Doxapram Elisa Kit Instructions Product #106219 & 106216 Forensic Use Only
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Properties and Units in Clinical Pharmacology and Toxicology
Pure Appl. Chem., Vol. 72, No. 3, pp. 479–552, 2000. © 2000 IUPAC INTERNATIONAL FEDERATION OF CLINICAL CHEMISTRY AND LABORATORY MEDICINE SCIENTIFIC DIVISION COMMITTEE ON NOMENCLATURE, PROPERTIES, AND UNITS (C-NPU)# and INTERNATIONAL UNION OF PURE AND APPLIED CHEMISTRY CHEMISTRY AND HUMAN HEALTH DIVISION CLINICAL CHEMISTRY SECTION COMMISSION ON NOMENCLATURE, PROPERTIES, AND UNITS (C-NPU)§ PROPERTIES AND UNITS IN THE CLINICAL LABORATORY SCIENCES PART XII. PROPERTIES AND UNITS IN CLINICAL PHARMACOLOGY AND TOXICOLOGY (Technical Report) (IFCC–IUPAC 1999) Prepared for publication by HENRIK OLESEN1, DAVID COWAN2, RAFAEL DE LA TORRE3 , IVAN BRUUNSHUUS1, MORTEN ROHDE1, and DESMOND KENNY4 1Office of Laboratory Informatics, Copenhagen University Hospital (Rigshospitalet), Copenhagen, Denmark; 2Drug Control Centre, London University, King’s College, London, UK; 3IMIM, Dr. Aiguader 80, Barcelona, Spain; 4Dept. of Clinical Biochemistry, Our Lady’s Hospital for Sick Children, Crumlin, Dublin 12, Ireland #§The combined Memberships of the Committee and the Commission (C-NPU) during the preparation of this report (1994–1996) were as follows: Chairman: H. Olesen (Denmark, 1989–1995); D. Kenny (Ireland, 1996); Members: X. Fuentes-Arderiu (Spain, 1991–1997); J. G. Hill (Canada, 1987–1997); D. Kenny (Ireland, 1994–1997); H. Olesen (Denmark, 1985–1995); P. L. Storring (UK, 1989–1995); P. Soares de Araujo (Brazil, 1994–1997); R. Dybkær (Denmark, 1996–1997); C. McDonald (USA, 1996–1997). Please forward comments to: H. Olesen, Office of Laboratory Informatics 76-6-1, Copenhagen University Hospital (Rigshospitalet), 9 Blegdamsvej, DK-2100 Copenhagen, Denmark. E-mail: [email protected] Republication or reproduction of this report or its storage and/or dissemination by electronic means is permitted without the need for formal IUPAC permission on condition that an acknowledgment, with full reference to the source, along with use of the copyright symbol ©, the name IUPAC, and the year of publication, are prominently visible. -
![Doxapram Shortens Recovery Following Sevoflurane Anesthesia [Le Doxapram Hâte La Récupération Après Une Anesthésie Au Sévoflurane]](https://docslib.b-cdn.net/cover/1653/doxapram-shortens-recovery-following-sevoflurane-anesthesia-le-doxapram-h%C3%A2te-la-r%C3%A9cup%C3%A9ration-apr%C3%A8s-une-anesth%C3%A9sie-au-s%C3%A9voflurane-391653.webp)
Doxapram Shortens Recovery Following Sevoflurane Anesthesia [Le Doxapram Hâte La Récupération Après Une Anesthésie Au Sévoflurane]
456 CANADIAN JOURNALGENERAL OF ANESTHESIA ANESTHESIA Doxapram shortens recovery following sevoflurane anesthesia [Le doxapram hâte la récupération après une anesthésie au sévoflurane] Chi-Chen Wu MD,* Martin S. Mok MD,* Jui-Yuan Chen MD,† Gong-Jhe Wu MD,† Yeong-Ray Wen MD,† Chao-Shun Lin MD* Purpose: A randomized, double blind controlled trial was possibilité de serrer la main sur demande, le temps d’extubation et undertaken to investigate the effect of doxapram on recovery le score de récupération d’Aldrete. Les valeurs de l’index bispectral, times and bispectral index following sevoflurane anesthesia. la tension artérielle systolique et la fréquence cardiaque ont été Methods: Upon completion of surgery under sevoflurane anes- enregistrées avant l’anesthésie, pendant l’opération et à chaque thesia, 60 adult patients were randomly allocated to receive minute pendant 15 min après