(PB) Exposure in the Aging Brain: Relevance to Alzheimer's Disease
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Kupferstichkabinett-Liste-Stecher.Pdf
Stand Februar 2013 Name Vorname Lebensdaten Aarland W. tätig um 1860 Aarlander A.v.W. 19.Jh. Aartandi W. 19.Jh. Abbema Wilhelm von 1812 - 1889 Abel Gottlieb Friedrich 1763 - 1794 Aberli Johann Ludwig 1723 - 1786 Abramson Michel Jacob tätig Ende 18.Jh. Achenbach Hans 1891 - 1972 Achenbach Andreas 1815 - 1910 Acin Katja *1900 Ackermann Helmut *1936 Ackermann Peter *1934 Ackermann R. 19.Jh. Ackermann Franz *1934 Acqua Christoforo dall 1734 - 1787 Adam Jacob 1784 - 1811 Adam Benno 1812 - 1892 Adam Georg 1784 - 1823 Adam Franz 1815 - 1886 Adam Albrecht 1786 - 1862 Adam Victor Vincent 1801 - 1866 Adam Julius 1826 - 1874 Adam Philipp tätig 2.H. 17.Jh. Adam Pierre 1799 - 1853 Adam Michael 1865 - 1915 Adami Valerio 1935 - Ade J. tätig 2.H.19.Jh. Adler tätig um 1785 Adler Jankel 1895 - 1949 Adler W. 19.Jh. Adrien 19.Jh. Aelst Nicolaus van geb. um 1526 Agar Johann Samuel 1770 - 1820 Agostino da Verona tätig um 1647 Agricola Christoph Ludwig 1667 - 1719 Ahrendts Konrad 1855 - 1901 Ahrens P. 19.Jh. Ahrens Georg 1947 - Aigner Robert 1901 - 1966 Akai Fujio 1945 - Akcit Osman 1955 - Aken Jan van tätig 2.H. 17.Jh. Akersloot Willem Outgertsz tätig um 1624 - 1634 Akrel Carl Frederik af 1779 - 1868 Akrel Frederic 1784 - 1804 Albanesi Angelo tätig 2.H. 18.Jh. Albers Anton 1877 - 1915 Albers Josef 1888 - 1976 Albert Hermann 1937 - Albert Joseph 1825 - 1886 Stand Februar 2013 Albert Ferdinand von tätig um1732 Braunschweig Lüneburg Albert, Prinz von 1819 - 1861 Sachsen-Coburg und Gotha Alberti Gasparo 2.H. 16.Jh. Alberti Ignaz 1760 - 1794 Alberti auch gen. -

Creation of Ultracold Rbcs Ground-State Molecules Markus
Creation of Ultracold RbCs Ground-State Molecules DISSERTATION by Markus Debatin submitted to the Faculty of Mathematics, Computer Science and Physics of the University of Innsbruck in partial fulfillment of the requirements for the degree of doctor of science advisor: Univ.Prof. Dr. Hanns-Christoph N¨agerl, Institute of Experimental Physics, University of Innsbruck Innsbruck, July 2013 Abstract Control of external and internal degrees of freedom at the level of single quantum states is essential for a series of molecular physics experiments. Heteronuclear dimers feature a large electric dipole moment, which makes them particular interesting candidates for experiments with strongly interacting quantum gases. However efficient cooling schemes such as laser cooling are difficult to realize for molecules. Therefore we create ultracold molecules from already cooled ultracold gases. This this work presents a series of experiments that has been carried out in order to create the molecules in their lowest internal quantum state. Techniques to create ultracold atoms are readily available. However, the creation of stable mixtures is still a challenge since inter-species scattering plays a role in addition to intra-species scattering physics. This requires us to cool two clouds of 87Rb and 133Cs separately and merge the clouds afterwards. After overlapping the BECs, we produce weakly bound RbCs molecules using the Feshbach-association technique. We transfer the molecules from the weakly bound state to the lowest vibrational and rotational level of the X1Σ+ electronic ground-state potential. For the transfer the initial and the final state are linked with lasers to an intermediate electronically excited state. The transfer is achieved by the Stimulated Raman Adiabatic Passage (STIRAP) technique. -

