A Bendamustine Resistance Gene Signature in Diffuse Large B-Cell Lymphoma and Multiple Myeloma
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Hodgkin Lymphoma Treatment Regimens
HODGKIN LYMPHOMA TREATMENT REGIMENS (Part 1 of 5) Clinical Trials: The National Comprehensive Cancer Network recommends cancer patient participation in clinical trials as the gold standard for treatment. Cancer therapy selection, dosing, administration, and the management of related adverse events can be a complex process that should be handled by an experienced health care team. Clinicians must choose and verify treatment options based on the individual patient; drug dose modifications and supportive care interventions should be administered accordingly. The cancer treatment regimens below may include both U.S. Food and Drug Administration-approved and unapproved indications/regimens. These regimens are provided only to supplement the latest treatment strategies. These Guidelines are a work in progress that may be refined as often as new significant data become available. The NCCN Guidelines® are a consensus statement of its authors regarding their views of currently accepted approaches to treatment. Any clinician seeking to apply or consult any NCCN Guidelines® is expected to use independent medical judgment in the context of individual clinical circumstances to determine any patient’s care or treatment. The NCCN makes no warranties of any kind whatsoever regarding their content, use, or application and disclaims any responsibility for their application or use in any way. Classical Hodgkin Lymphoma1 Note: All recommendations are Category 2A unless otherwise indicated. Primary Treatment Stage IA, IIA Favorable (No Bulky Disease, <3 Sites of Disease, ESR <50, and No E-lesions) REGIMEN DOSING Doxorubicin + Bleomycin + Days 1 and 15: Doxorubicin 25mg/m2 IV push + bleomycin 10units/m2 IV push + Vinblastine + Dacarbazine vinblastine 6mg/m2 IV over 5–10 minutes + dacarbazine 375mg/m2 IV over (ABVD) (Category 1)2-5 60 minutes. -
![Bendamustine and Cytosine Arabinoside: a Highly Synergistic Combination Visco C*, Carli G and Rodeghiero F Further DNA Synthesis [24]](https://docslib.b-cdn.net/cover/9877/bendamustine-and-cytosine-arabinoside-a-highly-synergistic-combination-visco-c-carli-g-and-rodeghiero-f-further-dna-synthesis-24-599877.webp)
Bendamustine and Cytosine Arabinoside: a Highly Synergistic Combination Visco C*, Carli G and Rodeghiero F Further DNA Synthesis [24]
Open Access Austin Journal of Cancer and Clinical Research Editorial Bendamustine and Cytosine Arabinoside: A Highly Synergistic Combination Visco C*, Carli G and Rodeghiero F further DNA synthesis [24]. The synergistic effect of bendamustine Department of Cell Therapy and Hematology, San Bortolo and cytarabine could be related to the individual mechanism of Hospital, Italy action of the two drugs, whose serial administration would avoid *Corresponding author: Carlo Visco, Department of the saturation of the common pathways. Cells escaping the cell cycle Cell Therapy and Hematology, San Bortolo Hospital, Via arrest induced by bendamustine and trying to repair their damage Rodolfi 37, 36100 Vicenza, Italy, Tel: +39 0444 753626; would be prone to incorporate the metabolite ara-CTP into DNA, Fax: +39 0444 920708; Email: [email protected] as reported by Staib [19] in acute myeloid leukemia cells. Indeed, the Received: January 07, 2015; Accepted: March 16, sequential treatment with bendamustine followed by cytarabine was 2015; Published: April 03, 2015 proven to be more effective than simultaneous addition of the two