Bibliography
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-
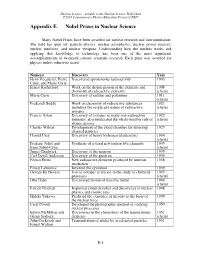
Appendix E Nobel Prizes in Nuclear Science
Nuclear Science—A Guide to the Nuclear Science Wall Chart ©2018 Contemporary Physics Education Project (CPEP) Appendix E Nobel Prizes in Nuclear Science Many Nobel Prizes have been awarded for nuclear research and instrumentation. The field has spun off: particle physics, nuclear astrophysics, nuclear power reactors, nuclear medicine, and nuclear weapons. Understanding how the nucleus works and applying that knowledge to technology has been one of the most significant accomplishments of twentieth century scientific research. Each prize was awarded for physics unless otherwise noted. Name(s) Discovery Year Henri Becquerel, Pierre Discovered spontaneous radioactivity 1903 Curie, and Marie Curie Ernest Rutherford Work on the disintegration of the elements and 1908 chemistry of radioactive elements (chem) Marie Curie Discovery of radium and polonium 1911 (chem) Frederick Soddy Work on chemistry of radioactive substances 1921 including the origin and nature of radioactive (chem) isotopes Francis Aston Discovery of isotopes in many non-radioactive 1922 elements, also enunciated the whole-number rule of (chem) atomic masses Charles Wilson Development of the cloud chamber for detecting 1927 charged particles Harold Urey Discovery of heavy hydrogen (deuterium) 1934 (chem) Frederic Joliot and Synthesis of several new radioactive elements 1935 Irene Joliot-Curie (chem) James Chadwick Discovery of the neutron 1935 Carl David Anderson Discovery of the positron 1936 Enrico Fermi New radioactive elements produced by neutron 1938 irradiation Ernest Lawrence -

Cockcroft: a Novel This One, the Comprehensive Compilation Lutely Fundamental Discovery
~7~~-------------------------------------SPRINGBCXJKS------------------------N_A_ru_~__ v_o_L._Jo_s_~_AP __ Rr_L_r9M_ same, when it's compiled as successfully as what was in the sheer physics of it an abso Cockcroft: a novel this one, the comprehensive compilation lutely fundamental discovery. The authors perspective undeniably earns its place in the library. of this book take in their stride Cockcroft I was an admirer of Cockcroft, a fine, and Walton's being made to wait so long William Cooper stocky, sensible, cautious Yorkshireman, theirs not to reason why, they say. But what who indulged in a minimum of speech and about ours? Did it take the Nobel Cockcroft and the Atom. in practically no change of facial committee nineteen years to recognize the By Guy Hartcup and T.E. Allibone. expression. (That expression was neverthe experiment as breaking open an entirely Adam Hilger, Bristol!Heyden, Phila- less amiable, thoughtful, smiling.) If one new line of powerful experiments to be delphia: 1984. Pp.320. £18.95, $34. addressed a remark to him, it was only made with particle-accelerators? Or did it when, after the ensuing silence, he either take those members who opposed the took the famous black notebook out of his award all that time to disappear from the SrNCE the title of Hartcup and Allibone's pocket or actually said something, that one scene? Or what? Someone must know. book may sound a bit vague, perhaps I knew for certain that he'd heard. It gave me There's a fascinating detail I should really should begin by saying what Cockcroft and lasting pleasure to think that at a dinner like to be told. -

