The New International System of Units: the Role of the Committee on Data for Science and Technology (CODATA)
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Avogadro Number, Boltzmann Constant, Planck Constant, Information Theory, Comparative and Relative Uncertainties
American Journal of Computational and Applied Mathematics 2018, 8(5): 93-102 DOI: 10.5923/j.ajcam.20180805.02 h, k, NA: Evaluating the Relative Uncertainty of Measurement Boris Menin Mechanical & Refrigeration Consultation Expert, Beer-Sheba, Israel Abstract For verification of the measurement accuracy of fundamental physical constants, the CODATA unique technique is used. This procedure is a complex process based on a careful discussion of the input data, and the justification and construction of tables of values sufficient for the direct use of the relative uncertainty are conducted using modern advanced statistical methods and powerful computers. However, at every stage of data processing, researchers must rely on common sense, that is, an expert conclusion. In this article, the author proposes a theoretical and informational grounded justification for calculating the relative uncertainty by using the comparative uncertainty. A detailed description of the data and the processing procedures do not require considerable time. Examples of measurements results of three fundamental constants are analysed, applying information-oriented approach. Comparison of the achieved relative and comparative uncertainties is introduced and discussed. Keywords Avogadro number, Boltzmann constant, Planck constant, Information theory, Comparative and relative uncertainties theories of physics can be proved by carefully studying the 1. Introduction numerical values of these constants, determined from different experiments in different fields of physics. It is expected that in 2018 that the International System of One needs to note the main feature of the CODATA Units (SI) will be redefined on the basis of certain values of technique in determining the relative uncertainty of one some fundamental constants [1]. -

X-Ray Crystal Density Method to Determine the Avogadro and Planck Constants
X-ray Crystal Density Method to Determine the Avogadro and Planck Constants Horst Bettin Physikalisch-Technische Bundesanstalt Braunschweig, Germany Proposed New Definition of the Kilogram The kilogram, symbol kg, is the SI unit of mass. It is defined by taking the fixed numerical value of the Planck constant h to be 6.626 070 040 x 10 -34 when expressed in the unit J s, which is equal to kg m2 s-1, where the metre and the second c ∆ν are defined in terms of and Cs . *) X represents one or more digits to be added at the time the new definition is finally adopted. α 2 - = M (e )c NAh 2 R∞ N h −10 A = 3.990 312 7110(18) × 10 Js/mol, Amedeo Avogadro Max Planck with relative uncertainty of 0.45 × 10 -9 (1776-1856) (1858-1947) Page 2 of 19 Avogadro Constant Definition of Avogadro constant NA • Number of molecules per mol • 6.022... x 10 23 mol -1 Amedeo Avogadro Current definition of mol (1776-1856) • Number “entities” like 12 C atoms in 12 g • i. e. 6.022... x 10 23 12 C atoms have a mass of 12 g 12 12 g/mol = NA m( C ) Faraday constant F = NA e (e: elementary charge) Molar gas constant R = NA k (k: Boltzmann constant) Page 3 of 19 Counting Atoms: XRCD Method 3 Use of a silicon crystal! 1. Volume a0 of the unit cell 3 2. Volume of an atom: a0 /8 3. Volume V of a sphere 4. Number N of the atoms 8 V8 VM N = = ⋅ mol A N 3 3 a0 a0msphere Page 4 of 19 Lattice parameter measurement (INRIM) d220 (2011)=192014712.67(67) am d220 (2014)=192014711.98(34) am -9 ur(2014) = 1.8 x 10 Page 5 of 19 Lattice parameter set-up at PTB S M1 M M2 AL AA OH Page 6 of 19 Sphere Interferometer of PTB mK-temperature stabilisation Camera 1 Camera 2 Fizeau- Fizeau- Collimator Objective 1 Objective 2 Diode laser Page 7 of 19 Diameter results (2014) Relative Mean apparent Sphere Lab. -

