Journal of Cell Science Travelling Fellowship Project Report. Does the Structure of the Pericentriolar Material Control Checkpoi
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Supplemental Information Proximity Interactions Among Centrosome
Current Biology, Volume 24 Supplemental Information Proximity Interactions among Centrosome Components Identify Regulators of Centriole Duplication Elif Nur Firat-Karalar, Navin Rauniyar, John R. Yates III, and Tim Stearns Figure S1 A Myc Streptavidin -tubulin Merge Myc Streptavidin -tubulin Merge BirA*-PLK4 BirA*-CEP63 BirA*- CEP192 BirA*- CEP152 - BirA*-CCDC67 BirA* CEP152 CPAP BirA*- B C Streptavidin PCM1 Merge Myc-BirA* -CEP63 PCM1 -tubulin Merge BirA*- CEP63 DMSO - BirA* CEP63 nocodazole BirA*- CCDC67 Figure S2 A GFP – + – + GFP-CEP152 + – + – Myc-CDK5RAP2 + + + + (225 kDa) Myc-CDK5RAP2 (216 kDa) GFP-CEP152 (27 kDa) GFP Input (5%) IP: GFP B GFP-CEP152 truncation proteins Inputs (5%) IP: GFP kDa 1-7481-10441-1290218-1654749-16541045-16541-7481-10441-1290218-1654749-16541045-1654 250- Myc-CDK5RAP2 150- 150- 100- 75- GFP-CEP152 Figure S3 A B CEP63 – – + – – + GFP CCDC14 KIAA0753 Centrosome + – – + – – GFP-CCDC14 CEP152 binding binding binding targeting – + – – + – GFP-KIAA0753 GFP-KIAA0753 (140 kDa) 1-496 N M C 150- 100- GFP-CCDC14 (115 kDa) 1-424 N M – 136-496 M C – 50- CEP63 (63 kDa) 1-135 N – 37- GFP (27 kDa) 136-424 M – kDa 425-496 C – – Inputs (2%) IP: GFP C GFP-CEP63 truncation proteins D GFP-CEP63 truncation proteins Inputs (5%) IP: GFP Inputs (5%) IP: GFP kDa kDa 1-135136-424425-4961-424136-496FL Ctl 1-135136-424425-4961-424136-496FL Ctl 1-135136-424425-4961-424136-496FL Ctl 1-135136-424425-4961-424136-496FL Ctl Myc- 150- Myc- 100- CCDC14 KIAA0753 100- 100- 75- 75- GFP- GFP- 50- CEP63 50- CEP63 37- 37- Figure S4 A siCtl -

Ninein, a Microtubule Minus-End Anchoring Protein 3015 Analysis As Described Previously (Henderson Et Al., 1994)
Journal of Cell Science 113, 3013-3023 (2000) 3013 Printed in Great Britain © The Company of Biologists Limited 2000 JCS1634 Microtubule minus-end anchorage at centrosomal and non-centrosomal sites: the role of ninein Mette M. Mogensen1,*, Azer Malik1, Matthieu Piel2, Veronique Bouckson-Castaing2 and Michel Bornens2 1Department of Anatomy and Physiology, MSI/WTB complex, Dow Street, University of Dundee, Dundee, DD1 5EH, UK 2Institute Curie, UMR 144-CNRS, 26 Rue d’Ulm, 75248 Paris Cedex 05, France *Author for correspondence (e-mail: [email protected]) Accepted 14 June; published on WWW 9 August 2000 SUMMARY The novel concept of a centrosomal anchoring complex, epithelial cells, where the vast majority of the microtubule which is distinct from the γ-tubulin nucleating complex, has minus-ends are associated with apical non-centrosomal previously been proposed following studies on cochlear sites, suggests that it is not directly involved in microtubule epithelial cells. In this investigation we present evidence nucleation. Ninein seems to play an important role in the from two different cell systems which suggests that the positioning and anchorage of the microtubule minus-ends centrosomal protein ninein is a strong candidate for the in these epithelial cells. Evidence is presented which proposed anchoring complex. suggests that ninein is released from the centrosome, Ninein has recently been observed in cultured fibroblast translocated with the microtubules, and is responsible for cells to localise primarily to the post-mitotic mother the anchorage of microtubule minus-ends to the apical centriole, which is the focus for a classic radial microtubule sites. We propose that ninein is a non-nucleating array. -

