Provider Manual
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

From the Academy
FROM THE ACADEMY Joint American Academy of DermatologyeNational Psoriasis Foundation guidelines of care for the management and treatment of psoriasis with phototherapy Craig A. Elmets, MD (Co-Chair),a HenryW.Lim,MD,b Benjamin Stoff, MD, MA,c Cody Connor, MD,a Kelly M. Cordoro, MD,d Mark Lebwohl, MD,e AprilW.Armstrong,MD,MPH,f Dawn M. R. Davis, MD,g Boni E. Elewski, MD,a Joel M. Gelfand, MD, MSCE,h Kenneth B. Gordon, MD,i AliceB.Gottlieb,MD,PhD,j Daniel H. Kaplan, MD, PhD,k Arthur Kavanaugh, MD,l Matthew Kiselica, BA/BS,m Dario Kivelevitch, MD,n Neil J. Korman, MD, PhD,o Daniela Kroshinsky, MD, MPH,p Craig L. Leonardi, MD,q Jason Lichten, MD,m NehalN.Mehta,MD,MSCE,r Amy S. Paller, MD,s Sylvia L. Parra, MD,t Arun L. Pathy, MD,u Elizabeth A. Farley Prater, MD,v Reena N. Rupani, MD,e Michael Siegel, PhD,m BruceE.Strober,MD,PhD,w,x Emily B. Wong, MD,y Jashin J. Wu, MD,z Vidhya Hariharan, PhD,aa and Alan Menter, MD (Co-Chair)n Birmingham, Alabama; Detroit, Michigan; Atlanta, Georgia; San Francisco, Los Angeles, San Diego, and Irvine, California; New York, New York; Rochester, Minnesota; Philadelphia and Pittsburgh, Pennsylva- nia; Milwaukee, Wisconsin; Portland, Oregon; Dallas and San Antonio, Texas; Cleveland, Ohio; Boston, Massachusetts; St. Louis, Missouri; Bethesda, Maryland; Chicago and Rosemont, Illinois; Sumter, South Carolina; Centennial, Colorado; Oklahoma City, Oklahoma; Farmington, Connecticut; and Waterloo, Ontario, Canada Psoriasis is a chronic inflammatory disease involving multiple organ systems and affecting approximately 3.2% of the world’s population. -

Crohn's & Colitis Program, Patient Information Guide
Inflammatory Bowel Disease Program Patient Information Guide Page # Contents ..........................................................................................................................................1 Welcome! Welcome Letter ....................................................................................................................3 Important Things to Know Up Front ....................................................................................4 Meet Your Crohn’s & Colitis Team .....................................................................................6 How to Contact Us Crohn’s & Colitis Clinic Schedule .....................................................................................11 Physician and Nurse Contact Information..........................................................................12 Appointment Scheduling and Other Contact Information..................................................13 The Basics of Inflammatory Bowel Disease (IBD) Basic Information about Inflammatory Bowel Disease (IBD) ...........................................14 Frequently Asked Questions about Inflammatory Bowel Disease (IBD) ..........................18 Testing in IBD Colonoscopy and Flexible Sigmoidoscopy ........................................................................20 Upper Endoscopy ...............................................................................................................21 Capsule Endoscopy and Deep Enteroscopy .......................................................................22 -

