Ca2+ Microdomains, Calcineurin and the Regulation of Gene Transcription
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Regulation of Calcium/Calmodulin-Dependent Protein Kinase II Activation by Intramolecular and Intermolecular Interactions
8394 • The Journal of Neuroscience, September 29, 2004 • 24(39):8394–8398 Mini-Review Regulation of Calcium/Calmodulin-Dependent Protein Kinase II Activation by Intramolecular and Intermolecular Interactions Leslie C. Griffith Department of Biology and Volen Center for Complex Systems, Brandeis University, Waltham, Massachusetts 02454-9110 Key words: calcium; calmodulin; learning; localization; NMDA; phosphatase; protein kinase As its name implies, calcium/calmodulin-dependent protein ki- The overlap of these subdomains is no accident. Binding of nase II (CaMKII) is calcium dependent. In its basal state, the Ca 2ϩ/CaM is the primary signal for release of autoinhibition. activity of CaMKII is extremely low. Regulation of intracellular Current models of activation posit that the binding of Ca 2ϩ/CaM calcium levels allows the neuron to link activity with phosphor- serves to disrupt the interactions of specific residues within the ylation by CaMKII. This review will briefly summarize our cur- autoinhibitory domain with the catalytic domain (Smith et al., rent understanding of the intramolecular mechanisms of activity 1992). Because there is no crystal structure for the catalytic and regulation and their modulation by Ca 2ϩ/CaM and will then regulatory parts of CaMKII, the interaction face of these two focus on the growing number of other modes of intermolecular domains has been inferred using the effects of charge-reversal regulation of CaMKII activity by substrate and scaffolding mutagenesis on activity and molecular modeling (Yang and molecules. Schulman, 1999). This study confirmed the role of Arg 297 at the P-3 position of the pseudosubstrate ligand (Mukherji and Soder- Regulation of CaMKII by its autoinhibitory domain ling, 1995) and identified residues in the catalytic domain that All members of the CaMKII family (␣, , ␥, and ␦ isozymes) share may have direct interactions with the regulatory region. -

Cooperation to Amplify Gene-Dosage-Imbalance Effects
Update TRENDS in Molecular Medicine Vol.12 No.10 Research Focus Cooperation to amplify gene-dosage-imbalance effects Susana de la Luna1 and Xavier Estivill2 1 ICREA and Gene Function Group, Genes and Disease Program, Center for Genomic Regulation-CRG, 08003-Barcelona, Spain 2 Genetic Causes of Disease Group, Genes and Disease Program, Center for Genomic Regulation-CRG and Pompeu Fabra University, Barcelona Biomedical Research Park, 08003-Barcelona, Spain Trisomy 21, also known as Down syndrome (DS), is a From gene-dosage imbalance to pathology complex developmental disorder that affects many ThepresenceofanextracopyofHSA21 genes predicts an organs, including the brain, heart, skeleton and increased expression of 1.5-fold at the RNA level for immune system. A working hypothesis for understand- those genes in trisomy. Experiments in which this effect ing the consequences of trisomy 21 is that the over- has been evaluated indicate that this is indeed the case expression of certain genes on chromosome 21, alone for most HSA21 genes in DS samples and for their or in cooperation, is responsible for the clinical features orthologs in mouse trisomic models [3].Inthesimplest of DS. There is now compelling evidence that the scenario, the overexpression of one specific gene would protein products of two genes on chromosome 21, lead to the disturbance of a biological process and, as a Down syndrome candidate region 1 (DSCR1)and result, a single gene would be responsible for each patho- dual-specificity tyrosine-(Y)-phosphorylation regulated logical feature of DS. However, it is more probable that kinase 1A (DYRK1A), interact functionally, and that the overexpression of several of the 250 HSA21 genes their increased dosage cooperatively leads to dysregu- would contribute to alter a functional pathway in a lation of the signaling pathways that are controlled by specific cell at a specific time. -