Dissertation Investigations of Nitrogen Oxide Plasmas
DISSERTATION INVESTIGATIONS OF NITROGEN OXIDE PLASMAS: FUNDAMENTAL CHEMISTRY AND SURFACE REACTIVITY AND MONITORING STUDENT PERCEPTIONS IN A GENERAL CHEMISTRY RECITATION Submitted by Joshua M. Blechle Department of Chemistry In partial fulfillment of the requirements For the Degree of Doctor of Philosophy Colorado State University Fort Collins, Colorado Fall 2016 Doctoral Committee: Advisor: Ellen R. Fisher Nancy E. Levinger Charles S. Henry Christopher R. Weinberger Copyright by Joshua M. Blechle 2016 All Rights Reserved ABSTRACT INVESTIGATIONS OF NITROGEN OXIDE PLASMAS: FUNDAMENTAL CHEMISTRY AND SURFACE REACTIVITY AND MONITORING STUDENT PERCEPTIONS IN A GENERAL CHEMISTRY RECITATION Part I of this dissertation focuses on investigations of nitrogen oxide plasma systems. With increasing concerns over the environmental presence of NxOy species, there is growing interest in utilizing plasma-assisted conversion techniques. Advances, however, have been limited because of the lack of knowledge regarding the fundamental chemistry of these plasma systems. Understanding the kinetics and thermodynamics of processes in these systems is vital to realizing their potential in a range of applications. Unraveling the complex chemical nature of these systems, however, presents numerous challenges. As such, this work serves as a foundational step in the diagnostics and assessment of these NxOy plasmas. The partitioning of energy within the plasma system is essential to unraveling these complications as it provides insight into both gas and surface reactivity. To obtain this information, techniques such as optical emission spectroscopy (OES), broadband absorption spectroscopy (BAS), and laser induced fluorescence (LIF) were utilized to determine species energetics (vibrational, rotational, translational temperatures). These temperature data provide mechanistic insight and establish the relationships between system parameters and energetic outcomes. -

MATERIALIEN (Hand-) 1990-2013
Sign. DSK Name/Bez. Name/Bez. (Forts.) Ort Sign. KMB 1. Veröffentlichungen, die sich ausschließlich mit Künstlerinnen befassen 1. 1 Kuenstlerinnen und ihre Werke in der... Staatsgalerie Stuttgart: Bestandsaufnahme Stuttgart (bis 2004) (Ordner) DSK: 1.1 1. 2 Kuenstlerinnen von der Antike... bis zur Gegenwart (Kirchb./Zonderg.) Dumont 1979 (in LabiblioS) BG 1979 A 1. 3 Adolf Hoelzel's Schuelerinnen Künstlerinnen setzen neue Maßstäbe Museum Stadt WN 1991 NDM 775 1991 1. 4 Deutsche Kuenstlerinnen des 20. JH Malerinnen, Bildhauerinnen,..., (Evers, U.) Schultheiss 1983 BGD 1 1983 1. 5 Kuenstlerinnen des 20. JH Museum WI 1990 YG WIESBA 30 1990 A 1. 6 Kuenstlerinnen International... 1877-1977 (1 Kop.-Bd.) Schl. B-Charlottenburg 1977 DSK: 1.6 1. 7 BRIGITTE- Serie Malerinnen BRIGITTE 1984 DSK: 1.7 1. 8 Malerinnen aus 5 JH (Gottfried Sello; Kopie) Ellert&Richter-V. 1988 R 1988 A 1. 9 Kuenstlerinnen aus Baden-Wuerttemberg von Maria Caspar-Filser bis heute Landes-Girokasse S 1992 YG STUTTG 9 1992 E 1. 10 Kuenstlerinnen von der Renaissance... bis zur Gegenwart (Heller, Nancy) vgs 1989 NCF 1989 A 1. 11 Und ich sehe nichts, nichts als die... (Berger, Renate) Fischer 1989 FU 3 1989 1. 12 Das Verborgene Museum I Dokum. z. Kunst v. Frauen in B öff. Sammlung. Hentrich 1987 YG BERLINW 20 1987/88 : 1 1. 13 Das Verborgene Museum II Dein Land ist Morgen Hentrich 1987 YG BERLINW 20 1987/88 : 2 1. 14 Kuenstlerinnen (Fischer, Mields, Nemeczek, Windheim) GHS KS 1984 DAD 1984 ZB 1. 15 Kunst von Frauen (Krull, Edith; Kopie) Weidlich 1984 DSK: 1.15 1. -