drugs (Figure 2) [21-23]. The S phase of the cell cycle is a crucial step Editorial of replication in mantle cell lymphoma (MCL) cells, where cyclin D1 Bendamustine is a bifunctional compound that has shown clinical overexpression deregulates the cell cycle at the G1/S phase transition, activity against various human cancers including non-Hodgkin’s and and is likely the engine continuously pushing cells towards S-phase Hodgkin’s lymphoma [1,2], chronic lymphocytic leukemia (CLL) [3], (Figure 3). Indeed, both drugs are known to be particularly active in multiple myeloma [4,5], breast cancer [6], and small-cell lung cancer patients with MCL. -

Bendamustine: Treanda®; Bendeka®; Belrapzo™
Bendamustine: Treanda®; Bendeka®; Belrapzo™ RTD (Intravenous) Document Number: IH-0130 Last Review Date: 03/01/2021 Date of Origin: 01/01/12 Dates Reviewed: 12/2011, 02/2013, 09/2013, 12/2013, 03/2014, 06/2014, 09/2014, 12/2014, 03/2015, 05/2015, 11/2015, 02/2016, 05/2016, 08/2016, 11/2016, 02/2017, 05/2017, 08/2017, 11/2017, 02/2018, 05/2018, 09/2018, 12/2018, 03/2019, 06/2019, 09/2019, 12/2019, 03/2020, 06/2020, 09/2020, 03/2021 I. Length of Authorization 1-3,5,8,13 • Non-Hodgkin’s Lymphoma (NHL), Chronic Lymphocytic Leukemia (CLL)/Small Lymphocytic Lymphoma (SLL), Waldenström’s Macroglobulinemia (WM)/Lymphoplasmacytic Lymphoma (LPL), Classic Hodgkin Lymphoma (cHL): o Coverage will be provided for six months and may NOT be renewed. • Multiple Myeloma: o Coverage will be provided for eight months and may NOT be renewed. II. Dosing Limits A. Quantity Limit (max daily dose) [NDC Unit]: Treanda 100 mg lyophilized powder for injection: 6 vials every 21 days Treanda 25 mg lyophilized powder for injection: 3 vials every 21 days Bendeka 100 mg/4 mL multi-dose vial: 6 vials every 21 days Belrapzo 100 mg/4 mL RTD multi-dose vial: 6 vials every 21 days B. Max Units (per dose and over time) [HCPCS Unit]: NHL: • 600 billable units every 21 days WM/LPL: • 450 billable units every 28 days cccHL:cHL: • 600 billable units every 28 days CLL/SLL & MMMultipleMultiple MMMyelomaMyelomayeloma:::: • 500 billable units every 28 days Proprietary & Confidential © 2021 Magellan Health, Inc. III. Initial Approval Criteria 1 Coverage is provided in the following -

Real Life Use of Bendamustine in Elderly Patients with Lymphoid Neoplasia
Journal of Personalized Medicine Article Real Life Use of Bendamustine in Elderly Patients with Lymphoid Neoplasia Irene Dogliotti 1, Simone Ragaini 2 , Francesco Vassallo 3, Elia Boccellato 2, Gabriele De Luca 2, Francesca Perutelli 2 , Carola Boccomini 3, Michele Clerico 2, Barbara Botto 3, Daniele Grimaldi 2, Lorella Orsucci 3, Simone Ferrero 2 , Candida Vitale 2 , Dario Ferrero 2, Marta Coscia 2,† and Federica Cavallo 2,*,† 1 Stem Cell Transplant Unit, A.O.U., Città della Salute e della Scienza di Torino, 10126 Turin, Italy; [email protected] 2 Division of Hematology, Department of Molecular Biotechnology and Health Sciences, University of Torino, A.O.U., Città della Salute e della Scienza di Torino, 10126 Turin, Italy; [email protected] (S.R.); [email protected] (E.B.); [email protected] (G.D.L.); [email protected] (F.P.); [email protected] (M.C.); [email protected] (D.G.); [email protected] (S.F.); [email protected] (C.V.); [email protected] (D.F.); [email protected] (M.C.) 3 Division of Hematology, A.O.U., Città della Salute e della Scienza di Torino, 10126 Turin, Italy; [email protected] (F.V.); [email protected] (C.B.); [email protected] (B.B.); [email protected] (L.O.) * Correspondence: [email protected]; Tel.: +39-01-1633-4556; Fax: +39-01-1633-6507 † These authors equally contributed to the manuscript. Abstract: Background. Bendamustine is a cytotoxic alkylating drug with a broad range of indications as a single agent or in combination therapy in lymphoid neoplasia patients. -