Copyrighted Material
ftoc.qrk 5/24/04 1:46 PM Page iii Contents Timeline v de Sitter,Willem 72 Dukas, Helen 74 Introduction 1 E = mc2 76 Eddington, Sir Arthur 79 Absentmindedness 3 Education 82 Anti-Semitism 4 Ehrenfest, Paul 85 Arms Race 8 Einstein, Elsa Löwenthal 88 Atomic Bomb 9 Einstein, Mileva Maric 93 Awards 16 Einstein Field Equations 100 Beauty and Equations 17 Einstein-Podolsky-Rosen Besso, Michele 18 Argument 101 Black Holes 21 Einstein Ring 106 Bohr, Niels Henrik David 25 Einstein Tower 107 Books about Einstein 30 Einsteinium 108 Born, Max 33 Electrodynamics 108 Bose-Einstein Condensate 34 Ether 110 Brain 36 FBI 113 Brownian Motion 39 Freud, Sigmund 116 Career 41 Friedmann, Alexander 117 Causality 44 Germany 119 Childhood 46 God 124 Children 49 Gravitation 126 Clothes 58 Gravitational Waves 128 CommunismCOPYRIGHTED 59 Grossmann, MATERIAL Marcel 129 Correspondence 62 Hair 131 Cosmological Constant 63 Heisenberg, Werner Karl 132 Cosmology 65 Hidden Variables 137 Curie, Marie 68 Hilbert, David 138 Death 70 Hitler, Adolf 141 iii ftoc.qrk 5/24/04 1:46 PM Page iv iv Contents Inventions 142 Poincaré, Henri 220 Israel 144 Popular Works 222 Japan 146 Positivism 223 Jokes about Einstein 148 Princeton 226 Judaism 149 Quantum Mechanics 230 Kaluza-Klein Theory 151 Reference Frames 237 League of Nations 153 Relativity, General Lemaître, Georges 154 Theory of 239 Lenard, Philipp 156 Relativity, Special Lorentz, Hendrik 158 Theory of 247 Mach, Ernst 161 Religion 255 Mathematics 164 Roosevelt, Franklin D. 258 McCarthyism 166 Russell-Einstein Manifesto 260 Michelson-Morley Experiment 167 Schroedinger, Erwin 261 Millikan, Robert 171 Solvay Conferences 265 Miracle Year 174 Space-Time 267 Monroe, Marilyn 179 Spinoza, Baruch (Benedictus) 268 Mysticism 179 Stark, Johannes 270 Myths and Switzerland 272 Misconceptions 181 Thought Experiments 274 Nazism 184 Time Travel 276 Newton, Isaac 188 Twin Paradox 279 Nobel Prize in Physics 190 Uncertainty Principle 280 Olympia Academy 195 Unified Theory 282 Oppenheimer, J. -

Physicists in Times of War” Has Gained New Topicality As a Result of the US Doctrine of Preemptive Strikes Adopted by the Bush Administration
Physicists in times of war Bert Schroer CBPF, Rua Dr. Xavier Sigaud 150 22290-180 Rio de Janeiro, Brazil and Institut fuer Theoretische Physik der FU Berlin, Germany December 2005 Abstract Though the majority of physicists would probably not support pre- emptive wars, nuclear and other weapons of mass destruction would not exist without their contributions. Einstein’s anti-militaristic position has been well-documented and the present essay recalls the role of some contemporary and past physicists on this issue. The idea that the rationality of scientific thought is a reliable antidote against supporting wars in order to achieve political or ideological aims was neither correct in the past nor is it presently valid. In the physics community there always existed a minority of supporters of wars of conquest of territory, domination of people beyond the borders or regime change. The “preemptive” war for the US hegemony in the middle east has given the problem of “physicists in times of war” new actuality, especially on the third anniversary of its start with a country slipping into civil war and culprit of this bloody mess talking about the fight for victory. One of the most perplexing appologists of the agressive war of Nazi- Germany against “the Bolshevist peril” has been the physicist Pascual Jordan whose interesting scientific and controversial political biography is the main issue of this essay. 1 Introductory remarks As a result of a significant coincidence 2005 was not only the centennial year of Einstein’s greatest discoveries but it also marks the 60 year commemoration of the end of the second world war, which was perhaps one of the greatest arXiv:physics/0603095v2 [physics.soc-ph] 20 Mar 2006 man-made tragedy and certainly 20thcenturies darkest episode. -

The Early History of the Niels Bohr Institute
CHAPTER 4.1 Birthplace of a new physics - the early history of the Niels Bohr Institute Peter Robertson* Abstract SCI.DAN.M. The foundation in 1921 of the Niels Bohr Institute in Copenhagen was to prove an important event in the I birth of modern physics. From its modest beginnings as • ONE a small three-storey building and a handful of physicists, HUNDRED the Institute underwent a rapid expansion over the fol lowing years. Under Bohr’s leadership, the Institute provided the principal centre for the emergence of quan YEARS tum mechanics and a new understanding of Nature at OF the atomic level. Over sixty physicists from 17 countries THE came to collaborate with the Danish physicists at the In BOHR stitute during its first decade. The Bohr Institute was the ATOM: first truly international centre in physics and, indeed, one of the first in any area of science. The Institute pro PROCEEDINGS vided a striking demonstration of the value of interna tional cooperation in science and it inspired the later development of similar centres elsewhere in Europe and FROM the United States. In this article I will discuss the origins and early development of the Institute and examine the A CONFERENCE reasons why it became such an important centre in the development of modern physics. Keywords: Niels Bohr Institute; development of quan tum mechanics; international cooperation in science. * School of Physics, University of Melbourne, Melbourne, Vic. 3010, Australia. Email: [email protected]. 481 PETER ROBERTSON SCI.DAN.M. I 1. Planning and construction of the Institute In 1916 Niels Bohr returned home to Copenhagen over four years since his first visit to Cambridge, England, and two years after a second visit to Manchester, working in the group led by Ernest Ru therford. -

