The Calcium/Calmodulin-Dependent Kinases II and IV As Therapeutic Targets in Neurodegenerative and Neuropsychiatric Disorders
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Gene Symbol Gene Description ACVR1B Activin a Receptor, Type IB
Table S1. Kinase clones included in human kinase cDNA library for yeast two-hybrid screening Gene Symbol Gene Description ACVR1B activin A receptor, type IB ADCK2 aarF domain containing kinase 2 ADCK4 aarF domain containing kinase 4 AGK multiple substrate lipid kinase;MULK AK1 adenylate kinase 1 AK3 adenylate kinase 3 like 1 AK3L1 adenylate kinase 3 ALDH18A1 aldehyde dehydrogenase 18 family, member A1;ALDH18A1 ALK anaplastic lymphoma kinase (Ki-1) ALPK1 alpha-kinase 1 ALPK2 alpha-kinase 2 AMHR2 anti-Mullerian hormone receptor, type II ARAF v-raf murine sarcoma 3611 viral oncogene homolog 1 ARSG arylsulfatase G;ARSG AURKB aurora kinase B AURKC aurora kinase C BCKDK branched chain alpha-ketoacid dehydrogenase kinase BMPR1A bone morphogenetic protein receptor, type IA BMPR2 bone morphogenetic protein receptor, type II (serine/threonine kinase) BRAF v-raf murine sarcoma viral oncogene homolog B1 BRD3 bromodomain containing 3 BRD4 bromodomain containing 4 BTK Bruton agammaglobulinemia tyrosine kinase BUB1 BUB1 budding uninhibited by benzimidazoles 1 homolog (yeast) BUB1B BUB1 budding uninhibited by benzimidazoles 1 homolog beta (yeast) C9orf98 chromosome 9 open reading frame 98;C9orf98 CABC1 chaperone, ABC1 activity of bc1 complex like (S. pombe) CALM1 calmodulin 1 (phosphorylase kinase, delta) CALM2 calmodulin 2 (phosphorylase kinase, delta) CALM3 calmodulin 3 (phosphorylase kinase, delta) CAMK1 calcium/calmodulin-dependent protein kinase I CAMK2A calcium/calmodulin-dependent protein kinase (CaM kinase) II alpha CAMK2B calcium/calmodulin-dependent -

Regulation of Calcium/Calmodulin-Dependent Protein Kinase II Activation by Intramolecular and Intermolecular Interactions
8394 • The Journal of Neuroscience, September 29, 2004 • 24(39):8394–8398 Mini-Review Regulation of Calcium/Calmodulin-Dependent Protein Kinase II Activation by Intramolecular and Intermolecular Interactions Leslie C. Griffith Department of Biology and Volen Center for Complex Systems, Brandeis University, Waltham, Massachusetts 02454-9110 Key words: calcium; calmodulin; learning; localization; NMDA; phosphatase; protein kinase As its name implies, calcium/calmodulin-dependent protein ki- The overlap of these subdomains is no accident. Binding of nase II (CaMKII) is calcium dependent. In its basal state, the Ca 2ϩ/CaM is the primary signal for release of autoinhibition. activity of CaMKII is extremely low. Regulation of intracellular Current models of activation posit that the binding of Ca 2ϩ/CaM calcium levels allows the neuron to link activity with phosphor- serves to disrupt the interactions of specific residues within the ylation by CaMKII. This review will briefly summarize our cur- autoinhibitory domain with the catalytic domain (Smith et al., rent understanding of the intramolecular mechanisms of activity 1992). Because there is no crystal structure for the catalytic and regulation and their modulation by Ca 2ϩ/CaM and will then regulatory parts of CaMKII, the interaction face of these two focus on the growing number of other modes of intermolecular domains has been inferred using the effects of charge-reversal regulation of CaMKII activity by substrate and scaffolding mutagenesis on activity and molecular modeling (Yang and molecules. Schulman, 1999). This study confirmed the role of Arg 297 at the P-3 position of the pseudosubstrate ligand (Mukherji and Soder- Regulation of CaMKII by its autoinhibitory domain ling, 1995) and identified residues in the catalytic domain that All members of the CaMKII family (␣, , ␥, and ␦ isozymes) share may have direct interactions with the regulatory region. -