Standard Oncology Criteria C16154-A
Prior Authorization Criteria Standard Oncology Criteria Policy Number: C16154-A CRITERIA EFFECTIVE DATES: ORIGINAL EFFECTIVE DATE LAST REVIEWED DATE NEXT REVIEW DATE DUE BEFORE 03/2016 12/2/2020 1/26/2022 HCPCS CODING TYPE OF CRITERIA LAST P&T APPROVAL/VERSION N/A RxPA Q1 2021 20210127C16154-A PRODUCTS AFFECTED: See dosage forms DRUG CLASS: Antineoplastic ROUTE OF ADMINISTRATION: Variable per drug PLACE OF SERVICE: Retail Pharmacy, Specialty Pharmacy, Buy and Bill- please refer to specialty pharmacy list by drug AVAILABLE DOSAGE FORMS: Abraxane (paclitaxel protein-bound) Cabometyx (cabozantinib) Erwinaze (asparaginase) Actimmune (interferon gamma-1b) Calquence (acalbrutinib) Erwinia (chrysantemi) Adriamycin (doxorubicin) Campath (alemtuzumab) Ethyol (amifostine) Adrucil (fluorouracil) Camptosar (irinotecan) Etopophos (etoposide phosphate) Afinitor (everolimus) Caprelsa (vandetanib) Evomela (melphalan) Alecensa (alectinib) Casodex (bicalutamide) Fareston (toremifene) Alimta (pemetrexed disodium) Cerubidine (danorubicin) Farydak (panbinostat) Aliqopa (copanlisib) Clolar (clofarabine) Faslodex (fulvestrant) Alkeran (melphalan) Cometriq (cabozantinib) Femara (letrozole) Alunbrig (brigatinib) Copiktra (duvelisib) Firmagon (degarelix) Arimidex (anastrozole) Cosmegen (dactinomycin) Floxuridine Aromasin (exemestane) Cotellic (cobimetinib) Fludara (fludarbine) Arranon (nelarabine) Cyramza (ramucirumab) Folotyn (pralatrexate) Arzerra (ofatumumab) Cytosar-U (cytarabine) Fusilev (levoleucovorin) Asparlas (calaspargase pegol-mknl Cytoxan (cyclophosphamide) -

Immunomodulatory Effects of Bendamustine in Hematopoietic Cell Transplantation
cancers Review Immunomodulatory Effects of Bendamustine in Hematopoietic Cell Transplantation Jessica Stokes 1, Megan S. Molina 1,2 , Emely A. Hoffman 1, Richard J. Simpson 1,2,3,4 and Emmanuel Katsanis 1,2,4,5,6,* 1 Department of Pediatrics, University of Arizona, Tucson, AZ 85721, USA; [email protected] (J.S.); [email protected] (M.S.M.); [email protected] (E.A.H.); [email protected] (R.J.S.) 2 Department of Immunobiology, University of Arizona, Tucson, AZ 85721, USA 3 Department of Nutritional Sciences, University of Arizona, Tucson, AZ 85721, USA 4 The University of Arizona Cancer Center, Tucson, AZ 85721, USA 5 Department of Medicine, University of Arizona, Tucson, AZ 85721, USA 6 Department of Pathology, University of Arizona, Tucson, AZ 85721, USA * Correspondence: [email protected]; Tel.: +1-520-626-7053 Simple Summary: Bendamustine is a chemotherapeutic agent used to treat a variety of cancers. It has recently been used in the context of allogeneic hematopoietic cell transplantation (HCT), a treatment mostly used to treat blood cancers. Given before or after transplantation of donor blood or bone marrow cells, bendamustine has been shown to reduce the side effects of the transplant, including graft-versus-host disease, where the donated cells attack the recipient’s tissues, while also promoting the anti-cancer effects of the transplant. These are exciting findings and show that bendamustine may be used to influence the immune system, called immunomodulation, in a beneficial manner. We report our research and review the available literature outlining these immunomodulatory effects of bendamustine, in hopes that it will promote further investigations utilizing this agent in allogeneic Citation: Stokes, J.; Molina, M.S.; transplants, ultimately improving patient outcomes. -