l’administration du médicament. either doxapram hydrochloride 1 mg·kg–1 iv or saline placebo. Résultats : Le temps écoulé avant l’ouverture des yeux a été plus Clinical recovery from anesthesia was assessed by time to eye court avec le doxapram qu’avec le placebo (6,9 ± 2,2 min vs 9,9 opening on verbal command, hand squeezing on command, ± 3,1 min, P < 0,05). Les scores moyens de l’index bispectral ont time to extubation, and the Aldrete recovery score. Bispectral été aussi plus élevés avec le doxapram sept à huit minutes après index values, systolic blood pressure, and heart rate were l’administration du médicament expérimental (P < 0,05). Un recorded at baseline (before anesthesia), during surgery, and retour à la conscience plus rapide a été associé à une plus grande every minute for 15 min after administration of the study drug. -

Title 16. Crimes and Offenses Chapter 13. Controlled Substances Article 1
TITLE 16. CRIMES AND OFFENSES CHAPTER 13. CONTROLLED SUBSTANCES ARTICLE 1. GENERAL PROVISIONS § 16-13-1. Drug related objects (a) As used in this Code section, the term: (1) "Controlled substance" shall have the same meaning as defined in Article 2 of this chapter, relating to controlled substances. For the purposes of this Code section, the term "controlled substance" shall include marijuana as defined by paragraph (16) of Code Section 16-13-21. (2) "Dangerous drug" shall have the same meaning as defined in Article 3 of this chapter, relating to dangerous drugs. (3) "Drug related object" means any machine, instrument, tool, equipment, contrivance, or device which an average person would reasonably conclude is intended to be used for one or more of the following purposes: (A) To introduce into the human body any dangerous drug or controlled substance under circumstances in violation of the laws of this state; (B) To enhance the effect on the human body of any dangerous drug or controlled substance under circumstances in violation of the laws of this state; (C) To conceal any quantity of any dangerous drug or controlled substance under circumstances in violation of the laws of this state; or (D) To test the strength, effectiveness, or purity of any dangerous drug or controlled substance under circumstances in violation of the laws of this state. (4) "Knowingly" means having general knowledge that a machine, instrument, tool, item of equipment, contrivance, or device is a drug related object or having reasonable grounds to believe that any such object is or may, to an average person, appear to be a drug related object. -

Examination Questions
EXAMINATION QUESTIONS CHAPTER I. GENERAL PHARMACOLOGY AND PRESCRIPTION 1. Essence of pharmacology as a science. Parts and fields of modern pharmacology. The main terms and concepts of pharmacology – pharmacological activity, action, efficiency. 2. Sources and stages of drug development. Drugs – generics, placebo effects. Definition of such concepts as medicinal agent (medicinal drug, drug), medicinal substance, medicinal form. 3. Routes of drug administration into the body and their characteristic. Presystemic drug elimination. 4. Drug transfer through biological barriers and their types. The main factors influencing on the drug transfer in the body. 5. Drug transfer of variable ionization substances through membranes (Henderson-Hasselbach's equation of ionization). Principles of transfer management. 6. Drug transfer in the body. Aqueous diffusion and lipid diffusion (Fick's diffusion equation). Active transport. 