Publication List 2013 * External Authors Are Starred Environmental Health
2014-01-01 Publication List 2013 * external authors are starred Environmental Health Abbas, S.*, Linseisen, J., Rohrmann, S.*, Chang-Claude, J.*, Peeters, P.H.*, Engel, P.*, Brustad, M.*, Lund, E.*, Skeie, G.*, Olsen, A.*, Tjonneland, A.*, Overvad, K.*, Boutron-Ruault, M.C.*, Clavel- Chapelon, F.*, Fagherazzi, G.*, Kaaks, R.*, Boeing, H.*, Buijsse, B.*, Adarakis, G.*, Ouranos, V.*, Trichopoulou, A.*, Masala, G.*, Krogh, V.*, Mattiello, A.*, Tumino, R.*, Sacerdote, C.*, Buckland, G.*, Suarez, M.V.A.*, Sánchez, M.J.*, Chirlaque, M.D.*, Barricarte, A.*, Amiano, P.*, Manjer, J.*, Wirfält, E.*: Dietary intake of vitamin D and calcium and breast cancer risk in the European Prospective Investigation into Cancer and Nutrition. Nutr. Cancer 65 , 178-187 (2013) Accordini, S.*, Corsico, A.G.*, Braggion, M.*, Gerbase, M.W.*, Gislason, D.*, Gulsvik, A.*, Heinrich, J., Janson, C.*, Jarvis, D.*, Jõgi, R.*, Pin, I.*, Schoefer, Y., Bugiani, M.*, Cazzoletti, L.*, Cerveri, I.*, Marcon, A.*, de Marco, R.*: The cost of persistent asthma in Europe: An international population- based study in adults. Int. Arch. Allergy Immunol. 160 , 93-101 (2013) Aichler, M.Y., Elsner, M., Ludyga, N., Feuchtinger, A., Zangen, V., Maier, S.K., Balluff, B., Schöne, C., Hierber, L., Braselmann, H., Meding, S., Rauser, S., Zischka, H., Aubele, M., Schmitt, M.*, Feith, M.*, Hauck, S.M., Ueffing, M., Langer, R.*, Kuester, B.*, Zitzelsberger, H.F., Höfler, H., Walch, A.K.: Clinical response to chemotherapy in oesophageal adenocarcinoma patients is linked to defects in mitochondria. J. Pathol. 230 , 410-419 (2013) Aichler, M.Y., Walch, A.K.: In brief: The (molecular) pathogenesis of Barrett's oesophagus. -

The Schneider Family of Nöttingen Germany 1650-1744 and the Snider Family in the Colony and State of South Carolina
The Schneider Family of Nöttingen Germany 1650-1744 And the Snider Family in the Colony and State of South Carolina From 1745 to 1900 by Dewey Gene Snyder Seventh great grandson of Hans Jacob Schneider July 15, 2016 If you can contribute material or your comments Please contact: [email protected] © 2016 Dewey Gene Snyder The Schneider Family of Nöttingen Germany Table of Contents Table of Contents ...................................................................................................................... i Table of Figures and illustrations ............................................................................................ iv Acknowledgements ................................................................................................................. ix Elloree Heritage Museum and Cultural Center , Elloree, SC ........................................ ix The Schneider Family from Nöttingen Germany ................................................................... 11 The story behind the story: Nöttingen Germany .................................................................... 12 Explanations of the Plaque Narrative ............................................................................. 16 References for the Schneider Family of Nöttingen Germany ................................................ 18 Descendants of Hans Jacob Schneider b. abt. 1650 ........................................................... 21 Latter Day Saints Family History Center Birth Records ................................................... -