2 Weighing and Preparation of Aqueous Solutions
2 WEIGHING AND PREPARATION OF AQUEOUS SOLUTIONS Theory In chemistry, a solution is a homogeneous mixture composed of two or more substances. In such a mixture, a solute is dissolved in another substance, known as a solvent. An aqueous solution is a solution in which the solvent is water. Concentration is the measure of how much of a given substance (solute) there is mixed with another substance (solvent). This can apply to any sort of chemical mixture, but most frequently the concept is limited to homogeneous solutions, where it refers to the amount of solute in a solution. For scientific or technical applications, concentration is expressed in a quantitative way. There are a number of different ways to quantitatively express concentration; the most common are listed below. They are based on mass or volume or both. Depending on what they are based on it is not always trivial to convert one measure to the other, because knowledge of the density might be needed to do so. At times this information may not be available, particularly if the temperature varies. Mass can be determined at a precision of ~ 0.1 mg on a routine basis with an analytical balance. Both solids and liquids are easily quantified by weighing. Some units of concentration - particularly the most popular one (molarity) - require to express the mass of substance in the moles. The mol is the SI basic unit that measures an amount of substance. „One mole contains exactly 6.022 140 76 × 1023 elementary entities. This number is the −1 fixed numerical value of the Avogadro constant, NA, when expressed in the unit mol and is called the Avogadro number.“ The previous definition was based on the number of carbon atoms in 12 g of material and was the following; a mol is the amount of substance of a system which contains as many elementary entities as there are atoms in 0.012 kilogram (or 12 g) of carbon-12 (12C), where the carbon – 12 atoms are unbound, at rest and in their ground state. -

Topic720 Composition: Mole Fraction: Molality: Concentration a Solution Comprises at Least Two Different Chemical Substances
Topic720 Composition: Mole Fraction: Molality: Concentration A solution comprises at least two different chemical substances where at least one substance is in vast molar excess. The term ‘solution’ is used to describe both solids and liquids. Nevertheless the term ‘solution’ in the absence of the word ‘solid’ refers to a liquid. Chemists are particularly expert at identifying the number and chemical formulae of chemical substances present in a given closed system. Here we explore how the chemical composition of a given system is expressed. We consider a simple system prepared using water()l and urea(s) at ambient temperature and pressure. We designate water as chemical substance 1 and urea as chemical substance j, so that the closed system contains an aqueous solution. The amounts of the two substances are given by n1 = = ()wM11 and nj ()wMjj where w1 and wj are masses; M1 and Mj are the molar masses of the two chemical substances. In these terms, n1 and nj are extensive variables. = ⋅ + ⋅ Mass of solution, w n1 M1 n j M j (a) = ⋅ Mass of solvent, w1 n1 M1 (b) = -1 For water, M1 0.018 kg mol . However in reviewing the properties of solutions, chemists prefer intensive composition variables. Mole Fraction The mole fractions of the two substances x1 and xj are given by the following two equations: =+ =+ xnnn111()j xnnnjj()1 j (c) += Here x1 x j 10. In general terms for a system comprising i - chemical substances, the mole fraction of substance k is given by equation (d). ji= = xnkk/ ∑ n j (d) j=1 ji= = Hence ∑ x j 10. -

Avogadro's Constant
Avogadro's Constant Nancy Eisenmenger Question Artem, 8th grade How \Avogadro constant" was invented and how scientists calculated it for the first time? Answer What is Avogadro's Constant? • Avogadros constant, NA: the number of particles in a mole • mole: The number of Carbon-12 atoms in 0.012 kg of Carbon-12 23 −1 • NA = 6:02214129(27) × 10 mol How was Avogadro's Constant Invented? Avogadro's constant was invented because scientists were learning about measuring matter and wanted a way to relate the microscopic to the macroscopic (e.g. how many particles (atoms, molecules, etc.) were in a sample of matter). The first scientists to see a need for Avogadro's constant were studying gases and how they behave under different temperatures and pressures. Amedeo Avogadro did not invent the constant, but he did state that all gases with the same volume, pressure, and temperature contained the same number of gas particles, which turned out to be very important to the development of our understanding of the principles of chemistry and physics. The constant was first calculated by Johann Josef Loschmidt, a German scientist, in 1865. He actually calculated the Loschmidt number, a constant that measures the same thing as Avogadro's number, but in different units (ideal gas particles per cubic meter at 0◦C and 1 atm). When converted to the same units, his number was off by about a factor of 10 from Avogadro's number. That may sound like a lot, but given the tools he had to work with for both theory and experiment and the magnitude of the number (∼ 1023), that is an impressively close estimate. -

