Method 26A Determination of Hydrogen Halide and Halogen
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

An Investigation of the Crystal Growth of Heavy Sulfides in Supercritical
AN ABSTRACT OF THE THESIS OF LEROY CRAWFORD LEWIS for the Ph. D. (Name) (Degree) in CHEMISTRY presented on (Major) (Date) Title: AN INVESTIGATION OF THE CRYSTAL GROWTH OF HEAVY SULFIDES IN SUPERCRITICAL HYDROGEN SULFIDE Abstract approved Redacted for privacy Dr. WilliarriIJ. Fredericks Solubility studies on the heavy metal sulfides in liquid hydrogen sulfide at room temperature were carried out using the isopiestic method. The results were compared with earlier work and with a theoretical result based on Raoult's Law. A relative order for the solubilities of sulfur and the sulfides of tin, lead, mercury, iron, zinc, antimony, arsenic, silver, and cadmium was determined and found to agree with the theoretical result. Hydrogen sulfide is a strong enough oxidizing agent to oxidize stannous sulfide to stannic sulfide in neutral or basic solution (with triethylamine added). In basic solution antimony trisulfide is oxi- dized to antimony pentasulfide. In basic solution cadmium sulfide apparently forms a bisulfide complex in which three moles of bisul- fide ion are bonded to one mole of cadmium sulfide. Measurements were made extending the range over which the volumetric properties of hydrogen sulfide have been investigated to 220 °C and 2000 atm. A virial expression in density was used to represent the data. Good agreement, over the entire range investi- gated, between the virial expressions, earlier work, and the theorem of corresponding states was found. Electrical measurements were made on supercritical hydro- gen sulfide over the density range of 10 -24 moles per liter and at temperatures from the critical temperature to 220 °C. Dielectric constant measurements were represented by a dielectric virial ex- pression. -

Influence of Water on Conductivity of Aluminium Halide Solutions in Acetonitrile
Influence of water on conductivity of aluminium halide solutions in acetonitrile L. LUX Department of Chemistry, Faculty of Metallurgy, Technical University, CS-043 85 Košice Received 7 September 1981 Accepted for publication 8 February 1982 Equilibria of the substitution and association reactions of AICI3 and AlBr3 with acetonitrile were investigated by measuring the conductivity of solutions at varying concentrations of A1C13, AlBr3, and water in the acetonitrile solution. Both halides behave in acetonitrile in the absence of water as relatively strong electrolytes. It has been revealed that water displaces acetoni trile from the solvate sphere of cation of both halides. As for AlBr3, the bromide ion is also displaced from the coordination sphere of aluminium and replaced by a neutral water molecule, which simultaneously results in an increased number of ionized particles in solution. The hydrate A1X3 -6H20 was identified as final product which separates from the solution by adding water. Изучены равновесия реакций замещения и ассоциации А1С13 и А1Вг3 с ацетонитрилом посредством измерения электропроводности раствора в зависимости от концентрации А1С13, А1Вг3, как и в зависимости от концентрации воды в растворе ацетонитрила. Оба галогенида ведут себя в ацетонитриле как сравнительно сильные электролиты в отсутствии воды. Обнаружено, что вода вытесняет ацетонитрил из сольватной обо лочки катиона у обоих галогенидов. У А1Вг3 доходит и к вытеснению иона брома из координационной сферы алюминия и его замещению нейтраль ной молекулой воды, что одновременно приводит к увеличению числа ионизированных частиц в растворе. Конечным продуктом, выделенным из раствора при добавлении воды был идентифицированный гидрат А1Х3-6Н20. Acetonitrile is a typical aprotic solvent which is widely used in electrochemical research because of its profitable properties, i.e. -