Molecular Physics
This article was downloaded by: [University of Montana] On: 04 December 2013, At: 09:15 Publisher: Taylor & Francis Informa Ltd Registered in England and Wales Registered Number: 1072954 Registered office: Mortimer House, 37-41 Mortimer Street, London W1T 3JH, UK Molecular Physics: An International Journal at the Interface Between Chemistry and Physics Publication details, including instructions for authors and subscription information: http://www.tandfonline.com/loi/tmph20 Manipulation of molecules with electromagnetic fields Mikhail Lemeshko a b c , Roman V. Krems c d , John M. Doyle b & Sabre Kais e a ITAMP, Harvard-Smithsonian Center for Astrophysics , Cambridge , MA , 02138 , USA b Physics Department , Harvard University , Cambridge , MA , 02138 , USA c Kavli Institute for Theoretical Physics , University of California , Santa Barbara , CA , 93106 , USA d Department of Chemistry , University of British Columbia , BC V6T 1Z1, Vancouver , Canada e Departments of Chemistry and Physics , Purdue University , West Lafayette , IN , 47907 , USA Accepted author version posted online: 14 Jun 2013.Published online: 28 Aug 2013. To cite this article: Mikhail Lemeshko , Roman V. Krems , John M. Doyle & Sabre Kais (2013) Manipulation of molecules with electromagnetic fields, Molecular Physics: An International Journal at the Interface Between Chemistry and Physics, 111:12-13, 1648-1682, DOI: 10.1080/00268976.2013.813595 To link to this article: http://dx.doi.org/10.1080/00268976.2013.813595 PLEASE SCROLL DOWN FOR ARTICLE Taylor & Francis makes every effort to ensure the accuracy of all the information (the “Content”) contained in the publications on our platform. However, Taylor & Francis, our agents, and our licensors make no representations or warranties whatsoever as to the accuracy, completeness, or suitability for any purpose of the Content. -

Brooklyn College Commencement
Brooklyn College COMMENCEMENT May 27, 2021 The Star-Spangled Banner (The National Anthem) O say can you see, by the dawn’s early light, What so proudly we hail’d at the twilight’s last gleaming? Whose broad stripes and bright stars through the perilous fight O’er the ramparts we watch’d were so gallantly streaming? And the rocket’s red glare, the bombs bursting in air, Gave proof through the night that our flag was still there. O say does that star-spangled banner yet wave O’er the land of the free and the home of the brave? Fanfare and Alma Mater On campus green, with towers of marble Lifting white spires in air. Oh, Brooklyn is our Alma Mater, And she is wondrous fair. Within the gray halls ever lit, The lamps of learning burn, And ever on to goals of glory Young eager spirits yearn. Traditional Tune, Arranged by Malcolm J. Merriweather Words by Robert Friend, Class of 1934 #BCGrad2021 Cheers to the Class of 2021! Post your photos and videos to Facebook, Instagram, and Twitter using the hashtag#BCGrad2021. Brooklyn College Commencement Ceremony • May 27, 2021 Table of Contents Message From the President ........................................................................................................ 2 Message From the President of the Brooklyn College Alumni Association ....................................................................... 3 College Administration, Schools, Departments, and Interdisciplinary Programs ......................................................................................... 4 BC Celebrates: Resilience -

Download Program Book