RCAN2 Isoform 2 Recombinant Protein Cat
RCAN2 Isoform 2 Recombinant Protein Cat. No.: 95-114 RCAN2 Isoform 2 Recombinant Protein Specifications SPECIES: Mouse SOURCE SPECIES: E. coli SEQUENCE: aa 2 - 197 FUSION TAG: Fusion Partner: C-terminal His-tag TESTED APPLICATIONS: ELISA, WB APPLICATIONS: This recombinant protein can be used for WB and ELISA. For research use only. PREDICTED MOLECULAR 26 kDa (Calculated) WEIGHT: Properties PURITY: ~95% PHYSICAL STATE: Liquid 100mM sodium phosphate, 10mM Tris, 500mM NaCl, 25 mM imidazole, 2mM MgCl2, 10% BUFFER: gycerol Store in working aliquots at -70˚C. Avoid freeze/thaw cycles. When working with proteins STORAGE CONDITIONS: care should be taken to keep recombinant protein at a cool and stable temperature. September 29, 2021 1 https://www.prosci-inc.com/rcan2-isoform-2-recombinant-protein-95-114.html Additional Info OFFICIAL SYMBOL: Rcan2 RCAN2 Antibody: Csp2, MCIP2, ZAKI-4, Dscr1l1, Zaki4, Calcipressin-2, Calcineurin inhibitory ALTERNATE NAMES: protein ZAKI-4 ACCESSION NO.: AAH62141 PROTEIN GI NO.: 38328420 GENE ID: 53901 Background and References Regulator of calcineurin 2 (RCAN2), also known as ZAKI4 and DSCR1L1, is expressed as two isoforms differing at their N-terminus. The longer of the two (isoform 1) is expressed exclusively in the brain, while isoform 2 is ubiquitously expressed, with highest expression in brain, heart, and muscle (1,2). Both isoforms bind to the catalytic subunit of calcineurin, a Ca++-dependent protein phosphatase involved in several neuronal functions, though BACKGROUND: their C-terminal region and inhibit calcineurin’s activity (3). Unlike isoform 1 of RCAN2, the expression of the second isoform is not induced by the thyroid hormone T3 (3). -

Regulation of Calmodulin-Stimulated Cyclic Nucleotide Phosphodiesterase (PDE1): Review
95-105 5/6/06 13:44 Page 95 INTERNATIONAL JOURNAL OF MOLECULAR MEDICINE 18: 95-105, 2006 95 Regulation of calmodulin-stimulated cyclic nucleotide phosphodiesterase (PDE1): Review RAJENDRA K. SHARMA, SHANKAR B. DAS, ASHAKUMARY LAKSHMIKUTTYAMMA, PONNIAH SELVAKUMAR and ANURAAG SHRIVASTAV Department of Pathology and Laboratory Medicine, College of Medicine, University of Saskatchewan, Cancer Research Division, Saskatchewan Cancer Agency, 20 Campus Drive, Saskatoon SK S7N 4H4, Canada Received January 16, 2006; Accepted March 13, 2006 Abstract. The response of living cells to change in cell 6. Differential inhibition of PDE1 isozymes and its environment depends on the action of second messenger therapeutic applications molecules. The two second messenger molecules cAMP and 7. Role of proteolysis in regulating PDE1A2 Ca2+ regulate a large number of eukaryotic cellular events. 8. Role of PDE1A1 in ischemic-reperfused heart Calmodulin-stimulated cyclic nucleotide phosphodiesterase 9. Conclusion (PDE1) is one of the key enzymes involved in the complex interaction between cAMP and Ca2+ second messenger systems. Some PDE1 isozymes have similar kinetic and 1. Introduction immunological properties but are differentially regulated by Ca2+ and calmodulin. Accumulating evidence suggests that the A variety of cellular activities are regulated through mech- activity of PDE1 is selectively regulated by cross-talk between anisms controlling the level of cyclic nucleotides. These Ca2+ and cAMP signalling pathways. These isozymes are mechanisms include synthesis, degradation, efflux and seque- also further distinguished by various pharmacological agents. stration of cyclic adenosine 3':5'-monophosphate (cAMP) and We have demonstrated a potentially novel regulation of PDE1 cyclic guanosine 3':5'- monophosphate (cGMP) within the by calpain. -