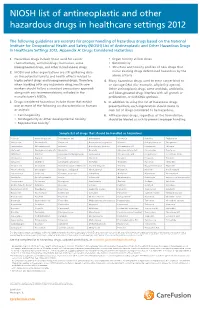
NIOSH List of Antineoplastic and Other Hazardous Drugs in Healthcare Settings 2012
NIOSH list of antineoplastic and other hazardous drugs in healthcare settings 2012 The following guidelines are excerpts for proper handling of hazardous drugs based on the National Institute for Occupational Health and Safety (NIOSH) List of Antineoplastic and Other Hazardous Drugs in Healthcare Settings 2012, Appendix A: Drugs Considered Hazardous 1. Hazardous drugs include those used for cancer • Organ toxicity at low doses* chemotherapy, antiviral drugs, hormones, some • Genotoxicity** bioengineered drugs, and other miscellaneous drugs. • Structure and toxicity profiles of new drugs that 2. NIOSH and other organizations are still gathering data mimic existing drugs determined hazardous by the on the potential toxicity and health effects related to above criteria highly potent drugs and bioengineered drugs. Therefore, 4. Many hazardous drugs used to treat cancer bind to when working with any hazardous drug, health care or damage DNA (for example, alkylating agents). workers should follow a standard precautions approach Other antineoplastic drugs, some antivirals, antibiotics, along with any recommendations included in the and bioengineered drugs interfere with cell growth or manufacturer’s MSDSs. proliferation, or with DNA synthesis. 3. Drugs considered hazardous include those that exhibit 5. In addition to using the list of hazardous drugs one or more of the following six characteristics in humans presented here, each organization should create its or animals: own list of drugs considered to be hazardous. • Carcinogenicity 6. All hazardous -

Bendamustine
Bendamustine DRUG NAME: Bendamustine SYNONYM(S): COMMON TRADE NAME(S): TREANDA® CLASSIFICATION: Alkylating agent Special pediatric considerations are noted when applicable, otherwise adult provisions apply. MECHANISM OF ACTION: Bendamustine is a bifunctional mechlorethamine derivative containing a purine-like benzimidazole ring. Its exact mechanism of action is unknown, but it may cause apoptotic and non-apoptotic death of malignant cells by damaging both single- and double-strand DNA, increasing the expression of pro-apoptotic genes, and inhibiting mitotic control. Bendamustine is active against both quiescent and dividing cells.1 PHARMACOKINETICS: Distribution not extensively distributed in tissues cross blood brain barrier? no information found volume of distribution 20-40 L plasma protein binding 94-96% (in vitro); however not considered likely to displace or be displaced by highly protein-bound drugs Metabolism primarily via hydrolysis; also via oxidative and conjugative pathways active metabolite(s) gamma-hydroxy bendamustine and N-desmethyl- bendamustine (via CYP 1A2); cytotoxic activity primarily from parent compound2 inactive metabolite(s) monohydroxy and dihydroxy-bendamustine (via hydrolysis) Excretion rapid elimination of bendamustine and active metabolites urine 3% (as parent compound); <1% (as active metabolites); <5% (as inactive metabolites) feces 90%3 terminal half life 40 min (parent compound); 3.7 h (active metabolites) clearance 32-42 L/h Sex no significant differences Elderly no significant differences Ethnicity limited data; effect of race has not been established Adapted from standard reference4,5 unless specified otherwise. USES: Primary uses: Other uses: *Lymphoma, non-Hodgkin’s Lymphoma, Hodgkin’s6 *Leukemia, chronic lymphocytic *Health Canada approved indication BC Cancer Drug Manual© Page 1 of 8 Bendamustine Developed: 1 May 2013 Revised: 1 October 2018 Bendamustine SPECIAL PRECAUTIONS: Contraindications: history of hypersensitivity reaction to bendamustine or mechlorethamine. -