Annual Report on Annual Reports 2016
ANNUAL REPORT ON ANNUAL REPORTS 2016 TOP 400 ANNUAL REPORTS WHO RANKS WHERE? 100 ANNUALS IN BRIEF BEST REPORTING PRACTICES Company Value > Report Value Annual Report on Annual Reports 2016 Contents Report rating scale 3 Top 400 annual reports 4 Who ranks where? 25 From ABB to ZTE 100 annuals in brief 58 From Abbott to Yamaha How important is the annual report today? 92 Views from Cecilia Ketels, Kellie Friery, Renee Carter, David Robinson, Kaevan Gazdar, Elena Moskvina, Thomas Rosenmayr, Rob Stangroom, Andrey Kozhevnikov, Ana Santamarina, Katie Holcomb, Ananda Jagoda Best practices on key report attibutes 100 Strategy, message, investor information, risks, style, online… How we make it 121 How is your report doing? The report scan 127 The report rating panel 128 Robert Berick, Susan Blesener, Renee Carter, Vero Escarmelle, Helena Fournial, Kaevan Gazdar, Mike Guillaume, Pradip Seth, Eva Wolosiuk Making reports pay off 133 e.com – ReportWatch 135 2 Report rating scale A+ ééééé First-rate A éééé(é) Excellent A- éééé Very good B+ ééé(é) Sound B ééé Average B- éé(é) Uneven C+ éé Common C é(é) Substandard C- é Poor D (é) Uncompetitive 3 Top 400 annual reports AkzoNobel (No. 1) Electrolux (No. 2 ) SCA (No. 3) Volvo (No. 4) 4 Report rank Company Country Report rating Compare 1 AKZONOBEL Netherlands A+ DUPONT 2 ELECTROLUX Sweden A+ WHIRLPOOL 3 SCA Sweden A+ KIMBERLY-CLARK 4 VOLVO Sweden A+ DAIMLER 5 POTASHCORP Canada A+ AGRIUM 6 ATLAS COPCO Sweden A SANDVIK 7 STORA ENSO Finland A UPM 8 BOLIDEN Sweden A GLENCORE 9 WIENERBERGER Austria A BORAL 10 -

Inflammatory Bowel Disease Agents
Therapeutic Class Overview Inflammatory Bowel Disease Agents INTRODUCTION • Inflammatory bowel disease (IBD) is a spectrum of chronic idiopathic inflammatory intestinal conditions that cause gastrointestinal symptoms including diarrhea, abdominal pain, bleeding, fatigue, and weight loss. The exact cause of IBD is unknown; however, proposed etiologies involve a combination of infectious, genetic, and lifestyle factors (Bernstein et al 2015, Peppercorn 2019[a], Peppercorn 2020[c]). • Complications of IBD include hemorrhage, rectal fissures, fistulas, peri-rectal and intra-abdominal abscesses, and colon cancer. Possible extra-intestinal complications include hepatobiliary complications, anemia, arthritis and arthralgias, uveitis, skin lesions, and mood and anxiety disorders (Bernstein et al 2015). • Ulcerative colitis (UC) and Crohn’s disease (CD) are 2 forms of IBD that differ in pathophysiology and presentation; as a result of these differences, the approach to the treatment of each condition often differs (Peppercorn 2019[a]). • UC is characterized by recurrent episodes of inflammation of the mucosal layer of the colon. The inflammation, limited to the mucosa, commonly involves the rectum and may extend in a proximal and continuous fashion to affect other parts of the colon. The hallmark clinical symptom is an inflamed rectum with symptoms of urgency, bleeding, and tenesmus (Peppercorn 2020[c], Rubin et al 2019). • CD can involve any part of the gastrointestinal tract and is characterized by transmural inflammation and “skip areas.” Transmural inflammation may lead to fibrosis, strictures, sinus tracts, and fistulae (Peppercorn 2019[b]). • The immune system is known to play a critical role in the underlying pathogenesis of IBD. It is suggested that abnormal responses of both innate and adaptive immunity mechanisms induce aberrant intestinal tract inflammation in IBD patients (Geremia et al 2014). -