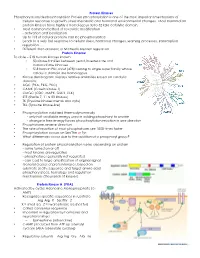
Protein Kinases Phosphorylation/Dephosphorylation Protein Phosphorylation Is One of the Most Important Mechanisms of Cellular Re
Protein Kinases Phosphorylation/dephosphorylation Protein phosphorylation is one of the most important mechanisms of cellular responses to growth, stress metabolic and hormonal environmental changes. Most mammalian protein kinases have highly a homologous 30 to 32 kDa catalytic domain. • Most common method of reversible modification - activation and localization • Up to 1/3 of cellular proteins can be phosphorylated • Leads to a very fast response to cellular stress, hormonal changes, learning processes, transcription regulation .... • Different than allosteric or Michealis Menten regulation Protein Kinome To date – 518 human kinases known • 50 kinase families between yeast, invertebrate and mammaliane kinomes • 518 human PKs, most (478) belong to single super family whose catalytic domain are homologous. • Kinase dendrogram displays relative similarities based on catalytic domains. • AGC (PKA, PKG, PKC) • CAMK (Casein kinase 1) • CMGC (CDC, MAPK, GSK3, CLK) • STE (Sterile 7, 11 & 20 kinases) • TK (Tryosine kinases memb and cyto) • TKL (Tyrosine kinase-like) • Phosphorylation stabilized thermodynamically - only half available energy used in adding phosphoryl to protein - change in free energy forces phosphorylation reaction in one direction • Phosphatases reverse direction • The rate of reaction of most phosphatases are 1000 times faster • Phosphorylation occurs on Ser/The or Tyr • What differences occur due to the addition of a phosphoryl group? • Regulation of protein phosphorylation varies depending on protein - some turned on or off -

Regulator of Calcineurin (RCAN): Beyond Down
Molecules and Cells Minireview Regulator of Calcineurin (RCAN): Beyond Down Syndrome Critical Region Sun-Kyung Lee1,2,* and Joohong Ahnn1,2,* 1Department of Life Science, 2Research Institute for Natural Sciences, College of Natural Sciences, Hanyang University, Seoul 04763, Korea *Correspondence: [email protected] (SKL); [email protected] (JA) https://doi.org/10.14348/molcells.2020.0060 www.molcells.org The regulator of calcineurin (RCAN) was first reported as RCAN3 a novel gene called DSCR1, encoded in a region termed the Down syndrome critical region (DSCR) of human chromosome 21. Genome sequence comparisons across INTRODUCTION species using bioinformatics revealed three members of the RCAN gene family, RCAN1, RCAN2, and RCAN3, present in The regulator of calcineurin (RCAN) was first reported as a most jawed vertebrates, with one member observed in most Down syndrome critical region 1 (DSCR1), which is encoded invertebrates and fungi. RCAN is most highly expressed in in a region that at that time was thought to participate in the brain and striated muscles, but expression has been reported onset of Down syndrome (DS) (Antonarakis, 2017; Fuentes in many other tissues, as well, including the heart and et al., 1995). Soon after, evidence showed that RCAN binds kidneys. Expression levels of RCAN homologs are responsive to and regulates the Ca2+/calmodulin-dependent serine/thre- to external stressors such as reactive oxygen species, Ca2+, onine phosphatase calcineurin, whose substrates include nu- amyloid β, and hormonal changes and upregulated in clear factor of activated T cells (NFAT), the transcription factor pathological conditions, including Alzheimer’s disease, that regulates gene expression in many cell types, including cardiac hypertrophy, diabetes, and degenerative neuropathy. -