7. Central postulate of pharmacokinetics: concentration of medicinal substance in blood plasma – the main parameter for management of the pharmacological effect. The tasks solved on the basis of this postulate. 8. Pharmacokinetic models (one-compartment and two-compartment), quantitative laws of absorption and drug elimination. 9. Bioavailability of drugs – definition, essence, quantitative expression, determinants. 10. Drug distribution in the body: compartments, ligands, the main determinants of distribution. 11. Elimination rate constant, its essence, dimension, connection with other pharmacokinetic parameters. 12. Excretion half-life of drugs, its essence, dimension, connection with other pharmacokinetic parameters. 13. Clearance as the main parameter of pharmacokinetics for management of the dosing regimen. Its essence, dimension and connection with other pharmacokinetic parameters. 14. Dose. Types of doses. Units of drug dosage. Aims of drug dosage, ways and variants of administration of drugs, dosing interval. -

Prohibited Substances List
Prohibited Substances List This is the Equine Prohibited Substances List that was voted in at the FEI General Assembly in November 2009 alongside the new Equine Anti-Doping and Controlled Medication Regulations(EADCMR). Neither the List nor the EADCM Regulations are in current usage. Both come into effect on 1 January 2010. The current list of FEI prohibited substances remains in effect until 31 December 2009 and can be found at Annex II Vet Regs (11th edition) Changes in this List : Shaded row means that either removed or allowed at certain limits only SUBSTANCE ACTIVITY Banned Substances 1 Acebutolol Beta blocker 2 Acefylline Bronchodilator 3 Acemetacin NSAID 4 Acenocoumarol Anticoagulant 5 Acetanilid Analgesic/anti-pyretic 6 Acetohexamide Pancreatic stimulant 7 Acetominophen (Paracetamol) Analgesic/anti-pyretic 8 Acetophenazine Antipsychotic 9 Acetylmorphine Narcotic 10 Adinazolam Anxiolytic 11 Adiphenine Anti-spasmodic 12 Adrafinil Stimulant 13 Adrenaline Stimulant 14 Adrenochrome Haemostatic 15 Alclofenac NSAID 16 Alcuronium Muscle relaxant 17 Aldosterone Hormone 18 Alfentanil Narcotic 19 Allopurinol Xanthine oxidase inhibitor (anti-hyperuricaemia) 20 Almotriptan 5 HT agonist (anti-migraine) 21 Alphadolone acetate Neurosteriod 22 Alphaprodine Opiod analgesic 23 Alpidem Anxiolytic 24 Alprazolam Anxiolytic 25 Alprenolol Beta blocker 26 Althesin IV anaesthetic 27 Althiazide Diuretic 28 Altrenogest (in males and gelidngs) Oestrus suppression 29 Alverine Antispasmodic 30 Amantadine Dopaminergic 31 Ambenonium Cholinesterase inhibition 32 Ambucetamide Antispasmodic 33 Amethocaine Local anaesthetic 34 Amfepramone Stimulant 35 Amfetaminil Stimulant 36 Amidephrine Vasoconstrictor 37 Amiloride Diuretic 1 Prohibited Substances List This is the Equine Prohibited Substances List that was voted in at the FEI General Assembly in November 2009 alongside the new Equine Anti-Doping and Controlled Medication Regulations(EADCMR). -

“Inactive” Ingredients in Pharmaceutical Products: Update (Subject Review)
AMERICAN ACADEMY OF PEDIATRICS Committee on Drugs “Inactive” Ingredients in Pharmaceutical Products: Update (Subject Review) ABSTRACT. Because of an increasing number of re- bronchospasm from antiasthmatic drugs, aspartame- ports of adverse reactions associated with pharmaceutical induced headache and seizures, saccharin-induced excipients, in 1985 the Committee on Drugs issued a cross-sensitivity reactions in children with sulfon- position statement1 recommending that the Food and amide allergy, benzyl alcohol toxicity in neonates Drug Administration mandate labeling