2.1 Relative Atomic and Molecular Masses, the Avogadro Constant, and the Mole Calculations AQA Chemistry
2.1 Relative atomic and molecular AQA Chemistry masses, the Avogadro constant, and the mole Calculations The Avogadro constant Specification reference 3.1.2.2 MS 0.1 Recognise and use expressions in decimal and ordinary form MS 0.4 Use calculators to find and use power, exponential, and logarithmic functions MS 1.1Use an appropriate number of significant figures. Learning objectives After completing the worksheet you should be able to: carry out calculations using the Avogadro constant carry out calculations using numbers in standard and ordinary form substitute numerical values into algebraic equations, and change the subject of equations report calculations to an appropriate number of significant figures. Introduction One mole of any substance contains the same number of particles as the number of atoms in 12 g of carbon-12. This number of particles is a huge number and is called the Avogadro constant. It has a value of 6.02 1023 and is given the units of mol−1 (per mole). The relative atomic mass of an element in grams contains 6.02 1023 atoms or one mole of that element. The relative formula mass of a compound in grams contains 6.02 1023 particles (molecules or ions) or one mole of that compound. Therefore we use the term molar mass to describe the relative atomic or formula mass of a substance in grams and give it the unit g mol−1. To calculate the number of particles in a certain mass of a substance we can use the following equation. mass m (g) Number of particles = ´ 6.02 ´ 1023(mol-1) molar mass M (g mol-1) Worked example Question 1 How many molecules are there in 42.5 g of ammonia, NH3? Answer © Oxford University Press 2015 www.oxfordsecondary.co.uk/acknowledgements This resource sheet may have been changed from the original 1 2.1 Relative atomic and molecular AQA Chemistry masses, the Avogadro constant, and the mole Calculations Step 1 Calculate the molar mass of ammonia using the relative atomic masses of nitrogen and hydrogen from the periodic table. -
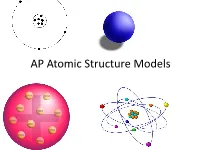
AP Atomic Structure Models What Is a Model?
AP Atomic Structure Models What is a Model? On a scrap piece of paper, write down your definition of a model with at least two examples. A model is a representation of an object, idea, action, or concept. Models A model in science is usually an approximation of the “real thing.” This means that all models have simplifications that may lead to omitted information. Models When a model is selected for a specific purpose, it should be able to help predict a “behavior.” The Four Historical Models of the Atom 1. Particle model 2. Plum Pudding model 3. Rutherford model 4. Bohr model Modern Model Quantum Mechanical Model Why do we have so many historical models? Are some of them still useful? When we initially began the process of determining the structure of the atom, we first had to overcome the prevailing scientific concept that matter was continuous. This idea of continuous matter was from Aristotle. (a Greek philosopher 384-322 B.C.E.) Particle Model John Dalton’s four ideas about the particle nature of matter helped push forward the acceptance of the first model. 1. All elements are composed of tiny indivisible particles called atoms. 2. Atoms of the same element are identical. The atoms of any one element are different from those of any other element. John Dalton 3. Atoms of different elements can physically mix together or can chemically combine in simple whole-number ratios to form compounds. 4. Chemical reactions occur when atoms are separated, joined, or rearranged. Atoms of one element, however, are never changed into atoms of another element as a result of a chemical reaction. -