The Genetic Architecture of Down Syndrome Phenotypes Revealed by High-Resolution Analysis of Human Segmental Trisomies
The genetic architecture of Down syndrome phenotypes revealed by high-resolution analysis of human segmental trisomies Jan O. Korbela,b,c,1, Tal Tirosh-Wagnerd,1, Alexander Eckehart Urbane,f,1, Xiao-Ning Chend, Maya Kasowskie, Li Daid, Fabian Grubertf, Chandra Erdmang, Michael C. Gaod, Ken Langeh,i, Eric M. Sobelh, Gillian M. Barlowd, Arthur S. Aylsworthj,k, Nancy J. Carpenterl, Robin Dawn Clarkm, Monika Y. Cohenn, Eric Dorano, Tzipora Falik-Zaccaip, Susan O. Lewinq, Ira T. Lotto, Barbara C. McGillivrayr, John B. Moeschlers, Mark J. Pettenatit, Siegfried M. Pueschelu, Kathleen W. Raoj,k,v, Lisa G. Shafferw, Mordechai Shohatx, Alexander J. Van Ripery, Dorothy Warburtonz,aa, Sherman Weissmanf, Mark B. Gersteina, Michael Snydera,e,2, and Julie R. Korenbergd,h,bb,2 Departments of aMolecular Biophysics and Biochemistry, eMolecular, Cellular, and Developmental Biology, and fGenetics, Yale University School of Medicine, New Haven, CT 06520; bEuropean Molecular Biology Laboratory, 69117 Heidelberg, Germany; cEuropean Molecular Biology Laboratory (EMBL) Outstation Hinxton, EMBL-European Bioinformatics Institute, Wellcome Trust Genome Campus, Hinxton, Cambridge CB10 1SA, United Kingdom; dMedical Genetics Institute, Cedars–Sinai Medical Center, Los Angeles, CA 90048; gDepartment of Statistics, Yale University, New Haven, CT 06520; Departments of hHuman Genetics, and iBiomathematics, University of California, Los Angeles, CA 90095; Departments of jPediatrics and kGenetics, University of North Carolina, Chapel Hill, NC 27599; lCenter for Genetic Testing, -

DYRK1A and RCAN1
BMB reports Mini Review Two key genes closely implicated with the neuropathological characteristics in Down syndrome: DYRK1A and RCAN1 Joongkyu Park#, Yohan Oh# & Kwang Chul Chung* Department of Biology, College of Life Science and Biotechnology, Yonsei University, Seoul 120-749, Korea The most common genetic disorder Down syndrome (DS) dis- 21) (2, 3). Other phenotypic features of DS include cognitive plays various developmental defects including mental re- impairment, learning and memory deficit, a high risk of leuke- tardation, learning and memory deficit, the early onset of mia, a decreased risk of solid tumors, congenital heart disease Alzheimer’s disease (AD), congenital heart disease, and cranio- and hypotonia (3-5). Moreover, DS brains show additional facial abnormalities. Those characteristics result from the ex- neuropathological outcomes such as the arrest of neurogenesis tra-genes located in the specific region called ‘Down syn- and synaptogenesis, and neuronal differentiation defects (6, 7). drome critical region (DSCR)’ in human chromosome 21. In They also exhibit a lower brain weight with reduced neuronal this review, we summarized the recent findings of the density, number, and volume regardless of region and age (6, DYRK1A and RCAN1 genes, which are located on DSCR and 8, 9). Although the cause of these CNS hypoplasias in DS pa- thought to be closely associated with the typical features of DS tients remains unclear, several reports using DS brains and cul- patients, and their implication to the pathogenesis of neural tured DS fibroblasts suggest that it may result from enhanced defects in DS. DYRK1A phosphorylates several transcriptional cell death and impaired cell proliferation (6, 7, 10, 11). -

Calcium/Calmodulin-Dependent Protein Kinase II: an Unforgettable Kinase