Mtor Coordinates Transcriptional Programs and Mitochondrial Metabolism of Activated Treg Subsets to Protect Tissue Homeostasis
ARTICLE DOI: 10.1038/s41467-018-04392-5 OPEN mTOR coordinates transcriptional programs and mitochondrial metabolism of activated Treg subsets to protect tissue homeostasis Nicole M. Chapman1, Hu Zeng 1, Thanh-Long M. Nguyen1, Yanyan Wang1, Peter Vogel 2, Yogesh Dhungana1, Xiaojing Liu3, Geoffrey Neale 4, Jason W. Locasale 3 & Hongbo Chi1 1234567890():,; Regulatory T (Treg) cells derived from the thymus (tTreg) and periphery (pTreg) have central and distinct functions in immunosuppression, but mechanisms for the generation and acti- vation of Treg subsets in vivo are unclear. Here, we show that mechanistic target of rapamycin (mTOR) unexpectedly supports the homeostasis and functional activation of tTreg and pTreg cells. mTOR signaling is crucial for programming activated Treg-cell function to protect immune tolerance and tissue homeostasis. Treg-specific deletion of mTOR drives sponta- neous effector T-cell activation and inflammation in barrier tissues and is associated with reduction in both thymic-derived effector Treg (eTreg) and pTreg cells. Mechanistically, mTOR functions downstream of antigenic signals to drive IRF4 expression and mitochondrial metabolism, and accordingly, deletion of mitochondrial transcription factor A (Tfam) severely impairs Treg-cell suppressive function and eTreg-cell generation. Collectively, our results show that mTOR coordinates transcriptional and metabolic programs in activated Treg subsets to mediate tissue homeostasis. 1 Department of Immunology, St. Jude Children’s Research Hospital, 262 Danny Thomas Place, MS 351, Memphis, TN 38105, USA. 2 Department of Pathology, St. Jude Children’s Research Hospital, 262 Danny Thomas Place, MS 250, Memphis, TN 38105, USA. 3 Department of Pharmacology & Cancer Biology, Duke University School of Medicine, Levine Science Research Center C266, Box 3813, Durham, NC 27710, USA. -

Support Info
Electronic Supplementary Material (ESI) for RSC Advances. This journal is © The Royal Society of Chemistry 2014 Supporting Information Design and synthesis of pyrrole–5-(2,6-dichlorobenzyl)sulfonylindolin-2-ones with C- 3’ side chains as potent Met kinase inhibitors Chia-Wei Liu,a Chun-Liang Lai,a Yu-Hsiang Lin,a Li-Wei Teng,a Sheng-chuan Yang,a Win-Yin Wei,a Shu Fu Lin,a Ju-Ying Yang,a Hung-Jyun Huang,a Ru-Wen Wang,a Chao-Cheng Chiang,a Mei-Hui Lee,a Yu- Chuan Wang,b Shih-Hsien Chuang,a Jia-Ming Chang,a Ying-Shuan E. Lee,a and Jiann-Jyh Huang*a,b aDevelopment Center for Biotechnology, No. 101, Lane 169, Kangning St., Xizhi District, New Taipei City 22180, Taiwan bDepartment of Applied Chemistry, National Chiayi University, No. 300, Syuefu Rd., Chiayi City 60004, Taiwan *Corresponding Author. Tel.: +886 5 271 7959; Fax: +886 5 271 7901. E-mail address: [email protected] (J.-J. Huang) Table of Contents: Page Supporting Figure. Ligplot diagrams of the ATP binding site of Met S2 complexed with compounds 2 and 20. Supporting Table. Kinase profiling data of compound 20. S3 References S10 - S1 - Supporting Figure. Ligplot diagrams1 of the ATP binding site of Met complexed with compounds 2 and 20: (A) Met with 2, and (B) Met with 20. - S2 - Supporting Table. Kinase profiling data of 20. Ambit KinomeScan Kinase Profiling (1.0 μM test concentration): Percentage of Percentage of Ambit Gene Symbol control (%) Ambit Gene Symbol control (%) 20 20 AAK1 68 ARK5 27 ABL1(E255K)-phosphorylated 85 ASK1 100 ABL1(F317I)-nonphosphorylated 78 ASK2 67 -

Profiling Data
