RT-NASBA) Robert M. Ulrich a, David E. John b, Geran W. Barton c, Gary S. Hendrick c, David P. Fries c, John H. Paul a,* a College of Marine Science, MSL 119, University of South Florida, 140 Seventh Ave. South, St. Petersburg, FL 33701, USA b Department of Biological Sciences, University of South Florida St. Petersburg, 140 Seventh Ave. S., St. Petersburg, FL 33701, USA c EcoSystems Technology Group, College of Marine Science, University of South Florida, 140 Seventh Ave. S., St. Petersburg, FL 33701, USA article info abstract Article history: Grouper are one of the most economically important seafood products in the state of Florida and their Received 19 September 2012 popularity as a high-end restaurant dish is increasing across the U.S. There is an increased incidence rate Accepted 3 November 2012 of the purposeful, fraudulent mislabeling of less costly and more readily available fish species as grouper in the U.S., particularly in Florida. This is compounded by commercial quotas on grouper becoming Keywords: increasingly more restrictive, which continues to drive both wholesale and restaurant prices higher each RT-NASBA year. Currently, the U.S. Food and Drug Administration recognize 56 species of fish that can use “grouper” FDA seafood list as an acceptable market name for interstate commerce. This group of fish includes species from ten Grouper fi fi Mislabeling different genera, making accurate taxonomic identi cation dif cult especially if distinguishing features such as skin, head, and tail have been removed. -

Philippines RVS Fish (866) 874-7639 (855) 225-8086
American Ingenuity Tranship www.livestockusa.org Philippines RVS Fish (866) 874-7639 (855) 225-8086 Tranship - F.O.B. Manila Sunday to LAX - Monday to You Animal cost plus landing costs Order Cut-off is on Thursdays! See landing costs below No guaranty on specialty fish over $75 January 19, 2020 Excellent quality fish! The regals eat, etc.! Code Common Name Binomial - scientific name Price Stock MADAGASCAR FISH RS0802 GOLDEN PUFFER (SHOW SIZE) AROTHRON CITRINELLUS $600.00 1 RS0409 GEM TANG ZEBRASOMA GEMMATUM $775.00 46 RS0408 BLOND NASO TANG NASO HEXACANTHUS $60.00 2 RS0411 POWDER BLUE TANG (MADAGASCAR) ACANTHURUS LEUCOSTERNON $51.75 1 RS0609 MADAGASCAR FLASHER WRASSE (MALE) PARACHEILINUS HEMITAENIATUS $300.00 2 RS0609F MADAGASCAR FLASHER WRASSE (FEMALE) PARACHEILINUS HEMITAENIATUS $200.00 2 RS0107 FLAMEBACK ANGEL (MADAGASCAR) CENTROPYGE ACANTHOPS $55.00 8 RS1301 CORAZON'S DAMSELFISH POMACENTRUS VATOSOA $51.75 3 WEST AFRICAN FISH WA0601 WEST AFRICAN BLACK BAR HOGFISH BODIANUS SPECIOSUS $75.00 4 WA0902 WHITE SPOTTED DRAGON EEL (S) MURAENA MELANOTIS $215.63 1 WA0902XX WHITE SPOTTED DRAGON EEL (SHOW) MURAENA MELANOTIS $431.25 2 WA1501 WEST AFRICAN RED BISCUIT STARFISH TOSIA QUEENSLANDENSIS $60.38 10 WA0101 BLUE SPOT CORAL GROUPER (WEST AFRICAN) CEPHALOPHOLIS TAENIOPS $51.75 1 PHILIPPINES FISH 01010 BLUE KORAN ANGEL JUV (L) POMACANTHUS SEMICIRCULATUS $20.55 1 01011 BLUE KORAN ANGEL JUV (M) POMACANTHUS SEMICIRCULATUS $16.43 3 01016 SIX BAR ANGEL ADULT EUXIPHIPOPS SEXTRIATUS $13.17 2 01017 SIX BAR ANGEL JUVENILE (S) EUXIPHIPOPS SEXTRIATUS $7.43 -