Periodic Trends in the Main Group Elements
Chemistry of The Main Group Elements 1. Hydrogen Hydrogen is the most abundant element in the universe, but it accounts for less than 1% (by mass) in the Earth’s crust. It is the third most abundant element in the living system. There are three naturally occurring isotopes of hydrogen: hydrogen (1H) - the most abundant isotope, deuterium (2H), and tritium 3 ( H) which is radioactive. Most of hydrogen occurs as H2O, hydrocarbon, and biological compounds. Hydrogen is a colorless gas with m.p. = -259oC (14 K) and b.p. = -253oC (20 K). Hydrogen is placed in Group 1A (1), together with alkali metals, because of its single electron in the valence shell and its common oxidation state of +1. However, it is physically and chemically different from any of the alkali metals. Hydrogen reacts with reactive metals (such as those of Group 1A and 2A) to for metal hydrides, where hydrogen is the anion with a “-1” charge. Because of this hydrogen may also be placed in Group 7A (17) together with the halogens. Like other nonmetals, hydrogen has a relatively high ionization energy (I.E. = 1311 kJ/mol), and its electronegativity is 2.1 (twice as high as those of alkali metals). Reactions of Hydrogen with Reactive Metals to form Salt like Hydrides Hydrogen reacts with reactive metals to form ionic (salt like) hydrides: 2Li(s) + H2(g) 2LiH(s); Ca(s) + H2(g) CaH2(s); The hydrides are very reactive and act as a strong base. It reacts violently with water to produce hydrogen gas: NaH(s) + H2O(l) NaOH(aq) + H2(g); It is also a strong reducing agent and is used to reduce TiCl4 to titanium metal: TiCl4(l) + 4LiH(s) Ti(s) + 4LiCl(s) + 2H2(g) Reactions of Hydrogen with Nonmetals Hydrogen reacts with nonmetals to form covalent compounds such as HF, HCl, HBr, HI, H2O, H2S, NH3, CH4, and other organic and biological compounds. -

United States Patent Office 2,807,613
United States Patent Office 2,807,613 Patented Sept. 24, 1957 s s 2 2,807,613 nol, where it may exist in the form of a hemiformal, or PREPARATION OF 6-METHYL-6-PHENYLTETRA of a revertible polymer. Usually formaldehyde is used in HYDRO-1,3-OXAZINES excess based on molar proportions referred to the a Claude 5. Schmide, Moorestown, and Richard C. Maas 5 methylstyrene, proportions from about 1.5:1 to 5:1 being field, Haddon afield, N. J., assignors to Rohm & Hiaas practical. Of course, with less than a 2:1 proportion Company, Philadelphia, Pa., a corporatica of Delaware unreacted starting materials may be present in the react ing mixture, Preferred proportions are from 2:1 to 4:1. No Drawing. Application April 3, 1955, Ammonia may be supplied as a gas or as an aqueous Serial No. 577,944 solution or in the form of ammonium chloride or bromide. 7 Claims. (C. 260-244) 0 Of course, if ammonia or ammonium hydroxide is used, This invention deals with a method for improving yields it will react with the hydrochloric or hydrobromic acid of 6-methyl-6-phenyltetrahydro-1,3-oxazines when made which is added as catalyst. The same final result is ob from an O-methylstyrene, formaldehyde, and ammonia. tained by use of the preformed ammonium halide, which In United States Patent 2,647,117, there is described 5 Supplies both the ammonia and the catalyst. The amount the reaction of olefins, including c-methylstyrene, with of ammonia or ammonium compound is usually at least ammonia and formaldehyde in the presence of hydrogen equivalent to the c-methylstyrene and may be in consid -chloride as a catalyst. -

Molecular Dynamics Simulation of Ion Mobility. 2. Alkali Metal and Halide Ions Using the SPC/E Model for Water at 25 °C†
+ + 1420 J. Phys. Chem. 1996, 100, 1420-1425 Molecular Dynamics Simulation of Ion Mobility. 2. Alkali Metal and Halide Ions Using the SPC/E Model for Water at 25 °C† Song Hi Lee Department of Chemistry, Kyungsung UniVersity, Pusan 608-736, Korea Jayendran C. Rasaiah* Department of Chemistry, UniVersity of Maine, Orono, Maine 04469 ReceiVed: October 16, 1995; In Final Form: NoVember 10, 1995X We present results of computer simulations of the mobilities of the alkali metal ions (Li+, Na+,K+, Rb+, and Cs+) and the halides (F-, Cl-, Br-, and I-) at 25 °C using the SPC/E model for water and ion-water parameters fitted to the binding energies of small clusters of ions. A simple truncation of the ion-water and water- water potentials was used, and the mobilities calculated from the mean square displacement and the velocity autocorrelation functions, respectively, were found to be in good agreement with each other. The calculations demonstrate, for the first time, cation and anion mobilities that fall on separate curves, as functions of ion size, with distinct maxima. This is in complete accord with experimental trends observed in water at 25 °C. The cation mobilities are also in better agreement with the measured values than the calculations done earlier (J. Chem. Phys. 1994, 101, 6964) using the TIP4P model. The mobilities of the halides calculated here for the SPC/E model are however slightly lower than the experimental results. The residence times of water in the hydration shells around an ion are found to decrease dramatically with its size. Stereoscopic pictures show that the structure of the solvent cage around an ion is qualitatively different for the larger ions, implicating both solvent dynamics and structure as important factors in explaining ion mobility in aqueous systems. -