Trastuzumab-Associated Flagellate Erythema: Report in a Woman with Metastatic Breast Cancer and Review of Antineoplastic Therapy-Induced Flagellate Dermatoses
Dermatol Ther (Heidelb) DOI 10.1007/s13555-015-0085-2 CASE REPORT Trastuzumab-Associated Flagellate Erythema: Report in a Woman with Metastatic Breast Cancer and Review of Antineoplastic Therapy-Induced Flagellate Dermatoses Philip R. Cohen To view enhanced content go to www.dermtherapy-open.com Received: September 13, 2015 Ó The Author(s) 2015. This article is published with open access at Springerlink.com ABSTRACT relevant manuscripts, along with their reference citations, were evaluated. Introduction: Flagellate erythema presents as Results: The woman’s pruritus and skin lesions erythematous, individual and intermingled, promptly resolved after treatment with linear streaks in a whiplash-like pattern. corticosteroids (oral and topical) and Several conditions, including antineoplastic antihistamines (oral); premedication with agents, have been associated with flagellate dexamethasone prior to each subsequent erythema. A woman with metastatic breast trastuzumab treatment prevented recurrence of cancer who developed flagellate erythema after flagellate erythema. Chemotherapy-induced receiving trastuzumab is described and the flagellate erythema was initially described in features of flagellate erythema associated with oncology patients who received bleomycin. In other antineoplastic agents are reviewed. addition to trastuzumab, other antineoplastic Methods: PubMed was used to search the agents that have been associated with the following terms, separately and in development of flagellate erythema include combination: agent, antineoplastic, bendamustine, -

For Health Professionals Who Care for Cancer Patients
October 2018 ♦ Vol. 21 ♦ No. 10 For Health Professionals Who Care For Cancer Patients Inside This Issue: . Editor’s Choice – New Programs: Osimertinib for Advanced . List of New and Revised Protocols, Provincial Pre-Printed EGFR T790M Mutation-Positive NSCLC Following Progression Orders and Patient Handouts – New: ULUAVOSI; Revised: with EGFR TKI ( ULUAVOSI); Revised Programs: Expanded BRAJDCARBT, BRAJZOL2, BRAJZOL5, UBRAVPALAI, BRAVTEST, Eligibility Criteria for Palbociclib with Aromatase Inhibitor in BRAVTRVIN, CNBEV, UGILAN, UGOOVOLAPM, GUBEP, Advanced Breast Cancer (UBRAVPALAI) ULYMIBRU, MOIT, UMYCARDEX, UMYCARLD, UMYLDF, . Drug Update – Drug Shortages Resolved: Docetaxel, UMYLDREL, UMYLENMTN, MYPAM, UMYPOMDEX, MYZOL, Vinorelbine SAAJGI, SCPAINLI; Deleted: GUBCV, GUEMCYT, GUKIFN, PUM . Benefit Drug List – New: ULUAVOSI; Deleted: GUBCV, . Website Resources and Contact Information GUEMCYT, GUKIFN, PUM . Cancer Drug Manual – New: Venetoclax; Revised: Bendamustine, Osimertinib EDITOR’S CHOICE NEW PROGRAMS Effective 01 October 2018, the BC Cancer Provincial Systemic Therapy Program has approved the following treatment program: Lung: Osimertinib for Epidermal Growth Factor Receptor (EGFR) T790M Mutation-Positive Advanced Non- Small Cell Lung Cancer (ULUAVOSI) – The BC Cancer Lung Tumour Group has implemented this new treatment program for patients who acquired the T790M mutation following progression on prior EGFR tyrosine kinase inhibitor (TKI) therapy (e.g. afatinib, geftinib, erlotinib), and for those with de novo T790M mutation-positive disease. In a phase III randomized controlled trial (AURA 3), osimertinib demonstrated superior progression-free survival (PFS) compared to platinum-doublet chemotherapy (median PFS 10.1 mo vs. 4.4 mo, HR 0.30 95% CI 0.23-0.41) and improved tolerability.1 The most commonly reported adverse events associated with osimertinib were diarrhea (41%), rash (34%), dry skin (23%) and paronychia (22%). -