Compound Name DiscoveRx Gene Symbol Entrez Gene Percent Compound Symbol Control Concentration (nM) JNK-IN-8 AAK1 AAK1 69 1000 JNK-IN-8 ABL1(E255K)-phosphorylated ABL1 100 1000 JNK-IN-8 ABL1(F317I)-nonphosphorylated ABL1 87 1000 JNK-IN-8 ABL1(F317I)-phosphorylated ABL1 100 1000 JNK-IN-8 ABL1(F317L)-nonphosphorylated ABL1 65 1000 JNK-IN-8 ABL1(F317L)-phosphorylated ABL1 61 1000 JNK-IN-8 ABL1(H396P)-nonphosphorylated ABL1 42 1000 JNK-IN-8 ABL1(H396P)-phosphorylated ABL1 60 1000 JNK-IN-8 ABL1(M351T)-phosphorylated ABL1 81 1000 JNK-IN-8 ABL1(Q252H)-nonphosphorylated ABL1 100 1000 JNK-IN-8 ABL1(Q252H)-phosphorylated ABL1 56 1000 JNK-IN-8 ABL1(T315I)-nonphosphorylated ABL1 100 1000 JNK-IN-8 ABL1(T315I)-phosphorylated ABL1 92 1000 JNK-IN-8 ABL1(Y253F)-phosphorylated ABL1 71 1000 JNK-IN-8 ABL1-nonphosphorylated ABL1 97 1000 JNK-IN-8 ABL1-phosphorylated ABL1 100 1000 JNK-IN-8 ABL2 ABL2 97 1000 JNK-IN-8 ACVR1 ACVR1 100 1000 JNK-IN-8 ACVR1B ACVR1B 88 1000 JNK-IN-8 ACVR2A ACVR2A 100 1000 JNK-IN-8 ACVR2B ACVR2B 100 1000 JNK-IN-8 ACVRL1 ACVRL1 96 1000 JNK-IN-8 ADCK3 CABC1 100 1000 JNK-IN-8 ADCK4 ADCK4 93 1000 JNK-IN-8 AKT1 AKT1 100 1000 JNK-IN-8 AKT2 AKT2 100 1000 JNK-IN-8 AKT3 AKT3 100 1000 JNK-IN-8 ALK ALK 85 1000 JNK-IN-8 AMPK-alpha1 PRKAA1 100 1000 JNK-IN-8 AMPK-alpha2 PRKAA2 84 1000 JNK-IN-8 ANKK1 ANKK1 75 1000 JNK-IN-8 ARK5 NUAK1 100 1000 JNK-IN-8 ASK1 MAP3K5 100 1000 JNK-IN-8 ASK2 MAP3K6 93 1000 JNK-IN-8 AURKA AURKA 100 1000 JNK-IN-8 AURKA AURKA 84 1000 JNK-IN-8 AURKB AURKB 83 1000 JNK-IN-8 AURKB AURKB 96 1000 JNK-IN-8 AURKC AURKC 95 1000 JNK-IN-8 -

Regulation of Calmodulin-Stimulated Cyclic Nucleotide Phosphodiesterase (PDE1): Review
95-105 5/6/06 13:44 Page 95 INTERNATIONAL JOURNAL OF MOLECULAR MEDICINE 18: 95-105, 2006 95 Regulation of calmodulin-stimulated cyclic nucleotide phosphodiesterase (PDE1): Review RAJENDRA K. SHARMA, SHANKAR B. DAS, ASHAKUMARY LAKSHMIKUTTYAMMA, PONNIAH SELVAKUMAR and ANURAAG SHRIVASTAV Department of Pathology and Laboratory Medicine, College of Medicine, University of Saskatchewan, Cancer Research Division, Saskatchewan Cancer Agency, 20 Campus Drive, Saskatoon SK S7N 4H4, Canada Received January 16, 2006; Accepted March 13, 2006 Abstract. The response of living cells to change in cell 6. Differential inhibition of PDE1 isozymes and its environment depends on the action of second messenger therapeutic applications molecules. The two second messenger molecules cAMP and 7. Role of proteolysis in regulating PDE1A2 Ca2+ regulate a large number of eukaryotic cellular events. 8. Role of PDE1A1 in ischemic-reperfused heart Calmodulin-stimulated cyclic nucleotide phosphodiesterase 9. Conclusion (PDE1) is one of the key enzymes involved in the complex interaction between cAMP and Ca2+ second messenger systems. Some PDE1 isozymes have similar kinetic and 1. Introduction immunological properties but are differentially regulated by Ca2+ and calmodulin. Accumulating evidence suggests that the A variety of cellular activities are regulated through mech- activity of PDE1 is selectively regulated by cross-talk between anisms controlling the level of cyclic nucleotides. These Ca2+ and cAMP signalling pathways. These isozymes are mechanisms include synthesis, degradation, efflux and seque- also further distinguished by various pharmacological agents. stration of cyclic adenosine 3':5'-monophosphate (cAMP) and We have demonstrated a potentially novel regulation of PDE1 cyclic guanosine 3':5'- monophosphate (cGMP) within the by calpain. -

Camk4 Compromises Podocyte Function in Autoimmune and Nonautoimmune Kidney Disease