Otto Stern Annalen 4.11.11
(To be published by Annalen der Physik in December 2011) Otto Stern (1888-1969): The founding father of experimental atomic physics J. Peter Toennies,1 Horst Schmidt-Böcking,2 Bretislav Friedrich,3 Julian C.A. Lower2 1Max-Planck-Institut für Dynamik und Selbstorganisation Bunsenstrasse 10, 37073 Göttingen 2Institut für Kernphysik, Goethe Universität Frankfurt Max-von-Laue-Strasse 1, 60438 Frankfurt 3Fritz-Haber-Institut der Max-Planck-Gesellschaft Faradayweg 4-6, 14195 Berlin Keywords History of Science, Atomic Physics, Quantum Physics, Stern- Gerlach experiment, molecular beams, space quantization, magnetic dipole moments of nucleons, diffraction of matter waves, Nobel Prizes, University of Zurich, University of Frankfurt, University of Rostock, University of Hamburg, Carnegie Institute. We review the work and life of Otto Stern who developed the molecular beam technique and with its aid laid the foundations of experimental atomic physics. Among the key results of his research are: the experimental test of the Maxwell-Boltzmann distribution of molecular velocities (1920), experimental demonstration of space quantization of angular momentum (1922), diffraction of matter waves comprised of atoms and molecules by crystals (1931) and the determination of the magnetic dipole moments of the proton and deuteron (1933). 1 Introduction Short lists of the pioneers of quantum mechanics featured in textbooks and historical accounts alike typically include the names of Max Planck, Albert Einstein, Arnold Sommerfeld, Niels Bohr, Max von Laue, Werner Heisenberg, Erwin Schrödinger, Paul Dirac, Max Born, and Wolfgang Pauli on the theory side, and of Wilhelm Conrad Röntgen, Ernest Rutherford, Arthur Compton, and James Franck on the experimental side. However, the records in the Archive of the Nobel Foundation as well as scientific correspondence, oral-history accounts and scientometric evidence suggest that at least one more name should be added to the list: that of the “experimenting theorist” Otto Stern. -

Bibliography
Bibliography F.H. Attix, Introduction to Radiological Physics and Radiation Dosimetry (John Wiley & Sons, New York, New York, USA, 1986) V. Balashov, Interaction of Particles and Radiation with Matter (Springer, Berlin, Heidelberg, New York, 1997) British Journal of Radiology, Suppl. 25: Central Axis Depth Dose Data for Use in Radiotherapy (British Institute of Radiology, London, UK, 1996) J.R. Cameron, J.G. Skofronick, R.M. Grant, The Physics of the Body, 2nd edn. (Medical Physics Publishing, Madison, WI, 1999) S.R. Cherry, J.A. Sorenson, M.E. Phelps, Physics in Nuclear Medicine, 3rd edn. (Saunders, Philadelphia, PA, USA, 2003) W.H. Cropper, Great Physicists: The Life and Times of Leading Physicists from Galileo to Hawking (Oxford University Press, Oxford, UK, 2001) R. Eisberg, R. Resnick, Quantum Physics of Atoms, Molecules, Solids, Nuclei and Particles (John Wiley & Sons, New York, NY, USA, 1985) R.D. Evans, TheAtomicNucleus(Krieger, Malabar, FL USA, 1955) H. Goldstein, C.P. Poole, J.L. Safco, Classical Mechanics, 3rd edn. (Addison Wesley, Boston, MA, USA, 2001) D. Greene, P.C. Williams, Linear Accelerators for Radiation Therapy, 2nd Edition (Institute of Physics Publishing, Bristol, UK, 1997) J. Hale, The Fundamentals of Radiological Science (Thomas Springfield, IL, USA, 1974) W. Heitler, The Quantum Theory of Radiation, 3rd edn. (Dover Publications, New York, 1984) W. Hendee, G.S. Ibbott, Radiation Therapy Physics (Mosby, St. Louis, MO, USA, 1996) W.R. Hendee, E.R. Ritenour, Medical Imaging Physics, 4 edn. (John Wiley & Sons, New York, NY, USA, 2002) International Commission on Radiation Units and Measurements (ICRU), Electron Beams with Energies Between 1 and 50 MeV,ICRUReport35(ICRU,Bethesda, MD, USA, 1984) International Commission on Radiation Units and Measurements (ICRU), Stopping Powers for Electrons and Positrons, ICRU Report 37 (ICRU, Bethesda, MD, USA, 1984) 646 Bibliography J.D. -