Prediction of Premature Termination Codon Suppressing Compounds for Treatment of Duchenne Muscular Dystrophy Using Machine Learning
Prediction of Premature Termination Codon Suppressing Compounds for Treatment of Duchenne Muscular Dystrophy using Machine Learning Kate Wang et al. Supplemental Table S1. Drugs selected by Pharmacophore-based, ML-based and DL- based search in the FDA-approved drugs database Pharmacophore WEKA TF 1-Palmitoyl-2-oleoyl-sn-glycero-3- 5-O-phosphono-alpha-D- (phospho-rac-(1-glycerol)) ribofuranosyl diphosphate Acarbose Amikacin Acetylcarnitine Acetarsol Arbutamine Acetylcholine Adenosine Aldehydo-N-Acetyl-D- Benserazide Acyclovir Glucosamine Bisoprolol Adefovir dipivoxil Alendronic acid Brivudine Alfentanil Alginic acid Cefamandole Alitretinoin alpha-Arbutin Cefdinir Azithromycin Amikacin Cefixime Balsalazide Amiloride Cefonicid Bethanechol Arbutin Ceforanide Bicalutamide Ascorbic acid calcium salt Cefotetan Calcium glubionate Auranofin Ceftibuten Cangrelor Azacitidine Ceftolozane Capecitabine Benserazide Cerivastatin Carbamoylcholine Besifloxacin Chlortetracycline Carisoprodol beta-L-fructofuranose Cilastatin Chlorobutanol Bictegravir Citicoline Cidofovir Bismuth subgallate Cladribine Clodronic acid Bleomycin Clarithromycin Colistimethate Bortezomib Clindamycin Cyclandelate Bromotheophylline Clofarabine Dexpanthenol Calcium threonate Cromoglicic acid Edoxudine Capecitabine Demeclocycline Elbasvir Capreomycin Diaminopropanol tetraacetic acid Erdosteine Carbidopa Diazolidinylurea Ethchlorvynol Carbocisteine Dibekacin Ethinamate Carboplatin Dinoprostone Famotidine Cefotetan Dipyridamole Fidaxomicin Chlormerodrin Doripenem Flavin adenine dinucleotide -

The Efficacy of Probiotics, Prebiotic Inulin-Type Fructans, and Synbiotics in Human Ulcerative Colitis: a Systematic Review and Meta-Analysis
Supplementary Materials and Data of Review: The Efficacy of Probiotics, Prebiotic Inulin-Type Fructans, and Synbiotics in Human Ulcerative Colitis: A Systematic Review and Meta-Analysis Supplementary Data S1 Search strategy Only clinical trial publications and human filters were used in the literature search performed in PUBMED. The studies were searched using the terms “ulcerative colitis” in combination with “probiotic”, “synbiotic”, and “inflammatory bowel disease” in combination with “prebiotic”, “inulin”, “FOS” and “fructo-oligosaccharides”. The search in SCOPUS was performed using combinations of the terms “ulcerative colitis” AND “inulin” limited to document types “Article”. Another search used “ulcerative colitis” AND “probiotic” AND “human” limited to document type “Article” and the subareas “Medicine”, “Immunology and Microbiology” and “Health Professions” and the exact key words “Controlled Study”, “Clinical Article”, “Clinical Trials”, “Controlled Clinical Trial” and “Double-Blind Method”. An additional search using the combinations “ulcerative colitis” AND “synbiotic” AND “human” limited to document type “Article” and excluding the exact key word “Crohn Disease”. In all cases, the search was limited to the languages “Spanish” and “English” Supplementary Table S1 Risk of bias of selected studies and and data data bias bias of generation generation assessment) reporting outcome concealment sources (participants personnel) personnel) Sequence (outcome Selective Other Allocation Incomplete Blinding Random Blinding Matsuoka et al. 2018 ? + + + + + + Tamaki et al. 2016 ? + + ? - ? + Yoshimatsu et al. 2015 ? ? + ? - + + Petersen et al. 2014 ? + + ? + + + Groeger et al. 2013 - - + ? - ? + Oliva et al. 2012 + + + ? - + + Wildt et al. 2011 ? + + ? + + + Tursi et al. 2010 ? + + ? + ? + Matthes et al. 2010 ? + + ? - + + Ng et al. 2010 ? - + ? + ? + Sood et al. 2009 + + + ? - ? + Miele et al. 2009 + + + ? - ? + Furrie et al. -