of over-the- receiving high-dose continuous infusion with pre- counter and prescription formulations to include a qual- served medications, dye-related cross-reactions in itative list of inactive ingredients. However, labeling of inactive ingredients remains voluntary. Adverse reac- children with aspirin intolerance, lactose-induced di- tions continue to be reported, although some are no arrhea, and propylene glycol-induced hyperosmola- longer considered clinically significant, and other new lity and lactic acidosis. Although many other excipi- reactions have emerged. The original statement, there- ents have been implicated in causing adverse fore, has been updated and its information expanded. reactions, these are the most significant in the pedi- atric population. ABBREVIATIONS. FDA, Food and Drug Administration; MDIs, metered-dose inhalers ANTIASTHMATIC MEDICATIONS It is readily appreciated that some percentage of asthmatic children will develop a “paradoxical” Pharmaceutical products often contain agents that bronchospasm after they inhale their medication. Be- have a variety of purposes, including improvement cause many of these reactions were attributed to of the appearance, bioavailability, stability, and pal- sulfite, which had been highly publicized as a caus- atability of the product. Excipients (substances ative agent, it was often first suspected. -

Neonatal Medications & Nutrition
Neonatal Medications & Nutrition A Comprehensive Guide 3rd Edition KARIN E. ZENK, PHARMD, FASHP Associate Clinical Professor of Pediatrics, University of California, Irvine, and in private consulting practice Formerly, Pharmacist-Specialist in Pediatrics and Neonatology, University of California, Irvine Medical Center JACK H. SILLS, MD Medical Director, Neonatal Intensive Care Unit, University of California, Irvine Medical Center, Clinical Professor of Pediatrics, University of California, Irvine ROBIN M. KOEPPEL, RNC, MS, CPNP, CS Neonatal Clinical Nurse Specialist/Pediatric Nurse Practitioner, University of California, Irvine Medical Center NK BOOK PUBLISHERS SAN T-A ROSA, CALIFORNIA Neonatal Medications & Nutrition TABLE OF CONTENTS Table of Contents by Therapeutic Categories and Indications xv Notice xxvi How to Use This Book xxvii Special Considerations in Neonatal Drug Therapy xxix SECTION I-DRUC MONOGRAPHS Acetaminophen (Tylenol) 1 Acetylcysteine (Mucomyst) 8 Acyclovir (Zovirax) 11 Adenosine (Adenocard) 13 Albumin 15 Albuterol (Proventil, Ventolin) .17 Alprostadil (Prostin VR Pediatric) 22 Alteplase, Recombinant (Activase, Cathflo Activase, Actilyse) 25 Aluminum and Magnesium Hydroxides (Maalox) 33 Amikacin (Amikin) 35 Aminocaproic Acid (Amicar) 37 Aminophylline 40 Amiodarone (Cordarone) 45 Amoxicillin (Amoxil) 53 Amphotericin B, Conventional (Fungizone) and Amphotericin B Lipid Based (Abelcet, Amphotec, AmBisome) 55 Ampicillin 60 Aquaphor Ointment 63 Arginine Hydrochloride (R-Gene 10) 66 Atraeurium (Tracrium) 75 Atropine 78 Aztreonam -
![Ehealth DSI [Ehdsi V2.2.2-OR] Ehealth DSI – Master Value Set](https://docslib.b-cdn.net/cover/8870/ehealth-dsi-ehdsi-v2-2-2-or-ehealth-dsi-master-value-set-1028870.webp)
Ehealth DSI [Ehdsi V2.2.2-OR] Ehealth DSI – Master Value Set