Periodic Table 1 Periodic Table
Periodic table 1 Periodic table This article is about the table used in chemistry. For other uses, see Periodic table (disambiguation). The periodic table is a tabular arrangement of the chemical elements, organized on the basis of their atomic numbers (numbers of protons in the nucleus), electron configurations , and recurring chemical properties. Elements are presented in order of increasing atomic number, which is typically listed with the chemical symbol in each box. The standard form of the table consists of a grid of elements laid out in 18 columns and 7 Standard 18-column form of the periodic table. For the color legend, see section Layout, rows, with a double row of elements under the larger table. below that. The table can also be deconstructed into four rectangular blocks: the s-block to the left, the p-block to the right, the d-block in the middle, and the f-block below that. The rows of the table are called periods; the columns are called groups, with some of these having names such as halogens or noble gases. Since, by definition, a periodic table incorporates recurring trends, any such table can be used to derive relationships between the properties of the elements and predict the properties of new, yet to be discovered or synthesized, elements. As a result, a periodic table—whether in the standard form or some other variant—provides a useful framework for analyzing chemical behavior, and such tables are widely used in chemistry and other sciences. Although precursors exist, Dmitri Mendeleev is generally credited with the publication, in 1869, of the first widely recognized periodic table. -

H-Diplo Article Review 20 19
H-Diplo Article Review 20 19 Article Review Editors: Thomas Maddux and Diane Labrosse H-Diplo Web and Production Editor: George Fujii @HDiplo Article Review No. 869 3 July 2019 Walter E. Grunden. “Physicists and ‘Fellow Travelers’: Nuclear Fear, the Red Scare, and Science Policy in Occupied Japan.” Journal of American-East Asian Relations 25:4 (2018): 343-383. DOI: https://doi.org/10.1163/18765610-02504001. https://hdiplo.org/to/AR869 Review by Dong-Won Kim, Harvard University hree decades have passed since the Berlin Wall collapsed in 1989, and most members of the younger generation now consider the Cold War to be as antique as both the First World War and the Second World War. In an era of global terrorism that shakes every corner of the world regardless of political Tand religious differences, the “red scare” of 1947-1960 can even seem to be unrealistic. Historians’ efforts to reinterpret the Cold War from different and distant perspectives are therefore only natural.1 Likewise, historians of science are now reinterpreting science and technology during the Cold War from new angles: for example, David Kaiser’s 2005 analysis of the unfair treatment of several theoretical physicists in the United States during the early Cold War period differs from the analyses offered by the historians of science in the 1980s and 1990s.2 Walter Grunden’s article, “Physicists and ‘Fellow Travelers’: Nuclear Fear, the Red Scare, and Science Policy in Occupied Japan,” offers a fresh understanding of the Cold War outside the United States. The paper is particularly welcome because it systematically analyzes how both the American Occupation (1945-1952) and 1 Two good examples are John Lewis Gaddis, The Cold War: A New History (New York: Penguin Books, 2005), and Odd Arne Westad, The Cold War: A World History (New York: Basic Books, 2017). -