The Journal of Neuroscience, September 29, 2004 • 24(39):8391–8393 • 8391 Mini-Review Calcium/Calmodulin-Dependent Protein Kinase II: An Unforgettable Kinase Leslie C. Griffith Department of Biology and Volen Center for Complex Systems, Brandeis University, Waltham, Massachusetts 02454-9110 Key words: calcium; calmodulin; learning; localization; LTP; NMDA; phosphatase; protein kinase; knock-out mice The search for “memory molecules” and why CaMKII is an mers of different isozymes are able to coassemble, allowing for a appealing candidate large number of possible holoenzyme compositions. Measure- Since the time neuroscientists first recognized that biochemical ment of the Stoke’s radii and sedimentation coefficients of rat events inside neurons could influence the function of the brain, brain and Drosophila CaMKIIs suggested a holoenzyme of 8–12 there have been people looking for “memory molecules.” This subunits (Bennett et al., 1983; Kuret and Schulman, 1984; elusive ion, small molecule, protein, or nucleic acid would be the GuptaRoy and Griffith, 1996). Early electron microscopic studies keystone on which memory was built; understanding the mem- of CaMKII purified from rat brain found some heterogeneity in ory molecule would allow us to unlock the mysteries of cognition. holoenzyme size, reporting both 8 and 10 subunit particles (Ka- Models of how this molecule could work abounded, but the idea naseki et al., 1991). More recent single-particle studies, using of a memory molecule as a kinase/phosphatase-based molecular recombinant rat CaMKII, have provided much higher-resolution switch emerged as an important contender (Lisman, 1985). The (21–25 Å) images (Kolodziej et al., 2000; Gaertner et al., 2004). -

GSK-3 Kinases Enhance Calcineurin Signaling by Phosphorylation of Rcns
Downloaded from genesdev.cshlp.org on September 25, 2021 - Published by Cold Spring Harbor Laboratory Press GSK-3 kinases enhance calcineurin signaling by phosphorylation of RCNs Zoe Hilioti,1 Deirdre A. Gallagher,1 Shalini T. Low-Nam,1 Priya Ramaswamy,1 Pawel Gajer,1 Tami J. Kingsbury,1 Christine J. Birchwood,1 Andre Levchenko,2 andKyle W. Cunningham 1,3 1Department of Biology and 2Whitaker Institute for Biomedical Engineering, Johns Hopkins University, Baltimore, Maryland 21218, USA The conservedRCN family of proteins can bindanddirectlyregulate calcin eurin, a Ca2+-activatedprotein phosphatase involvedin immunity, heart growth, muscle development,learning, andother processes. Whereas high levels of RCNs can inhibit calcineurin signaling in fungal andanimal cells, RCNs can also stimulate calcineurin signaling when expressedat endogenouslevels. Here we show t hat the stimulatory effect of yeast Rcn1 involves phosphorylation of a conservedserine residueby Mck1, a mem ber of the GSK-3 family of protein kinases. Mutations at the GSK-3 consensus site of Rcn1 andhuman DS CR1/MCIP1 abolish the stimulatory effects on calcineurin signaling. RCNs may therefore oscillate between stimulatory andinhibitory forms in vivo in a manner similar to the Inhibitor-2 regulators of type 1 protein phosphatase. Computational modeling indicates a biphasic response of calcineurin to increasing RCN concentration such that protein phosphatase activity is stimulatedby low concentrations of phospho-RCN andinhibitedby high concentrations of phospho- or dephospho-RCN. This prediction was verifiedexperimentally in yeast cells expressing Rcn1 or DSCR1/MCIP1 at different concentrations. Through the phosphorylation of RCNs, GSK-3 kinases can potentially contribute to a positive feedback loop involving calcineurin-dependent up-regulation of RCN expression. -

Juvenile Hormone-Activated Phospholipase C Pathway PNAS PLUS Enhances Transcriptional Activation by the Methoprene-Tolerant Protein