Chemistry-I Content
CLASS-11 th THE CENTRAL BOARD OF SECONDARY EDUCATION CHEMISTRY-I CONTENT Unit 1 1 Some basic concepts of chemistry Unit 2 21 Structure of Atom Unit 3 51 Classification of element and Periodicity in Properties Unit 4 80 Chemical bonding and molecular structure Unit 5 111 States of matter: Gases and Liquids Unit 6 134 Chemical Thermodynamics Unit 7 164 Equilibrium CHAPTER 1‒SOME BASIC CONCEPTS OF CHEMISTRY Content V Nature of matter V Properties of matter and their measurement V Uncertainty in measurement V Laws of chemical combination V Dalton’s atomic theory V Atomic and molecular masses V Percentage composition V Stoichiometry and stoichiometric calculation V Important Questions Importance of Chemistry 1) Many types of drugs, fairness products, medicines and many more things which we use in daily life are because of chemistry. 2) Drugs effective in cancer therapy – Cisplatin and Taxol. 3) Drug used for AIDS patients-AZT (azidothymidine). Nature of Matter Matter It is defined as “Anything that has mass, occupies space and can be felt by any one or more senses is called matter”. Physical classification of matter On the basis of physical properties matter can be classified into five states as discussed below 1) Solid 2) Liquid 3) Gas 4) Plasma (highest energetic state of matter) 5) BEC (Bose - Einstein condensate) But we usually study only first three because of their vast existence in our environment. Properties of matter 1) Solid It possesses definite shape and definite volume which means it cannot be compressed on applying pressure on it. www.toppersnotes.com 1 • Fixed shape and volume • Intermolecular space is very low • Intermolecular forces are very strong • They have rigidity (resistance to change in shape) • Rocks, metal, wood, Iron, copper sulphate, bricks etc. -

8-1 SECTION 8 AMOUNT of SUBSTANCE and ITS UNIT, the MOLE Amount of Substance: Symbol N, a Quantity Fundamental to Chemistry
8-1 SECTION 8 AMOUNT OF SUBSTANCE AND ITS UNIT, THE MOLE Suppose a chemist wishes to carry out the chemical reaction of adding bromine to hexene to give dibromohexane, C6H12 + Br2 → C6H12Br2, starting with a known amount of hexene. How does the chemist know how much bromine is needed? The chemical equation tells you that one molecule of dibromine is needed for each molecule of hexene. But the very large numbers of molecules required for reactions on a practicable scale cannot be counted. If the mass of hexene is known how could the mass of bromine required be calculated? This section tells how this type of question is answered. It requires the introduction of a new quantity specific to chemistry , amount of substance, and it it th l Amount of substance: symbol n, a quantity fundamental to chemistry. Atoms and molecules are much too small or light to be counted or weighed individually in the laboratory. The chemist therefore needs a unit to specify the quantity amount of substance of an appropriate magnitude (size) for laboratory or industrial scale work. The chosen unit is the mole. Mole: symbol mol, the unit of the quantity amount of substance. The mole is defined as the amount of substance of a system which contains as many elementary entities as there are atoms in 12 grams of carbon-12 (i.e. carbon consisting only of the isotope 12C). 12 g is an easily measurable mass. When the mole is used, the elementary entities must be specified and may be atoms, molecules, ions, electrons, other particles or specified groups of such particles. -