Occurrence of the Malabar Grouper Epinephelus Malabaricus (Bloch & Schneider, 1801) (Actinopterygii, Perciformes, Serranidae), in the Maltese Islands
Aquatic Invasions (2011) Volume 6, Supplement 1: S129–S132 doi: 10.3391/ai.2011.6.S1.029 Open Access © 2011 The Author(s). Journal compilation © 2011 REABIC Aquatic Invasions Records Occurrence of the Malabar grouper Epinephelus malabaricus (Bloch & Schneider, 1801) (Actinopterygii, Perciformes, Serranidae), in the Maltese Islands Patrick J. Schembri1* and Reno Tonna2 1Department of Biology, University of Malta, Msida MSD2080, Malta 2Namaste Flat 5, Triq il-Merzuq, Birzebbuga, Malta E-mail: [email protected] (PJS), [email protected] (RT) *Corresponding author Received: 3 August 2011 / Accepted: 12 October 2011 / Published online: 20 October 2011 Abstract The capture of a single specimen of the Malabar grouper Epinephelus malabaricus (Bloch & Schneider, 1801) from inside the breakwater at the entrance to the Grand Harbour at Valletta, Malta in July 2011 is reported here. Previously this Lessepsian immigrant species was only known from Israeli waters where it is established but rare. On the presently available evidence, the Maltese specimen has to be considered as a casual, most likely transported by shipping. Key words: Epinephelinae, Malta, central Mediterranean, non-indigenous species, Lessepsian immigrant Introduction and Malta (photographs and record published in FishBase and reported in a Maltese newspaper; Groupers of the subfamily Epinephelinae are Froese and Pauly 2011; Vassallo 2009), although represented by nine species in the Mediterranean, of one of us (PJS) knows of an earlier (2008) which six – Epinephelus aeneus (Geoffroy Saint- unpublished record from Malta. The species seems Hilaire, 1817), Epinephelus caninus (Valenciennes, to have become established in the Sicily Channel 1843), Epinephelus costae (Steindachner, 1878), (Guidetti et al. -

Workshop for Red List Assessments of Groupers
Workshop for Global Red List Assessments of Groupers Family Serranidae; subfamily Epinephelinae FINAL REPORT April 30th, 2007 Prepared by Yvonne Sadovy Chair, Groupers & Wrasses Specialist Group The University of Hong Kong (24 pages) Introduction The groupers (Family Serranidae; Subfamily Epinephelinae) comprise about 160 species globally in the tropics and sub-tropics. Many groupers are commercially important and assessments to date on a subset of species suggest that the group might be particularly vulnerable to fishing. An assessment of all grouper species is needed to examine the sub- family as a whole and set conservation and management priorities as indicated. The Serranidae is also a priority family for the Global Marine Species Assessment. This report summarizes the outcomes of the first complete red listing assessment for groupers conducted by the Groupers and Wrasses IUCN Specialist Group (GWSG) at a workshop in Hong Kong. The Workshop for Global Red List Assessments of Groupers took place 7-11 February, 2007, at the Robert Black College of the University of Hong Kong (HKU). The 5-day workshop was designed to complete red list assessments for all grouper species. Of a total of 161 grouper species globally, only 22 are included on the IUCN Red List with a currently valid assessment; several need to be reassessed and the remaining 100+ have never been assessed. The aim of the workshop, therefore, was to assess 139 groupers to complete all 161 species. The workshop had 23 participants, including many highly respected grouper experts, coming from eleven countries (see cover photo of participants). All members of the GWSG were invited in circulation. -