Stems for Nonproprietary Drug Names
USAN STEM LIST STEM DEFINITION EXAMPLES -abine (see -arabine, -citabine) -ac anti-inflammatory agents (acetic acid derivatives) bromfenac dexpemedolac -acetam (see -racetam) -adol or analgesics (mixed opiate receptor agonists/ tazadolene -adol- antagonists) spiradolene levonantradol -adox antibacterials (quinoline dioxide derivatives) carbadox -afenone antiarrhythmics (propafenone derivatives) alprafenone diprafenonex -afil PDE5 inhibitors tadalafil -aj- antiarrhythmics (ajmaline derivatives) lorajmine -aldrate antacid aluminum salts magaldrate -algron alpha1 - and alpha2 - adrenoreceptor agonists dabuzalgron -alol combined alpha and beta blockers labetalol medroxalol -amidis antimyloidotics tafamidis -amivir (see -vir) -ampa ionotropic non-NMDA glutamate receptors (AMPA and/or KA receptors) subgroup: -ampanel antagonists becampanel -ampator modulators forampator -anib angiogenesis inhibitors pegaptanib cediranib 1 subgroup: -siranib siRNA bevasiranib -andr- androgens nandrolone -anserin serotonin 5-HT2 receptor antagonists altanserin tropanserin adatanserin -antel anthelmintics (undefined group) carbantel subgroup: -quantel 2-deoxoparaherquamide A derivatives derquantel -antrone antineoplastics; anthraquinone derivatives pixantrone -apsel P-selectin antagonists torapsel -arabine antineoplastics (arabinofuranosyl derivatives) fazarabine fludarabine aril-, -aril, -aril- antiviral (arildone derivatives) pleconaril arildone fosarilate -arit antirheumatics (lobenzarit type) lobenzarit clobuzarit -arol anticoagulants (dicumarol type) dicumarol -
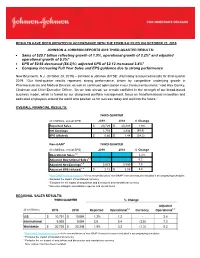
Sales of $20.7 Billion Reflecting Growth of 1.9%, Operational Growth of 3.2
RESULTS HAVE BEEN UPDATED IN ACCORDANCE WITH THE FORM 8-K FILED ON OCTOBER 23, 2019 JOHNSON & JOHNSON REPORTS 2019 THIRD-QUARTER RESULTS: • Sales of $20.7 billion reflecting growth of 1.9%, operational growth of 3.2%* and adjusted operational growth of 5.2%* • EPS of $0.66 decreased (54.2)%; adjusted EPS of $2.12 increased 3.4%* • Company increasing Full Year Sales and EPS guidance due to strong performance New Brunswick, N.J. (October 23, 2019) – Johnson & Johnson (NYSE: JNJ) today announced results for third-quarter 2019. “Our third-quarter results represent strong performance, driven by competitive underlying growth in Pharmaceuticals and Medical Devices, as well as continued optimization in our Consumer business,” said Alex Gorsky, Chairman and Chief Executive Officer. “As we look ahead, we remain confident in the strength of our broad-based business model, which is fueled by our disciplined portfolio management, focus on transformational innovation and dedicated employees around the world who position us for success today and well into the future.” OVERALL FINANCIAL RESULTS: THIRD QUARTER ($ in Millions, except EPS) 2019 2018 % Change Reported Sales $ 20,729 $ 20,348 1.9% Net Earnings 1,753 3,934 (55.4) EPS (diluted) $ 0.66 $ 1.44 (54.2) Non-GAAP* THIRD QUARTER ($ in Millions, except EPS) 2019 2018 % Change Operational Sales1,2 3.2% Adjusted Operational Sales1,3 5.2 Adjusted Net Earnings1,4 5,672 5,590 1.5 Adjusted EPS (diluted)1,4 $ 2.12 $ 2.05 3.4 1 Non-GAAP financial measure; refer to reconciliations of non-GAAP financial measures -