CaMK4 compromises podocyte function in autoimmune and nonautoimmune kidney disease Kayaho Maeda, … , Maria G. Tsokos, George C. Tsokos J Clin Invest. 2018;128(8):3445-3459. https://doi.org/10.1172/JCI99507. Research Article Autoimmunity Nephrology Podocyte malfunction occurs in autoimmune and nonautoimmune kidney disease. Calcium signaling is essential for podocyte injury, but the role of Ca2+/calmodulin–dependent kinase (CaMK) signaling in podocytes has not been fully explored. We report that podocytes from patients with lupus nephritis and focal segmental glomerulosclerosis and lupus- prone and lipopolysaccharide- or adriamycin-treated mice display increased expression of CaMK IV (CaMK4), but not CaMK2. Mechanistically, CaMK4 modulated podocyte motility by altering the expression of the GTPases Rac1 and RhoA and suppressed the expression of nephrin, synaptopodin, and actin fibers in podocytes. In addition, it phosphorylated the scaffold protein 14-3-3β, which resulted in the release and degradation of synaptopodin. Targeted delivery of a CaMK4 inhibitor to podocytes preserved their ultrastructure, averted immune complex deposition and crescent formation, and suppressed proteinuria in lupus-prone mice and proteinuria in mice exposed to lipopolysaccharide-induced podocyte injury by preserving nephrin/synaptopodin expression. In animals exposed to adriamycin, podocyte-specific delivery of a CaMK4 inhibitor prevented and reversed podocyte injury and renal disease. We conclude that CaMK4 is pivotal in immune and nonimmune podocyte injury and that its targeted cell-specific inhibition preserves podocyte structure and function and should have therapeutic value in lupus nephritis and podocytopathies, including focal segmental glomerulosclerosis. Find the latest version: https://jci.me/99507/pdf The Journal of Clinical Investigation RESEARCH ARTICLE CaMK4 compromises podocyte function in autoimmune and nonautoimmune kidney disease Kayaho Maeda,1 Kotaro Otomo,1 Nobuya Yoshida,1 Mones S. -
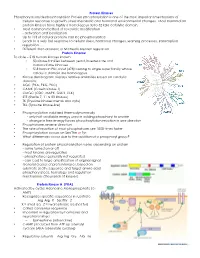
Protein Kinases Phosphorylation/Dephosphorylation Protein Phosphorylation Is One of the Most Important Mechanisms of Cellular Re
Protein Kinases Phosphorylation/dephosphorylation Protein phosphorylation is one of the most important mechanisms of cellular responses to growth, stress metabolic and hormonal environmental changes. Most mammalian protein kinases have highly a homologous 30 to 32 kDa catalytic domain. • Most common method of reversible modification - activation and localization • Up to 1/3 of cellular proteins can be phosphorylated • Leads to a very fast response to cellular stress, hormonal changes, learning processes, transcription regulation .... • Different than allosteric or Michealis Menten regulation Protein Kinome To date – 518 human kinases known • 50 kinase families between yeast, invertebrate and mammaliane kinomes • 518 human PKs, most (478) belong to single super family whose catalytic domain are homologous. • Kinase dendrogram displays relative similarities based on catalytic domains. • AGC (PKA, PKG, PKC) • CAMK (Casein kinase 1) • CMGC (CDC, MAPK, GSK3, CLK) • STE (Sterile 7, 11 & 20 kinases) • TK (Tryosine kinases memb and cyto) • TKL (Tyrosine kinase-like) • Phosphorylation stabilized thermodynamically - only half available energy used in adding phosphoryl to protein - change in free energy forces phosphorylation reaction in one direction • Phosphatases reverse direction • The rate of reaction of most phosphatases are 1000 times faster • Phosphorylation occurs on Ser/The or Tyr • What differences occur due to the addition of a phosphoryl group? • Regulation of protein phosphorylation varies depending on protein - some turned on or off -

Supplementary Table 1. in Vitro Side Effect Profiling Study for LDN/OSU-0212320. Neurotransmitter Related Steroids