UNITED STATES PATENT OFFICE 2,56,31 METHOD of REDUCING and by DRO GENATING CHEMICA, COMPOUNDS by REACTING WITE: ALUMNUM-CONAN NG BYOFREDES Hermann E
Patented Nov. 27, 1951 2,576,31 UNITED STATES PATENT OFFICE 2,56,31 METHOD OF REDUCING AND BY DRO GENATING CHEMICA, COMPOUNDS BY REACTING WITE: ALUMNUM-CONAN NG BYOFREDES Hermann E. Schlesirager and Albert E. Finholt, Chicago, Ill.; said Schlesinger assignor of one fourth to. Edaa, M. Schlesinger and said Fin holt assignor of one-fourth to Marion H. Finholt No Drawing. Application June 3, 1947, Serial No. 752,286 2 (Cairns. (C. 260-638) 2 This invention relates to methods of making LiAlH4. Although this new compound will be aluminum-containing hydrides and the reactions called lithium aluminum hydride in the present thereof, and also relates to products prepared by application, it may also be called lithium alumi said methods. nohydride or lithium tetrahydroaluminide. In This application is a continuation-in-part of one method of making lithium aluminum hydride, our copending application Serial No. 717,312, filed lithium hydride is reacted with an aluminum December 19, 1946, now Patent No. 2,567,972, halide such as aluminum chloride in the presence issued September 18, 1951. of a suitable liquid medium such as an ether. If We have discovered that these compounds, es the reagents are mixed in the proportions of the pecially the ether soluble lithium aluminum hy 0 following equation, or if an excess of lithium hy dride, are extremely useful chemical reagents. dride is used, the reaction proceeds as follows: - They may be employed for replacing halogens or Organic radicals by hydrogen in a great variety 4Li H--AlCl3->LiAlH4--3LiCl of compounds. As a result, their discovery has led to new methods, safer, more convenient, and 16 The liquid medium used is one in which one of more efficient than those hitherto known, for pro the reaction products, e. -

Halides and Halogens. What Do I Need to Know? John Vivari, Nordson EFD
Halides and Halogens. What do I need to know? John Vivari, Nordson EFD Abstract With halogen-containing substances in the public eye due to scrutiny by the European Union and a variety of non- governmental organizations (NGOs) as possible additions to the list of substances banned from electronics, we at EFD have received numerous inquiries from customers asking how this subject will affect them and their processes. Having just overcome the hurdle of RoHS (Restriction of Hazardous Substances), they want to know what halogens and halides are, and what changes they should be prepared for if required to stop using them. Halide-free materials are not new. Some segments of the electronics industry have been sensitive to halides and their significance for decades. This paper will give the reader a working knowledge of halogens and halides. Armed with this education, the reader will be able to make informed decisions when required to use halogen-free materials, either because regulations dictate it or social pressure makes acceptance preferable to resistance. Key Words: halide, halogen, bromine, chlorine, flame retardant, RoHS What are halogens and halides? damage. Brominated flame retardant use is not limited to electronics. It is also in common usage in furniture, At their most basic level, halogens are the electronegative construction materials and textiles. elements in column 17 of the periodic table, including fluorine (F), chlorine, (Cl), bromine (Br), iodine (I) and Other sources of halogens in circuit boards include astatine (At). In electronics fiberglass sizing, epoxy curing agents and accelerators, applications, iodine and resin wetting and de-foaming agents, flux residues, and astatine are rarely if ever contamination from handling. -