Amgen 2008 Annual Report and Financial Summary Pioneering Science Delivers Vital Medicines
Amgen 2008 Annual Report and Financial Summary Pioneering science delivers vital medicines About Amgen Products Amgen discovers, develops, manufactures Aranesp® (darbepoetin alfa) and delivers innovative human therapeutics. ® A biotechnology pioneer since 1980, Amgen Enbrel (etanercept) was one of the fi rst companies to realize ® the new science’s promise by bringing EPOGEN (Epoetin alfa) safe and effective medicines from the lab Neulasta® (pegfi lgrastim) to the manufacturing plant to patients. NEUPOGEN® (Filgrastim) Amgen therapeutics have changed the practice of medicine, helping millions of Nplate® (romiplostim) people around the world in the fi ght against ® cancer, kidney disease, rheumatoid Sensipar (cinacalcet) arthritis and other serious illnesses, and so Vectibix® (panitumumab) far, more than 15 million patients worldwide have been treated with Amgen products. With a broad and deep pipeline of potential new medicines, Amgen remains committed to advancing science to dramatically improve people’s lives. 0405 06 07 08 0405 06 07 08 0405 06 07 08 0405 06 07 08 Total revenues “Adjusted” earnings Cash fl ow from “Adjusted” research and ($ in millions) per share (EPS)* operations development (R&D) expenses* (Diluted) ($ in millions) ($ in millions) 2008 $15,003 2008 $4.55 2008 $5,988 2008 $2,910 2007 14,771 2007 4.29 2007 5,401 2007 3,064 2006 14,268 2006 3.90 2006 5,389 2006 3,191 2005 12,430 2005 3.20 2005 4,911 2005 2,302 2004 10,550 2004 2.40 2004 3,697 2004 1,996 * “ Adjusted” EPS and “adjusted” R&D expenses are non-GAAP fi nancial measures. See page 8 for reconciliations of these non-GAAP fi nancial measures to U.S. -

National Quality Forum
NQF #PSM-030-10 NATIONAL QUALITY FORUM Measure Evaluation 4.1 January 2010 This form contains the measure information submitted by stewards. Blank fields indicate no information was provided. Attachments also may have been submitted and are provided to reviewers. The sub-criteria and most of the footnotes from the evaluation criteria are provided in Word comments and will appear if your cursor is over the highlighted area (or in the margin if your Word program is set to show revisions in balloons). Hyperlinks to the evaluation criteria and ratings are provided in each section. TAP/Workgroup (if utilized): Complete all yellow highlighted areas of the form. Evaluate the extent to which each sub-criterion is met. Based on your evaluation, summarize the strengths and weaknesses in each section. Note: If there is no TAP or workgroup, the SC also evaluates the sub-criteria (yellow highlighted areas). Steering Committee: Complete all pink highlighted areas of the form. Review the workgroup/TAP assessment of the sub-criterion, noting any areas of disagreement; then evaluate the extent to which each major criterion is met; and finally, indicate your recommendation for the endorsement. Provide the rationale for your ratings. Evaluation ratings of the extent to which the criteria are met C = Completely (unquestionably demonstrated to meet the criterion) P = Partially (demonstrated to partially meet the criterion) M = Minimally (addressed BUT demonstrated to only minimally meet the criterion) N = Not at all (NOT addressed; OR incorrectly addressed; OR demonstrated to NOT meet the criterion) NA = Not applicable (only an option for a few sub-criteria as indicated) (for NQF staff use) NQF Review #: PSM-030-10 NQF Project: Patient Safety Measures MEASURE DESCRIPTIVE INFORMATION De.1 Measure Title: Patient(s) with inflammatory bowel disease taking methotrexate, sulfasalazine, mercaptopurine, or azathioprine that had a CBC in last 3 reported months. -