Derivation of Photon Mass and Avogadro Constant from Planck Units
Derivation of Photon Mass and Avogadro Constant from Planck units B. Ravi Sankar, ISRO Satellite Centre. (Dated: October 5, 2016) Originally proposed in 1899 by German physicist Max Planck, Planck units are also known as natural units because the origin of their definition comes only from properties of the fundamental physical theories and not from interchangeable experimental param- eters. It is widely accepted that Planck units are the most fundamental units. In this paper, few more fundamental constants are derived from Planck units. These constants are permutations and combinations of Planck units and hence by construct, they are also constants. The mass and radius of photon are derived. The Avogadro constant, Boltzmann constant and unified mass unit are also derived. The structure of the photon is explained. The meaning of Avogadro constant in terms of photon structure is also explained. The meaning of Planck mass is explained. As proof for the meaning of the Planck mass, the solar constant is derived. The solar constant is derived applying string theory as well. Finally revised Planck current, Planck voltage and Planck impedance are also derived. It is also proven that Planck mass is the energy emitted by any star per second per ray of proper length c. Apart from this, the energy emitted per second per ray of proper length c by a planet or communication antenna is not equal to Planck mass. I. INTRODUCTION TABLE I Base Planck units In physics, Planck units are physical units of mea- Name Expression Value (SI units) surement defined in terms of five universal physical con- q Planck mass m = ~c 2:17651 × 10−8 stants.They are particularly relevant in research on uni- p G q G~ −34 fied theories such as quantum gravity. -

Amount of Substance and Its Unit – Mole- Connecting the Invisible Micro World to the Observable Macro World Part 2 (English, Mp4)
Amount of Substance and Its Unit – Mole- Connecting the Invisible Micro World to the Observable Macro World Part 2 (English, mp4) [MUSIC PLAYING] Instructor: Hi, everyone. Welcome back. I hope you had some fun watching the BLOSSOMS lesson, How Big is a Mole? Did you find an answer to the question about eating rice? Can you believe it? My estimate is that it would take about seven million years for the people on earth to eat one mole of rice. Was your estimate close to this range? Are you impressed with the incredible size of a mole, which is around 6.02 times 10 to the 23rd. This number also goes by the name of Avogadro's number. And now I want to tell you how it got that name. Avogadro's number is named after the early 19th century Italian scientist Amedeo Avogadro, who, in 1811, first proposed that equal volumes of gases under the same conditions contain the same number of molecules. This hypothesis proved useful in determining the atomic and the molecular weights, and eventually led to the concept of a mole. Many years later, the French physicist Jean Perrin proposed naming the constant in honor of Avogadro. But Dr. Avogadro himself, during his life, never saw this number that is now named after him. Avogadro's number, NA is equal to 6.02 times 10 to the 23rd, like any pure number is dimensionless, which means there is no unit attached to it. However, it also defines the mole. So we can express NA as 6.02 times 10 to the 23rd per mole. -

Molecular Sociology: Postgenome Science Upsets the Avogadro's the Maximum Soluble Concentration of the Plasma Main Component Number
nalytic A al & B y i tr o s c i h e Lisitsa et al., Biochem Anal Biochem 2015, 4:4 m m e Biochemistry & i h s c t DOI: 10.4172/2161-1009.1000216 r o i y B ISSN: 2161-1009 Analytical Biochemistry Research Article Open Access Molar Concentration Welcomes Avogadro in Postgenomic Analytics Andrey V Lisitsa*, Elena A Ponomarenko, Olga I Kiseleva, Ekaterina V Poverennaya and Alexander I Archakov Institute of Biomedical Chemistry, 119121, Pogodinskaya Street 10, Moscow, Russian Federation Abstract The researchers working with high-throughput methods of genomics, transcriptomics, and proteomics reconsider the concept of concentration and evaluate the data obtained in the number of copies of biomacromolecules. Measurement of copy number reflects a steady trend in increasing the sensitivity of postgenomic analytical methods, up to the level of a single molecule. In this paper we review the physical meaning of the terms "molar concentration" and "Avogadro’s number" to establish a relationship between them. The relationship between the molar concentration and the number of copies of that same macromolecule in a certain volume is set through the reverse Avogadro’s number, the value of which (10-24 М) characterizes the molar concentration of a single molecule in 1 liter. Using the reverse Avogadro’s number, we deal with situations in analyzing homogeneous biological solutions and heterogeneous cellular material. Keywords: Avogadro’s number; Protein copy number; Blood plasma; that can typically represent all 4-5 liters of blood in human body. At the Cell line same time, the sensitivity of an analytical method should be taken into account, since the more sensitive method, the lesser amount of blood is Introduction required for an analysis.