The Leadership Alliance is a national consortium of leading research and teaching colleges, universities and private industry. The mission of the Alliance is to develop underrepresented students into outstanding leaders and role models in academia, business and the public sector. Member Institutions Cultivating Excellence Brooklyn College Princeton University THE LEADERSHIP ALLIANCE NATIONAL SYMPOSIUM 2013 Brown University Spelman College Chaminade University of Honolulu Stanford University and Empowering Great Minds Clafin University Tougaloo College Columbia University Tufts University Cornell University University of Chicago Dartmouth College University of Colorado Boulder Dillard University University of Maryland, Baltimore County Harvard University University of Miami Howard University University of Pennsylvania Hunter College University of Puerto Rico Johns Hopkins University University of Virginia Montana State University-Bozeman Vanderbilt University Morehouse College Washington University in St. Louis Morgan State University Xavier University of Louisiana New York University Yale University Associate Member Institution Novartis Institutes for BioMedical Research JULY 26-28, 2013 LEADERSHIP ALLIANCE NATIONAL SYMPOSIUM 2013 Executive Offce 133 Waterman Street • Box 1963 • Providence, RI 02912 401-863-1474 • www.theleadershipalliance.org July 26 – 28 Hilton Stamford Hotel & Executive Meeting Center Welcome to the 2013 Symposium 2013 Planning Committee Leadership Alliance National Symposium Jabbar Bennett Associate Dean, Graduate School (Chair) Associate Dean, Division of Biology and Medicine n behalf of the Leadership Alliance, it gives me great pleasure to welcome you to the 2013 Director, Ofce of Diversity and Multicultural Afairs OLeadership Alliance National Symposium (LANS)! Tis event never ceases to inspire as it Clinical Assistant Professor of Medicine, Te Warren Alpert Medical School features the intellectual capacity and fortitude of diverse scholars and researchers from across the Brown University country. -
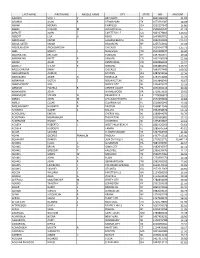
Last Name First Name Middle Name City State Npi Amount
LAST NAME FIRST NAME MIDDLE NAME CITY STATE NPI AMOUNT AARONS SCOTT P BAYTOWN TX 1841386919 31.89 ABANDO ALAN R CEDAR PARK TX 1073561387 18.69 ABAWI HEERAN FAIRFIELD OH 1932337649 16.80 ABBAS HUSAIN M JACKSONVILLE FL 1508026527 31.72 ABBOTT JOHN T FAYETTEVILLE GA 1861679805 120.55 ABBOTT LISA G RENO NV 1609883701 11.19 ABDOLLAHI KARIM LAGUNA BEACH CA 1942240403 215.91 ABDUL SATER SARAH BROOKLYN NY 1255742912 17.26 ABDULMASSIH ABDULMASSIH CHICAGO IL 1629045778 135.71 ABEL MARCY J NASHVILLE TN 1952388977 36.60 ABERNATHIE BRENON YONKERS NY 1497916977 20.44 ABERNATHY BRETT B DENVER CO 1437106028 55.98 ABOAF ALAN P CENTENNIAL CO 1003866393 15.57 ABOSEIF SHERIF R OXNARD CA 1093882995 116.39 ABOU GHAYDA RAMY CHICAGO IL 1093257446 109.36 ABOUHOSSEIN AHMAD DAYTON OH 1487693156 12.36 ABOUKASM AMER G ROSEVILLE MI 1194754085 19.26 ABRAHAM VICTOR E WILMINGTON NC 1003848540 46.67 ABRAMOWITZ JOEL JERSEY CITY NJ 1356429377 13.19 ABRAMS PAMELA B CENTER VALLEY PA 1851502215 30.40 ABRAMSON JOHN WYNNEWOOD PA 1710953633 20.02 ABRAMSON STEVEN N PRAIRIEVILLE LA 1720086820 13.21 ABRAN JOHN M CHICAGO HEIGHTS IL 1942293832 14.84 ABREU CESAR R CLEARWATER FL 1518995943 13.44 ABROMOWITZ HOWARD B DAYTON OH 1508812546 49.83 ACHARYA SUJEET S DALLAS TX 1962658674 54.76 ACHOLONU EMEKA CHERRY HILL NJ 1598944076 34.76 ACHUTHAN HEMAMALINI THORNTON CO 1205982881 17.41 ACKERMAN RANDY B VOORHEES NJ 1205893807 30.63 ACOSTA ROBERTO J WEST PALM BEACH FL 1861426306 53.63 ACOSTA RUDOLPH TAMPA FL 1285645564 43.51 ADAIR SANDRA FLOWER MOUND TX 1497784359 31.09 ADAM GEORGE FRANKLIN -