AILERON THERAPEUTICS, INC. (Name of Registrant As Specified in Its Charter)
Table of Contents UNITED STATES SECURITIES AND EXCHANGE COMMISSION Washington, D.C. 20549 SCHEDULE 14A Proxy Statement Pursuant to Section 14(a) of the Securities Exchange Act of 1934 (Amendment No. ) Filed by the Registrant ☒ Filed by a Party other than the Registrant ☐ Check the appropriate box: ☒ Preliminary Proxy Statement ☐ Confidential, for Use of the Commission Only (as permitted by Rule 14a-6(e)(2)) ☐ Definitive Proxy Statement ☐ Definitive Additional Materials ☐ Soliciting Material Pursuant to §240.14a-12 AILERON THERAPEUTICS, INC. (Name of Registrant as Specified in Its Charter) (Name of Person(s) Filing Proxy Statement, if Other Than the Registrant) Payment of Filing Fee (Check the appropriate box): ☒ No fee required. ☐ Fee computed on table below per Exchange Act Rules 14a-6(i)(1) and 0-11. (1) Title of each class of securities to which transaction applies: (2) Aggregate number of securities to which transaction applies: (3) Per unit price or other underlying value of transaction computed pursuant to Exchange Act Rule 0-11 (set forth the amount on which the filing fee is calculated and state how it was determined): (4) Proposed maximum aggregate value of transaction: (5) Total fee paid: ☐ Fee paid previously with preliminary materials. ☐ Check box if any part of the fee is offset as provided by Exchange Act Rule 0-11(a)(2) and identify the filing for which the offsetting fee was paid previously. Identify the previous filing by registration statement number, or the Form or Schedule and the date of its filing. (1) Amount previously paid: (2) Form, Schedule or Registration Statement No.: (3) Filing Party: (4) Date Filed: Table of Contents AILERON THERAPEUTICS, INC. -

UNITED STATES SECURITIES and EXCHANGE COMMISSION Form
Table of Contents UNITED STATES SECURITIES AND EXCHANGE COMMISSION Washington D.C. 20549 Form 10-K (Mark One) ANNUAL REPORT PURSUANT TO SECTION 13 OR 15(d) OF THE SECURITIES EXCHANGE ACT OF ☒ 1934 For the fiscal year ended December 31, 2008 OR TRANSITION REPORT PURSUANT TO SECTION 13 OR 15(d) OF THE SECURITIES EXCHANGE ACT ☐ OF 1934 Commission file number 000-12477 Amgen Inc. (Exact name of registrant as specified in its charter) Delaware 95-3540776 (State or other jurisdiction of (I.R.S. Employer incorporation or organization) Identification No.) One Amgen Center Drive, 91320-1799 Thousand Oaks, California (Zip Code) (Address of principal executive offices) (805) 447-1000 (Registrant’s telephone number, including area code) Securities registered pursuant to Section 12(g) of the Act: Common stock, $0.0001 par value; preferred share purchase rights (Title of class) Indicate by check mark if the registrant is a well-known seasoned issuer, as defined in Rule 405 of the Securities Act. Yes ☒ No ☐ Indicate by check mark if the registrant is not required to file reports pursuant to Section 13 or Section 15(d) of the Act. Yes ☐ No ☒ Indicate by check mark whether the registrant (1) has filed all reports required to be filed by Section 13 or Section 15(d) of the Securities Exchange Act of 1934 during the preceding 12 months (or for such shorter period that the registrant was required to file such reports), and (2) has been subject to such filing requirements for the past 90 days. Yes ☒ No ☐ Indicate by check mark if disclosure of delinquent filers pursuant to Item 405 of Regulation S-K is not contained herein, and will not be contained, to the best of registrant’s knowledge, in definitive proxy or information statements incorporated by reference in Part III of this Form 10-K or any amendment to this Form 10-K.