P – Block Elements SYJC
P – Block Elements Introduction The p-block elements are placed in groups 13 – 18 . The general electronic configuration is ns 2 np1 – 6. The groups included in the syllabus are 15, 16, 17 and 18. Group 15 Elements Nitrogen family: configuration is ns2np3. The elements of group 15 – nitrogen (N), phosphorus (P), arsenic (As), antimony (Sb) bismuth (Bi) All Group 15 Elements tend to follow the general periodic trends: Periodic properties Trends Electronegativity:(the atom's ability of Decreases down the group attracting electrons) Ionization Enthalpy (the amount of decreases energy required to remove an electron from the atom in it's gaseous phase) Atomic Radii (the radius of the atom) increases Electron Affinity (ability of the atom to decreases accept an electron) Melting Point (amount of energy increases going down the required to break bonds to change a group solid phase substance to a liquid phase) Boiling Point (amount of energy increases going down the required to break bonds to change a group liquid phase substance to a gas) Chemical properties Action of air;(high temp arc) N2 + O2 2NO Action oxidizing agents: P4 +20HNO3 4H3PO4 + 20 NO2+4 H20 As4 + 20 HNO3 4H3AsO4 + 20 NO2+4 H20 Action of hot conc H2SO4 P4 +10 H2SO4 4H3PO4 + 10 SO2+4 H20 As4 +10 H2SO4 4H3AsO4 + 4 Sb + 6 H2SO4 Sb2(SO4)3 + 3 Hydrides All form hydrides with formula EH3 ( E = N, P, As, Sb , Bi) oxidation state = – 3 Hydrogen bonding in NH3 The stability of hydrides decrease down the group due to decrease in bond Hydrides comparison Anomalous behaviour of -

Kinetics and Mechanism of Oxidation of Glycine and Alanine by Oxone R
http://dx.doi.org/10.5935/0103-5053.20140139 J. Braz. Chem. Soc., Vol. 25, No. 9, 1545-1551, 2014. Printed in Brazil - ©2014 Sociedade Brasileira de Química Article 0103 - 5053 $6.00+0.00 A Kinetics and Mechanism of Oxidation of Glycine and Alanine by Oxone® Catalyzed by Bromide Ion Malharrao R. Thombare and Gavisiddappa S. Gokavi* Kinetics and Catalysis Laboratory, Department of Chemistry, Shivaji University, 416004 Kolhapur, India A oxidação da glicina e alanina por Oxone®, catalisada por íons brometo foi estudada em meio ácido. A reação é iniciada pela oxidação do brometo ao bromo, que reage com o aminoácido. A formação de bromo é comprovada pelo exame espectrofotométrico da mistura reacional. O intermediário proposto envolve a formação de um complexo entre o bromo e o ânion do aminoácido. A taxa de reação é inibida por um aumento na concentração do íon hidrogênio devido ao equilíbrio de protonação dos aminoácidos. Um mecanismo é proposto e a lei da razão derivada foi verificada graficamente. O efeito da permissividade relativa, força iônica e temperatura também foram acompanhados e esses efeitos também dão suporte ao mecanismo proposto. Oxidation of glycine and alanine by Oxone® catalysed by bromide ions has been studied in acidic medium. The reaction is initiated by the oxidation of bromide to bromine, which then reacts with the amino acid. The formation of bromine is supported by the spectrophotometric examination of the reaction mixture. The proposed intermediate involves a complex formation between bromine and the anion of the amino acid. The rate of the reaction is inhibited by an increase in the hydrogen ion concentration due to the protonation equilibria of the amino acids. -