R-BAC Relapsed/Refractory Non-Hodgkin's Lymphoma
THE CLATTERBRIDGE CANCER CENTRE NHS FOUNDATION TRUST Systemic Anti-Cancer Therapy Protocol R-BAC Relapsed/Refractory Non- Hodgkin’s Lymphoma PROTOCOL REF: MPHARBACHA (Version No: 1.0) Approved for use in: Relapsed or refractory Mantle Cell Lymphoma First line treatment of Mantle Cell Lymphoma in less fit patients unsuitable for more intensive treatment Blueteq registration required (bendamustine) Dosage: Drug Dose Route Frequency Rituximab 375mg/m2 IV infusion Day 1 only Bendamustine 70mg/m2 IV infusion Days 2 and 3 (once daily) Cytarabine 500mg/m2 ** IV infusion Days 2 to 4 (once daily) **Dose can be increased to 800mg/m2 if patient tolerates first cycle well – clinical decision. Maximum of 6 cycles (cycle length 28 days) Administration: Due to the long retention time of rituximab in B cell depleted patients, women of childbearing potential must employ effective contraceptive methods during and for 12 months after treatment with rituximab. Patients will required irradiated blood products (lifelong) –the patients receive information booklets about irradiated blood when counselled by the specialist nurses. It Issue Date: 11th December 2020 Page 1 of 6 Protocol reference: MPHARBACHA Review Date: December 2023 Author: Aileen McCaughey Authorised by: DTC Version No: 1.0 THE CLATTERBRIDGE CANCER CENTRE NHS FOUNDATION TRUST contains an alert card that the patient carries around with them. The specialist nurses then contact the lab. Anti-emetic risk: Severely emetogenic. Supportive treatments: Rituximab pre-infusion medicines: Paracetamol -

Cancer Drug Costs for a Month of Treatment at Initial Food
Cancer drug costs for a month of treatment at initial Food and Drug Administration approval Year of FDA Monthly Cost Monthly cost (2013 Generic name Brand name(s) approval (actual $'s) $'s) Vinblastine Velban 1965 $78 $575 Thioguanine, 6-TG Thioguanine Tabloid 1966 $17 $122 Hydroxyurea Hydrea 1967 $14 $97 Cytarabine Cytosar-U, Tarabine PFS 1969 $13 $82 Procarbazine Matulane 1969 $2 $13 Testolactone Teslac 1969 $179 $1,136 Mitotane Lysodren 1970 $134 $801 Plicamycin Mithracin 1970 $50 $299 Mitomycin C Mutamycin 1974 $5 $22 Dacarbazine DTIC-Dome 1975 $29 $125 Lomustine CeeNU 1976 $10 $41 Carmustine BiCNU, BCNU 1977 $33 $127 Tamoxifen citrate Nolvadex 1977 $44 $167 Cisplatin Platinol 1978 $125 $445 Estramustine Emcyt 1981 $420 $1,074 Streptozocin Zanosar 1982 $61 $147 Etoposide, VP-16 Vepesid 1983 $181 $422 Interferon alfa 2a Roferon A 1986 $742 $1,573 Daunorubicin, Daunomycin Cerubidine 1987 $533 $1,090 Doxorubicin Adriamycin 1987 $521 $1,066 Mitoxantrone Novantrone 1987 $477 $976 Ifosfamide IFEX 1988 $1,667 $3,274 Flutamide Eulexin 1989 $213 $399 Altretamine Hexalen 1990 $341 $606 Idarubicin Idamycin 1990 $227 $404 Levamisole Ergamisol 1990 $105 $187 Carboplatin Paraplatin 1991 $860 $1,467 Fludarabine phosphate Fludara 1991 $662 $1,129 Pamidronate Aredia 1991 $507 $865 Pentostatin Nipent 1991 $1,767 $3,015 Aldesleukin Proleukin 1992 $13,503 $22,364 Melphalan Alkeran 1992 $35 $58 Cladribine Leustatin, 2-CdA 1993 $764 $1,229 Asparaginase Elspar 1994 $694 $1,088 Paclitaxel Taxol 1994 $2,614 $4,099 Pegaspargase Oncaspar 1994 $3,006 $4,713