Supplementary Table 1. In vitro side effect profiling study for LDN/OSU-0212320. Percent Inhibition Receptor 10 µM Neurotransmitter Related Adenosine, Non-selective 7.29% Adrenergic, Alpha 1, Non-selective 24.98% Adrenergic, Alpha 2, Non-selective 27.18% Adrenergic, Beta, Non-selective -20.94% Dopamine Transporter 8.69% Dopamine, D1 (h) 8.48% Dopamine, D2s (h) 4.06% GABA A, Agonist Site -16.15% GABA A, BDZ, alpha 1 site 12.73% GABA-B 13.60% Glutamate, AMPA Site (Ionotropic) 12.06% Glutamate, Kainate Site (Ionotropic) -1.03% Glutamate, NMDA Agonist Site (Ionotropic) 0.12% Glutamate, NMDA, Glycine (Stry-insens Site) 9.84% (Ionotropic) Glycine, Strychnine-sensitive 0.99% Histamine, H1 -5.54% Histamine, H2 16.54% Histamine, H3 4.80% Melatonin, Non-selective -5.54% Muscarinic, M1 (hr) -1.88% Muscarinic, M2 (h) 0.82% Muscarinic, Non-selective, Central 29.04% Muscarinic, Non-selective, Peripheral 0.29% Nicotinic, Neuronal (-BnTx insensitive) 7.85% Norepinephrine Transporter 2.87% Opioid, Non-selective -0.09% Opioid, Orphanin, ORL1 (h) 11.55% Serotonin Transporter -3.02% Serotonin, Non-selective 26.33% Sigma, Non-Selective 10.19% Steroids Estrogen 11.16% 1 Percent Inhibition Receptor 10 µM Testosterone (cytosolic) (h) 12.50% Ion Channels Calcium Channel, Type L (Dihydropyridine Site) 43.18% Calcium Channel, Type N 4.15% Potassium Channel, ATP-Sensitive -4.05% Potassium Channel, Ca2+ Act., VI 17.80% Potassium Channel, I(Kr) (hERG) (h) -6.44% Sodium, Site 2 -0.39% Second Messengers Nitric Oxide, NOS (Neuronal-Binding) -17.09% Prostaglandins Leukotriene, -

Supplementary Material DNA Methylation in Inflammatory Pathways Modifies the Association Between BMI and Adult-Onset Non- Atopic
Supplementary Material DNA Methylation in Inflammatory Pathways Modifies the Association between BMI and Adult-Onset Non- Atopic Asthma Ayoung Jeong 1,2, Medea Imboden 1,2, Akram Ghantous 3, Alexei Novoloaca 3, Anne-Elie Carsin 4,5,6, Manolis Kogevinas 4,5,6, Christian Schindler 1,2, Gianfranco Lovison 7, Zdenko Herceg 3, Cyrille Cuenin 3, Roel Vermeulen 8, Deborah Jarvis 9, André F. S. Amaral 9, Florian Kronenberg 10, Paolo Vineis 11,12 and Nicole Probst-Hensch 1,2,* 1 Swiss Tropical and Public Health Institute, 4051 Basel, Switzerland; [email protected] (A.J.); [email protected] (M.I.); [email protected] (C.S.) 2 Department of Public Health, University of Basel, 4001 Basel, Switzerland 3 International Agency for Research on Cancer, 69372 Lyon, France; [email protected] (A.G.); [email protected] (A.N.); [email protected] (Z.H.); [email protected] (C.C.) 4 ISGlobal, Barcelona Institute for Global Health, 08003 Barcelona, Spain; [email protected] (A.-E.C.); [email protected] (M.K.) 5 Universitat Pompeu Fabra (UPF), 08002 Barcelona, Spain 6 CIBER Epidemiología y Salud Pública (CIBERESP), 08005 Barcelona, Spain 7 Department of Economics, Business and Statistics, University of Palermo, 90128 Palermo, Italy; [email protected] 8 Environmental Epidemiology Division, Utrecht University, Institute for Risk Assessment Sciences, 3584CM Utrecht, Netherlands; [email protected] 9 Population Health and Occupational Disease, National Heart and Lung Institute, Imperial College, SW3 6LR London, UK; [email protected] (D.J.); [email protected] (A.F.S.A.) 10 Division of Genetic Epidemiology, Medical University of Innsbruck, 6020 Innsbruck, Austria; [email protected] 11 MRC-PHE Centre for Environment and Health, School of Public Health, Imperial College London, W2 1PG London, UK; [email protected] 12 Italian Institute for Genomic Medicine (IIGM), 10126 Turin, Italy * Correspondence: [email protected]; Tel.: +41-61-284-8378 Int. -

Camk4 Gene Deletion Induces Hypertension