Juvenile hormone-activated phospholipase C pathway PNAS PLUS enhances transcriptional activation by the methoprene-tolerant protein Pengcheng Liua, Hong-Juan Pengb, and Jinsong Zhua,1 aDepartment of Biochemistry, Virginia Polytechnic Institute and State University, Blacksburg, VA 24061; and bDepartment of Pathogen Biology, School of Public Health and Tropical Medicine, Southern Medical University, Guangzhou, Guangdong, 510515, China Edited by Lynn M. Riddiford, Howard Hughes Medical Institute Janelia Farm Research Campus, Ashburn, VA, and approved March 11, 2015 (received for review December 4, 2014) Juvenile hormone (JH) is a key regulator of a wide diversity of in the regulatory regions of JH-responsive genes, leading to developmental and physiological events in insects. Although the the transcriptional activation of these genes (12). This function intracellular JH receptor methoprene-tolerant protein (MET) func- of MET–TAI in the JH-induced gene expression seems to be tions in the nucleus as a transcriptional activator for specific JH- evolutionarily conserved in Ae. aegypti, D. melanogaster, the red regulated genes, some JH responses are mediated by signaling flour beetle Tribolium castaneum, the silkworm Bombyx mori,and pathways that are initiated by proteins associated with plasma the cockroach Bombyx mori (9, 13–16). membrane. It is unknown whether the JH-regulated gene expres- The mechanisms by which JH exerts pleiotropic functions are sion depends on the membrane-mediated signal transduction. In manifold in insects. Several studies suggest that JH can act via a Aedes aegypti mosquitoes, we found that JH activated the phos- receptor on plasma membrane (3, 17). For example, develop- pholipase C (PLC) pathway and quickly increased the levels of ino- ment of ovarian patency during vitellogenesis is stimulated by JH sitol 1,4,5-trisphosphate, diacylglycerol, and intracellular calcium, in some insects via transmembrane signaling cascades that in- leading to activation and autophosphorylation of calcium/calmod- volve second messengers (18, 19). -

A-Kinase Anchoring Protein–Calcineurin Signaling in Long-Term Depression of Gabaergic Synapses
2650 • The Journal of Neuroscience, February 6, 2013 • 33(6):2650–2660 Cellular/Molecular A-Kinase Anchoring Protein–Calcineurin Signaling in Long-Term Depression of GABAergic Synapses Matthieu Dacher, Shawn Gouty, Steven Dash, Brian M. Cox, and Fereshteh S. Nugent Department of Pharmacology, Uniformed Services University of the Health Sciences, Bethesda, Maryland 20814 The postsynaptic scaffolding A-kinase anchoring protein 79/150 (AKAP79/150) signaling complex regulates excitatory synaptic trans- mission and strength through tethering protein kinase A (PKA), PKC, and calcineurin (CaN) to the postsynaptic densities of neurons (Sanderson and Dell’Acqua, 2011), but its role in inhibitory synaptic transmission and plasticity is unknown. Using immunofluorescence and whole-cell patch-clamp recording in rat midbrain slices, we show that activation of postsynaptic D2-like family of dopamine (DA) receptor in the ventral tegmental area (VTA) induces long-term depression (LTD) of GABAergic synapses on DA neurons through an inositol triphosphate receptor-mediated local rise in postsynaptic Ca 2ϩ and CaN activation accompanied by PKA inhibition, which requires AKAP150 as a bridging signaling molecule. Our data also illuminate a requirement for a clathrin-mediated internalization of GABAA receptors in expression of LTDGABA. Moreover, disruption of AKAP–PKA anchoring does not affect glutamatergic synapses onto DA neurons, suggesting that the PKA–AKAP–CaN complex is uniquely situated at GABAA receptor synapses in VTA DA neurons to regulate plasticity associated with GABAA receptors. Drug-induced modulation of GABAergic plasticity in the VTA through such novel signaling mechanisms has the potential to persistently alter the output of individual DA neurons and of the VTA, which may contribute to the reinforcing or addictive properties of drugs of abuse.