View/Download
PERCIFORMES (part 4) · 1 The© Christopher ETYFishScharpf and Kenneth J. Lazara Project COMMENTS: v. 1.0 - 11 March 2021 Order PERCIFORMES (part 4) Suborder SERRANOIDEI (part 2 of 3) Family SERRANIDAE Sea Basses and Groupers (part 2 of 2) Subfamily Epinephelinae Groupers 17 genera · 189 species Aethaloperca Fowler 1904 aethalos, sooty or black, presumably referring to pale-brown to black color of A. rogaa; perca, perch, i.e., a perch-like fish [treated as a synonym of Cephalopholis by some workers] Aethaloperca rogaa (Fabricius 1775) Rogáa, Arabic name for the grouper along the Red Sea of Saudi Arabia Alphestes Bloch & Schneider 1801 ancient Greek name for a greedy, incontinent fish with a bad reputation, sometimes said to swim in pairs, one behind the other, possibly Symphodus tinca (per Jordan & Evermann 1896), a wrasse; its application to a grouper is not explained Alphestes afer (Bloch 1793) African, described from Guinea, West Africa (but also occurs in western Atlantic from Bermuda and North Carolina south to Uruguay, including southern Gulf of Mexico and Caribbean Sea) Alphestes immaculatus Breder 1936 im-, not; maculatus, spotted, referring to plain coloration (actually mottled, with spotted fins), compared to the profusely spotted P. multiguttatus Alphestes multiguttatus (Günther 1867) multi-, many; guttatus, spotted, referring to head and body profusely covered with dark-brown spots (which often coalesce to form horizontal streaks) Anyperodon Günther 1859 etymology not explained, presumably an-, not; [h]yper, upper; odon, tooth, referring to absence of teeth on palatine Anyperodon leucogrammicus (Valenciennes 1828) leucos, white; grammicus, lined, referring to three whitish longitudinal bands on sides Cephalopholis Bloch & Schneider 1801 cephalus, head; pholis, scale, referring to completely scaled head of C. -
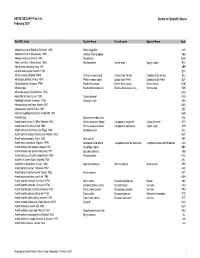
ASFIS ISSCAAP Fish List February 2007 Sorted on Scientific Name
ASFIS ISSCAAP Fish List Sorted on Scientific Name February 2007 Scientific name English Name French name Spanish Name Code Abalistes stellaris (Bloch & Schneider 1801) Starry triggerfish AJS Abbottina rivularis (Basilewsky 1855) Chinese false gudgeon ABB Ablabys binotatus (Peters 1855) Redskinfish ABW Ablennes hians (Valenciennes 1846) Flat needlefish Orphie plate Agujón sable BAF Aborichthys elongatus Hora 1921 ABE Abralia andamanika Goodrich 1898 BLK Abralia veranyi (Rüppell 1844) Verany's enope squid Encornet de Verany Enoploluria de Verany BLJ Abraliopsis pfefferi (Verany 1837) Pfeffer's enope squid Encornet de Pfeffer Enoploluria de Pfeffer BJF Abramis brama (Linnaeus 1758) Freshwater bream Brème d'eau douce Brema común FBM Abramis spp Freshwater breams nei Brèmes d'eau douce nca Bremas nep FBR Abramites eques (Steindachner 1878) ABQ Abudefduf luridus (Cuvier 1830) Canary damsel AUU Abudefduf saxatilis (Linnaeus 1758) Sergeant-major ABU Abyssobrotula galatheae Nielsen 1977 OAG Abyssocottus elochini Taliev 1955 AEZ Abythites lepidogenys (Smith & Radcliffe 1913) AHD Acanella spp Branched bamboo coral KQL Acanthacaris caeca (A. Milne Edwards 1881) Atlantic deep-sea lobster Langoustine arganelle Cigala de fondo NTK Acanthacaris tenuimana Bate 1888 Prickly deep-sea lobster Langoustine spinuleuse Cigala raspa NHI Acanthalburnus microlepis (De Filippi 1861) Blackbrow bleak AHL Acanthaphritis barbata (Okamura & Kishida 1963) NHT Acantharchus pomotis (Baird 1855) Mud sunfish AKP Acanthaxius caespitosa (Squires 1979) Deepwater mud lobster Langouste -

Monogenea, Diplectanidae
Pseudorhabdosynochus regius n. sp. (Monogenea, Diplectanidae) from the mottled grouper Mycteroperca rubra (Teleostei) in the Mediterranean Sea and Eastern Atlantic Amira Chaabane, Lassad Neifar, Jean-Lou Justine To cite this version: Amira Chaabane, Lassad Neifar, Jean-Lou Justine. Pseudorhabdosynochus regius n. sp. (Monogenea, Diplectanidae) from the mottled grouper Mycteroperca rubra (Teleostei) in the Mediterranean Sea and Eastern Atlantic. Parasite, EDP Sciences, 2015, 22, pp.9. 10.1051/parasite/2015005. hal-01271438 HAL Id: hal-01271438 https://hal.sorbonne-universite.fr/hal-01271438 Submitted on 9 Feb 2016 HAL is a multi-disciplinary open access L’archive ouverte pluridisciplinaire HAL, est archive for the deposit and dissemination of sci- destinée au dépôt et à la diffusion de documents entific research documents, whether they are pub- scientifiques de niveau recherche, publiés ou non, lished or not. The documents may come from émanant des établissements d’enseignement et de teaching and research institutions in France or recherche français ou étrangers, des laboratoires abroad, or from public or private research centers. publics ou privés. Distributed under a Creative Commons Attribution| 4.0 International License Parasite 2015, 22,9 Ó A. Chaabane et al., published by EDP Sciences, 2015 DOI: 10.1051/parasite/2015005 urn:lsid:zoobank.org:pub:EEB58B6E-4C70-4F30-AE5D-7169BA21D1C6 Available online at: www.parasite-journal.org RESEARCH ARTICLE OPEN ACCESS Pseudorhabdosynochus regius n. sp. (Monogenea, Diplectanidae) from the mottled -