Guide to the James Franck Papers 1882-1966
University of Chicago Library Guide to the James Franck Papers 1882-1966 © 2006 University of Chicago Library Table of Contents Acknowledgments 3 Descriptive Summary 3 Information on Use 3 Access 3 Citation 3 Biographical Note 4 Scope Note 15 Related Resources 21 Subject Headings 21 INVENTORY 22 Series I: Correspondence 22 Series II: Manuscripts 51 Subseries 1: Physics - work in Germany and Denmark, 1905-1934 51 Subseries 2: Physics - work in United States, 1935-1958 53 Subseries 3: Biophysics - work on Photosynthesis at Johns Hopkins, 1935-193855 Subseries 4: Biophysics - work on Photosynthesis at the University of Chicago,55 1938-48 Subseries 5: Biophysics - work on Photosynthesis after 1948 55 Subseries 6: General Articles and Talks on Science 71 Subseries 7: Papers by other scientists 72 Subseries 8: Notes, memoranda and fragments 76 Subseries 9: Atomic Scientists' Movement, 1944-1953 76 Subseries 10: Franck Memorial Symposium, May 12-13, 1966 79 Series III: Tape Recordings and Photographs 80 Subseries 1: Tape recordings 80 Subseries 2: Hertha Sponer's photograph album, Göttingen, 1920-1933 80 Series IV: Personal Documents and Memorabilia 90 Subseries 1: Documents 90 Subseries 2: Clippings 93 Subseries 3: Biographies and Obituaries 94 Subseries 4: Memorabilia; Scrolls, Certificates, Medals, Mementos 96 Series V: Robert Platzman's Editorial Papers for the "Selected Works of James98 Franck" Series VI: Addenda 103 Subseries 1: Correspondence between James Franck and his nephew and Dr. Heinz104 Kallman Subseries 2: Oversize 105 Descriptive Summary Identifier ICU.SPCL.FRANCK Title Franck, James. Papers Date 1882-1966 Size 20.5 linear feet (29 boxes) Repository Special Collections Research Center University of Chicago Library 1100 East 57th Street Chicago, Illinois 60637 U.S.A. -

Newly Opened Correspondence Illuminates Einstein's Personal Life
CENTER FOR HISTORY OF PHYSICS NEWSLETTER Vol. XXXVIII, Number 2 Fall 2006 One Physics Ellipse, College Park, MD 20740-3843, Tel. 301-209-3165 Newly Opened Correspondence Illuminates Einstein’s Personal Life By David C. Cassidy, Hofstra University, with special thanks to Diana Kormos Buchwald, Einstein Papers Project he Albert Einstein Archives at the Hebrew University of T Jerusalem recently opened a large collection of Einstein’s personal correspondence from the period 1912 until his death in 1955. The collection consists of nearly 1,400 items. Among them are about 300 letters and cards written by Einstein, pri- marily to his second wife Elsa Einstein, and some 130 letters Einstein received from his closest family members. The col- lection had been in the possession of Einstein’s step-daughter, Margot Einstein, who deposited it with the Hebrew University of Jerusalem with the stipulation that it remain closed for twen- ty years following her death, which occurred on July 8, 1986. The Archives released the materials to public viewing on July 10, 2006. On the same day Princeton University Press released volume 10 of The Collected Papers of Albert Einstein, con- taining 148 items from the collection through December 1920, along with other newly available correspondence. Later items will appear in future volumes. “These letters”, write the Ein- stein editors, “provide the reader with substantial new source material for the study of Einstein’s personal life and the rela- tionships with his closest family members and friends.” H. Richard Gustafson playing with a guitar to pass the time while monitoring the control room at a Fermilab experiment. -

The History and Impact of the CNO Cycles in Nuclear Astrophysics
Phys. Perspect. 20 (2018) 124–158 Ó 2018 Springer International Publishing AG, part of Springer Nature 1422-6944/18/010124-35 https://doi.org/10.1007/s00016-018-0216-0 Physics in Perspective The History and Impact of the CNO Cycles in Nuclear Astrophysics Michael Wiescher* The carbon cycle, or Bethe-Weizsa¨cker cycle, plays an important role in astrophysics as one of the most important energy sources for quiescent and explosive hydrogen burning in stars. This paper presents the intellectual and historical background of the idea of the correlation between stellar energy production and the synthesis of the chemical elements in stars on the example of this cycle. In particular, it addresses the contributions of Carl Friedrich von Weizsa¨cker and Hans Bethe, who provided the first predictions of the carbon cycle. Further, the experimental verification of the predicted process as it developed over the following decades is discussed, as well as the extension of the initial carbon cycle to the carbon- nitrogen-oxygen (CNO) multi-cycles and the hot CNO cycles. This development emerged from the detailed experimental studies of the associated nuclear reactions over more than seven decades. Finally, the impact of the experimental and theoretical results on our present understanding of hydrogen burning in different stellar environments is presented, as well as the impact on our understanding of the chemical evolution of our universe. Key words: Carl Friedrich von Weizsa¨cker; Hans Bethe; Carbon cycle; CNO cycle. Introduction The energy source of the sun and all other stars became a topic of great interests in the physics community in the second half of the nineteenth century.