Reactions of Alkenes and Alkynes
05 Reactions of Alkenes and Alkynes Polyethylene is the most widely used plastic, making up items such as packing foam, plastic bottles, and plastic utensils (top: © Jon Larson/iStockphoto; middle: GNL Media/Digital Vision/Getty Images, Inc.; bottom: © Lakhesis/iStockphoto). Inset: A model of ethylene. KEY QUESTIONS 5.1 What Are the Characteristic Reactions of Alkenes? 5.8 How Can Alkynes Be Reduced to Alkenes and 5.2 What Is a Reaction Mechanism? Alkanes? 5.3 What Are the Mechanisms of Electrophilic Additions HOW TO to Alkenes? 5.1 How to Draw Mechanisms 5.4 What Are Carbocation Rearrangements? 5.5 What Is Hydroboration–Oxidation of an Alkene? CHEMICAL CONNECTIONS 5.6 How Can an Alkene Be Reduced to an Alkane? 5A Catalytic Cracking and the Importance of Alkenes 5.7 How Can an Acetylide Anion Be Used to Create a New Carbon–Carbon Bond? IN THIS CHAPTER, we begin our systematic study of organic reactions and their mecha- nisms. Reaction mechanisms are step-by-step descriptions of how reactions proceed and are one of the most important unifying concepts in organic chemistry. We use the reactions of alkenes as the vehicle to introduce this concept. 129 130 CHAPTER 5 Reactions of Alkenes and Alkynes 5.1 What Are the Characteristic Reactions of Alkenes? The most characteristic reaction of alkenes is addition to the carbon–carbon double bond in such a way that the pi bond is broken and, in its place, sigma bonds are formed to two new atoms or groups of atoms. Several examples of reactions at the carbon–carbon double bond are shown in Table 5.1, along with the descriptive name(s) associated with each. -
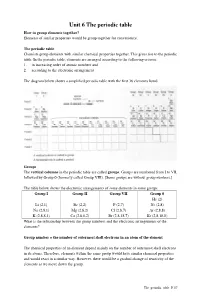
Unit 6 the Periodic Table How to Group Elements Together? Elements of Similar Properties Would Be Group Together for Convenience
Unit 6 The periodic table How to group elements together? Elements of similar properties would be group together for convenience. The periodic table Chemists group elements with similar chemical properties together. This gives rise to the periodic table. In the periodic table, elements are arranged according to the following criteria: 1. in increasing order of atomic numbers and 2. according to the electronic arrangement The diagram below shows a simplified periodic table with the first 36 elements listed. Groups The vertical columns in the periodic table are called groups . Groups are numbered from I to VII, followed by Group 0 (formerly called Group VIII). [Some groups are without group numbers.] The table below shows the electronic arrangements of some elements in some groups. Group I Group II Group VII Group 0 He (2) Li (2,1) Be (2,2) F (2,7) Ne (2,8) Na (2,8,1) Mg (2,8,2) Cl (2,8,7) Ar (2,8,8) K (2,8,8,1) Ca (2,8,8,2) Br (2,8,18,7) Kr (2,8,18,8) What is the relationship between the group numbers and the electronic arrangements of the elements? Group number = the number of outermost shell electrons in an atom of the element The chemical properties of an element depend mainly on the number of outermost shell electrons in its atoms. Therefore, elements within the same group would have similar chemical properties and would react in a similar way. However, there would be a gradual change of reactivity of the elements as we move down the group. -

The P-Block Elements Properties and Uses of Dioxygen Are Placed in Groups 13 to 18 of the Periodic Table
77UnitUnitUnit Objectives TheThe pp -Block-Block After studying this Unit, you will be able to ElementElementss • appreciate general trends in the chemistry of elements of groups 15,16,17 and 18; Diversity in chemistry is the hallmark of p–block elements manifested • learn the preparation, properties in their ability to react with the elements of s–, d– and f–blocks as and uses of dinitrogen and well as with their own. phosphorus and some of their important compounds; • describe the preparation, In Class XI, you have learnt that the p-block elements properties and uses of dioxygen are placed in groups 13 to 18 of the periodic table. and ozone and chemistry of some Their valence shell electronic configuration is ns2np1–6 simple oxides; (except He which has 1s2 configuration). The properties • know allotropic forms of sulphur, of p-block elements like that of others are greatly chemistry of its important influenced by atomic sizes, ionisation enthalpy, electron compounds and the structures of gain enthalpy and electronegativity. The absence of d- its oxoacids; orbitals in second period and presence of d or d and f • describe the preparation, orbitals in heavier elements (starting from third period properties and uses of chlorine onwards) have significant effects on the properties of and hydrochloric acid; elements. In addition, the presence of all the three types • know the chemistry of interhalogens and structures of of elements; metals, metalloids and non-metals bring oxoacids of halogens; diversification in chemistry of these elements. • enumerate the uses of noble Having learnt the chemistry of elements of Groups gases; 13 and 14 of the p-block of periodic table in Class XI, • appreciate the importance of you will learn the chemistry of the elements of these elements and their subsequent groups in this Unit.