3. List of Nominal Species of Groupers
click for previous page 306 FAO Species Cataogue Vol. 16 3. LIST OF NOMINAL SPECIES OF GROUPERS The following list gives in order (i) the scientific name as it originally appeared, in alphabetical order according to the specific name; (ii) the author(s) - Cuvier (1829) = in Cuvier and Valenciennes (1829); (iii) date of publication; and (iv) Present identification. NOMINAL SPECIES PRESENT ALLOCATION Serranus abdominalis Peters, 1855 Epinephelus lanceolatus Bodianus acanthistius Gilbert, 1892 Epinephelus acanthistius Serranus acutirostris Valenciennes, 1828 Mycteroperca acutirostris Trachinus Adscensionis Osbeck, 1765 Epinephelus adscensionis Serranus aeneus Geoffroy Saint-Hilaire, 1817 Epinephelus aeneus Epinephelus afer Bloch, 1793 Alphestes afer Trisotropis aguaji Poey, 1867 Mycteroperca bonaci Cephalopholis aitha Randall and Heemstra, 1991 Cephalopholis aitha Serranus aka-ara Temminck and Schlegel, 1842 Epinephelus akaara Serranus alatus Alleyne and Macleay, 1877 Epinephelus quoyanus Epinephelus albimaculatus Seale, 1909 Epinephelus bleekeri Variola albimarginata Baissac, 1953 Variola albimarginata Holocentrus albo -fuscus Lacepède, 1802 Epinephelus maculatus Serranus alboguttatus Valenciennes, 1828 Epinephelus caeruleopunctatus Cephalopholis albomarginatus Fowler and Bean, 1930 Gracila albomarginata Epinephelus albomarginatus Boulenger, 1903 Epinephelus albomarginatus Epinephelus albopunctulatus Boulenger, 1895 Epinephelus irroratus Dermatolepis aldabrensis Smith, 1955 Dermatolepis striolata Serranus Alexandrinus Valenciennes, 1828 -

Morphometric Variation and Allozyme Electrophoretic Studies in Hind
International Journal of Science and Research (IJSR) ISSN (Online): 2319-7064 Index Copernicus Value (2013): 6.14 | Impact Factor (2013): 4.438 Morphometric Variation and Allozyme Electrophoretic Studies in Hind Grouper Species of Genus Cephalopholis (Epinephelidae) off Visakhapatnam, Central Eastern Coast of India V. A. Iswarya Deepti1, K. V. L. Shrikanya2, K. Sujatha3 1 & 3 Department of Marine Living Resources, Andhra University, Visakhapatnam – 530 003, Andhra Pradesh, India 2 Present address: Department of Biotechnology, Vikrama Simhapuri University, Nellore - 524320 Andhra Pradesh, India Abstract: Most studies of groupers have focussed on species of commercially important genera. For smaller Epinephelines such as species of genus Cephalopholis studies are very little despite having commercial value as food in some regions and increasing inclusion in the aquarium trade. The present paper deals with morphometric and meristic characters used to identify six species of hind groupers of genus Cephalopholis that are represented in the coastal waters of Visakhapatnam . Multivariate analysis (Principal Component Analysis) has been carried out to study existing variation in biometric characters of two closely related species – C. nigripinnis (Valen- ciennes, 1828) and C . sonnerati (Valenciennes, 1828). Cephalopholis microprion (Bleeker, 1852) is new record from coastal waters of mainland India. The genetic variability in four species – C. formosa, C. miniata, C. nigripinnis and C. sonnerati was evidenced by al- lozyme electrophoretic studies carried out on eleven enzyme systems that revealed seventeen loci out of which ten are polymorphic. Spe- cies are easily distinguished and also diagnostic locus and alleles were identified that can aid in species identification. This data along with morphometric and meristic data has varied application in research on evolution, conservation and management of natural re- sources and genetic improvement programmes.