ORIGINAL RESEARCH CaMK4 Gene Deletion Induces Hypertension Gaetano Santulli, MD; Ersilia Cipolletta, MD; Daniela Sorriento, PhD; Carmine Del Giudice, MS; Antonio Anastasio, MS; Sara Monaco, PhD; Angela Serena Maione, BS; Gianluigi Condorelli, MD, PhD; Annibale Puca, MD, PhD; Bruno Trimarco, MD; Maddalena Illario, MD, PhD; Guido Iaccarino, MD, PhD Background-—The expression of calcium/calmodulin-dependent kinase IV (CaMKIV) was hitherto thought to be confined to the nervous system. However, a recent genome-wide analysis indicated an association between hypertension and a single-nucleotide polymorphism (rs10491334) of the human CaMKIV gene (CaMK4), which suggests a role for this kinase in the regulation of vascular tone. Methods and Results-—To directly assess the role of CaMKIV in hypertension, we characterized the cardiovascular phenotype of CaMK4À/À mice. They displayed a typical hypertensive phenotype, including high blood pressure levels, cardiac hypertrophy, vascular and kidney damage, and reduced tolerance to chronic ischemia and myocardial infarction compared with wild-type littermates. Interestingly, in vitro experiments showed the ability of this kinase to activate endothelial nitric oxide synthase. Eventually, in a population study, we found that the rs10491334 variant associates with a reduction in the expression levels of CaMKIV in lymphocytes from hypertensive patients. Conclusions-—Taken together, our results provide evidence that CaMKIV plays a pivotal role in blood pressure regulation through the control of endothelial nitric oxide synthase activity. ( J Am Heart Assoc. 2012;1:e001081 doi: 10.1161/JAHA.112.001081) Key Words: angiogenesis • arrhythmia • endothelium • hypertension • hypertrophy growing body of evidence bears out the rising interest in single-nucleotide polymorphism (SNP) of the human CaMKIV A the function of calcium/calmodulin-dependent kinases gene (CaMK4). -

Can PDE Inhibition Improve Cognition? : Translational Insights
Can PDE inhibition improve cognition? : Translational insights Citation for published version (APA): Reneerkens, O. A. H. (2013). Can PDE inhibition improve cognition? : Translational insights. Maastricht University. https://doi.org/10.26481/dis.20130418or Document status and date: Published: 01/01/2013 DOI: 10.26481/dis.20130418or Document Version: Publisher's PDF, also known as Version of record Please check the document version of this publication: • A submitted manuscript is the version of the article upon submission and before peer-review. There can be important differences between the submitted version and the official published version of record. People interested in the research are advised to contact the author for the final version of the publication, or visit the DOI to the publisher's website. • The final author version and the galley proof are versions of the publication after peer review. • The final published version features the final layout of the paper including the volume, issue and page numbers. Link to publication General rights Copyright and moral rights for the publications made accessible in the public portal are retained by the authors and/or other copyright owners and it is a condition of accessing publications that users recognise and abide by the legal requirements associated with these rights. • Users may download and print one copy of any publication from the public portal for the purpose of private study or research. • You may not further distribute the material or use it for any profit-making activity or commercial gain • You may freely distribute the URL identifying the publication in the public portal. If the publication is distributed under the terms of Article 25fa of the Dutch Copyright Act, indicated by the “Taverne” license above, please follow below link for the End User Agreement: www.umlib.nl/taverne-license Take down policy If you believe that this document breaches copyright please contact us at: [email protected] providing details and we will investigate your claim.