MTC eHealth DSI [eHDSI v2.2.2-OR] eHealth DSI – Master Value Set Catalogue Responsible : eHDSI Solution Provider PublishDate : Wed Nov 08 16:16:10 CET 2017 © eHealth DSI eHDSI Solution Provider v2.2.2-OR Wed Nov 08 16:16:10 CET 2017 Page 1 of 490 MTC Table of Contents epSOSActiveIngredient 4 epSOSAdministrativeGender 148 epSOSAdverseEventType 149 epSOSAllergenNoDrugs 150 epSOSBloodGroup 155 epSOSBloodPressure 156 epSOSCodeNoMedication 157 epSOSCodeProb 158 epSOSConfidentiality 159 epSOSCountry 160 epSOSDisplayLabel 167 epSOSDocumentCode 170 epSOSDoseForm 171 epSOSHealthcareProfessionalRoles 184 epSOSIllnessesandDisorders 186 epSOSLanguage 448 epSOSMedicalDevices 458 epSOSNullFavor 461 epSOSPackage 462 © eHealth DSI eHDSI Solution Provider v2.2.2-OR Wed Nov 08 16:16:10 CET 2017 Page 2 of 490 MTC epSOSPersonalRelationship 464 epSOSPregnancyInformation 466 epSOSProcedures 467 epSOSReactionAllergy 470 epSOSResolutionOutcome 472 epSOSRoleClass 473 epSOSRouteofAdministration 474 epSOSSections 477 epSOSSeverity 478 epSOSSocialHistory 479 epSOSStatusCode 480 epSOSSubstitutionCode 481 epSOSTelecomAddress 482 epSOSTimingEvent 483 epSOSUnits 484 epSOSUnknownInformation 487 epSOSVaccine 488 © eHealth DSI eHDSI Solution Provider v2.2.2-OR Wed Nov 08 16:16:10 CET 2017 Page 3 of 490 MTC epSOSActiveIngredient epSOSActiveIngredient Value Set ID 1.3.6.1.4.1.12559.11.10.1.3.1.42.24 TRANSLATIONS Code System ID Code System Version Concept Code Description (FSN) 2.16.840.1.113883.6.73 2017-01 A ALIMENTARY TRACT AND METABOLISM 2.16.840.1.113883.6.73 2017-01 -

Jp Xvii the Japanese Pharmacopoeia
JP XVII THE JAPANESE PHARMACOPOEIA SEVENTEENTH EDITION Official from April 1, 2016 English Version THE MINISTRY OF HEALTH, LABOUR AND WELFARE Notice: This English Version of the Japanese Pharmacopoeia is published for the convenience of users unfamiliar with the Japanese language. When and if any discrepancy arises between the Japanese original and its English translation, the former is authentic. The Ministry of Health, Labour and Welfare Ministerial Notification No. 64 Pursuant to Paragraph 1, Article 41 of the Law on Securing Quality, Efficacy and Safety of Products including Pharmaceuticals and Medical Devices (Law No. 145, 1960), the Japanese Pharmacopoeia (Ministerial Notification No. 65, 2011), which has been established as follows*, shall be applied on April 1, 2016. However, in the case of drugs which are listed in the Pharmacopoeia (hereinafter referred to as ``previ- ous Pharmacopoeia'') [limited to those listed in the Japanese Pharmacopoeia whose standards are changed in accordance with this notification (hereinafter referred to as ``new Pharmacopoeia'')] and have been approved as of April 1, 2016 as prescribed under Paragraph 1, Article 14 of the same law [including drugs the Minister of Health, Labour and Welfare specifies (the Ministry of Health and Welfare Ministerial Notification No. 104, 1994) as of March 31, 2016 as those exempted from marketing approval pursuant to Paragraph 1, Article 14 of the Same Law (hereinafter referred to as ``drugs exempted from approval'')], the Name and Standards established in the previous Pharmacopoeia (limited to part of the Name and Standards for the drugs concerned) may be accepted to conform to the Name and Standards established in the new Pharmacopoeia before and on September 30, 2017. -
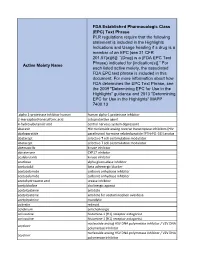
Active Moiety Name FDA Established Pharmacologic Class (EPC) Text
FDA Established Pharmacologic Class (EPC) Text Phrase PLR regulations require that the following statement is included in the Highlights Indications and Usage heading if a drug is a member of an EPC [see 21 CFR 201.57(a)(6)]: “(Drug) is a (FDA EPC Text Phrase) indicated for [indication(s)].” For Active Moiety Name each listed active moiety, the associated FDA EPC text phrase is included in this document. For more information about how FDA determines the EPC Text Phrase, see the 2009 "Determining EPC for Use in the Highlights" guidance and 2013 "Determining EPC for Use in the Highlights" MAPP 7400.13. .alpha. -