Annual Report 2009
Little Traverse Conservancy 3264 Powell Road Non-Profit Org. Carlin Smith, Chair Harbor Springs, MI 49740-9469 U.S. POSTAGE PAID John A. Griffin, Vice Chair 231.347.0991 Conway, MI 49722 Michael J. FitzSimons, Secretary Forwarding Service Requested Permit No. 908 Joey Arbaugh Do Not Return John T. Baker James Bartlett Warner Blow Ian R.N. Bund Nadine Cain Michael Cameron board of trustees George Covington Marilyn Damstra Michael Dow Mary Driggs Mary H. Faculak Jeffrey S. Ford Gregg Garver Jim Gillingham Charles Dawley Carol Jackson Thomas C. Bailey Doug Fuller George Jury EXECUTIVE DIRECTOR staff Cindy Mom Paul Keiswetter Thomas Lagerstrom LAND STEWARDSHIP Dave Kring Marta Olson ASSOCIATE DIRECTOR Kieran Fleming Dianne Litzenburger Mark Paddock Mary Anne Griffin Jay Neff Lisa Loyd R. Hamilton Schirmer Cacia McClain C. T. Martin Ty Ratliff Marsha Tompkins ADMINISTRATIVE STAFF Neil Marzella Mary Ann VanLokeren LAND PROTECTION William T. McCormick Alison Berry Edward G. Voss Mikki Snyder VanVuren Harriet K. McGraw Melissa Hansen Patsy Warner EDITOR Rob Mossburg Joan Winston, Trustee Emeritus ENVIRONMENTAL EDUCATION Anne Fleming COMMUNICATIONS Little Traverse Conservancy 2009 Annual Report page - www.landtrust.org Offield Nature Preserve by Martin McReynolds Offield Nature Preserve LITTLE TRAVERSE CONSERVANCY, INC. SCHEDULE OF FINANCIAL POSITION BY FUND 2009 as of December 31, 2009 Land 12 LAND OPERATING PROTECTION ENDOWMENT TOTAL Protection REVENUES, SUPPORT AND INVESTMENT RESULTS: Projects Contributions and grants $1,064,249 $ 2,693,165 -

2016 Honors Convocation Program
93RD ANNUAL HONORS CONVOCATION Hill Auditorium March 20, 2016 | 2:00 p.m. This year marks the 93rd Honors Convocation held at the University of Michigan since the first was instituted on May 13, 1924, by President Marion LeRoy Burton. On these occasions the University publicly recognizes and commends the undergraduate students in its schools and colleges who have earned distinguished academic records or have excelled as leaders in the community. It is highly appropriate that those students be honored who have most clearly and effectively demonstrated academic excellence, dynamic leadership, and inspirational volunteerism. The Honors Convocation takes rank with the Commencement Exercises among the important ceremonies of the University year. The names of the students who are honored for outstanding achievement this year appear in this program. They include all students who have earned University Honors in both Winter 2015 and Fall 2015, plus all seniors who have earned University Honors in either Winter 2015 or Fall 2015. The William J. Branstrom Freshman Prize recipients are listed, as well—recognizing first year undergraduate students whose academic achievement during their first semester on campus place them in the upper five percent of their school or college class. James B. Angell Scholars— students who receive all “A” grades over consecutive terms —are given a special place in the program. In addition, the student speaker is recognized individually for exemplary contributions to the University community. To all honored students, and to their parents, the University extends its hearty congratulations. Honored Students Honored Faculty Faculty Colleagues and Friends of the University It is a pleasure to welcome you to the 93rd University of Michigan Honors Convocation.