Marrakesh Agreement Establishing the World Trade Organization
No. 31874 Multilateral Marrakesh Agreement establishing the World Trade Organ ization (with final act, annexes and protocol). Concluded at Marrakesh on 15 April 1994 Authentic texts: English, French and Spanish. Registered by the Director-General of the World Trade Organization, acting on behalf of the Parties, on 1 June 1995. Multilat ral Accord de Marrakech instituant l©Organisation mondiale du commerce (avec acte final, annexes et protocole). Conclu Marrakech le 15 avril 1994 Textes authentiques : anglais, français et espagnol. Enregistré par le Directeur général de l'Organisation mondiale du com merce, agissant au nom des Parties, le 1er juin 1995. Vol. 1867, 1-31874 4_________United Nations — Treaty Series • Nations Unies — Recueil des Traités 1995 Table of contents Table des matières Indice [Volume 1867] FINAL ACT EMBODYING THE RESULTS OF THE URUGUAY ROUND OF MULTILATERAL TRADE NEGOTIATIONS ACTE FINAL REPRENANT LES RESULTATS DES NEGOCIATIONS COMMERCIALES MULTILATERALES DU CYCLE D©URUGUAY ACTA FINAL EN QUE SE INCORPOR N LOS RESULTADOS DE LA RONDA URUGUAY DE NEGOCIACIONES COMERCIALES MULTILATERALES SIGNATURES - SIGNATURES - FIRMAS MINISTERIAL DECISIONS, DECLARATIONS AND UNDERSTANDING DECISIONS, DECLARATIONS ET MEMORANDUM D©ACCORD MINISTERIELS DECISIONES, DECLARACIONES Y ENTEND MIENTO MINISTERIALES MARRAKESH AGREEMENT ESTABLISHING THE WORLD TRADE ORGANIZATION ACCORD DE MARRAKECH INSTITUANT L©ORGANISATION MONDIALE DU COMMERCE ACUERDO DE MARRAKECH POR EL QUE SE ESTABLECE LA ORGANIZACI N MUND1AL DEL COMERCIO ANNEX 1 ANNEXE 1 ANEXO 1 ANNEX -

Current Awareness in Clinical Toxicology Editors: Damian Ballam Msc and Allister Vale MD
Current Awareness in Clinical Toxicology Editors: Damian Ballam MSc and Allister Vale MD May 2016 CONTENTS General Toxicology 8 Metals 33 Management 16 Pesticides 35 Drugs 19 Chemical Warfare 37 Chemical Incidents & 28 Plants 37 Pollution Chemicals 29 Animals 38 CURRENT AWARENESS PAPERS OF THE MONTH Efficacy and effectiveness of anti-digoxin antibodies in chronic digoxin poisonings from the DORA study (ATOM-1) Chan BS, Isbister GK, O'Leary M, Chiew A, Buckley NA. Clin Toxicol 2016; online early: doi: 10.1080/15563650.2016.1175620: Context We hypothesized that in chronic digoxin toxicity, anti-digoxin antibodies (Fab) would be efficacious in binding digoxin, but this may not translate into improved clinical outcomes. Objective This study aims to investigate changes in free digoxin concentrations and clinical effects on heart rate and potassium concentrations in chronic digoxin poisoning when anti-digoxin Fab are given. Materials and methods This is a prospective observational study. Patients were recruited if they have been treated with anti-digoxin Fab for chronic digoxin poisoning. Data was entered into a standardised prospective form, supplemented with medical records. Their serum or plasma was collected, analysed for free and bound digoxin and free anti-digoxin Fab concentrations. Results From September 2013 to February 2015, 36 patients (median age, 78 years; 22 females) Current Awareness in Clinical Toxicology is produced monthly for the American Academy of Clinical Toxicology by the Birmingham Unit of the UK National Poisons Information Service, with contributions from the Cardiff, Edinburgh, and Newcastle Units. The NPIS is commissioned by Public Health England 2 were recruited from 18 hospitals.