Downloaded from Bioscientifica.Com at 10/04/2021 03:09:50PM Via Free Access
242 S-M Ho et al. Regulation of centrosome 24:2 83–96 Research duplication by BPA analogues Bisphenol A and its analogues disrupt centrosome cycle and microtubule dynamics in prostate cancer Shuk-Mei Ho1,2,3,4, Rahul Rao1, Sarah To1,5,6, Emma Schoch1 and Pheruza Tarapore1,2,3 1Department of Environmental Health, University of Cincinnati Medical Center, Cincinnati, Ohio, USA 2Center for Environmental Genetics, University of Cincinnati Medical Center, Cincinnati, Ohio, USA Correspondence 3Cincinnati Cancer Center, Cincinnati, Ohio, USA should be addressed 4Cincinnati Veteran Affairs Hospital Medical Center, Cincinnati, Ohio, USA to S-M Ho or P Tarapore 5Center for Cancer Research, Hudson Institute of Medical Research, Clayton, Victoria, Australia Email 6Monash University, Clayton, Victoria, Australia [email protected] or [email protected] Abstract Humans are increasingly exposed to structural analogues of bisphenol A (BPA), as Key Words BPA is being replaced by these compounds in BPA-free consumer products. We have f endocrine-disrupting previously shown that chronic and developmental exposure to BPA is associated with chemicals increased prostate cancer (PCa) risk in human and animal models. Here, we examine f bisphenol A analogues whether exposure of PCa cells (LNCaP, C4-2) to low-dose BPA and its structural analogues f BPA (BPS, BPF, BPAF, TBBPA, DMBPA and TMBPA) affects centrosome amplification (CA), f BPS a hallmark of cancer initiation and progression. We found that exposure to BPA, BPS, f BPF DMBPA and TBBPA, in descending order, increased the number of cells with CA, in a non- f TBBPA Endocrine-Related Cancer Endocrine-Related monotonic dose–response manner. -

The Emerging Role of Ncrnas and RNA-Binding Proteins in Mitotic Apparatus Formation
non-coding RNA Review The Emerging Role of ncRNAs and RNA-Binding Proteins in Mitotic Apparatus Formation Kei K. Ito, Koki Watanabe and Daiju Kitagawa * Department of Physiological Chemistry, Graduate School of Pharmaceutical Science, The University of Tokyo, Bunkyo, Tokyo 113-0033, Japan; [email protected] (K.K.I.); [email protected] (K.W.) * Correspondence: [email protected] Received: 11 November 2019; Accepted: 13 March 2020; Published: 20 March 2020 Abstract: Mounting experimental evidence shows that non-coding RNAs (ncRNAs) serve a wide variety of biological functions. Recent studies suggest that a part of ncRNAs are critically important for supporting the structure of subcellular architectures. Here, we summarize the current literature demonstrating the role of ncRNAs and RNA-binding proteins in regulating the assembly of mitotic apparatus, especially focusing on centrosomes, kinetochores, and mitotic spindles. Keywords: ncRNA; centrosome; kinetochore; mitotic spindle 1. Introduction Non-coding RNAs (ncRNAs) are defined as a class of RNA molecules that are transcribed from genomic DNA, but not translated into proteins. They are mainly classified into the following two categories according to their length—small RNA (<200 nt) and long non-coding RNA (lncRNA) (>200 nt). Small RNAs include traditional RNA molecules, such as transfer RNA (tRNA), small nuclear RNA (snRNA), small nucleolar RNA (snoRNA), PIWI-interacting RNA (piRNA), and micro RNA (miRNA), and they have been studied extensively [1]. Research on lncRNA is behind that on small RNA despite that recent transcriptome analysis has revealed that more than 120,000 lncRNAs are generated from the human genome [2–4]. -

CEP63 Deficiency Promotes P53-Dependent Microcephaly and Reveals a Role for the Centrosome in Meiotic Recombination
ARTICLE Received 25 Mar 2015 | Accepted 30 May 2015 | Published 9 Jul 2015 DOI: 10.1038/ncomms8676 CEP63 deficiency promotes p53-dependent microcephaly and reveals a role for the centrosome in meiotic recombination Marko Marjanovic´1,2, Carlos Sa´nchez-Huertas1,*, Berta Terre´1,*, Rocı´oGo´mez3, Jan Frederik Scheel4,w, Sarai Pacheco5,6, Philip A. Knobel1, Ana Martı´nez-Marchal5,6, Suvi Aivio1, Lluı´s Palenzuela1, Uwe Wolfrum4, Peter J. McKinnon7, Jose´ A. Suja3, Ignasi Roig5,6, Vincenzo Costanzo8, Jens Lu¨ders1 & Travis H. Stracker1 CEP63 is a centrosomal protein that facilitates centriole duplication and is regulated by the DNA damage response. Mutations in CEP63 cause Seckel syndrome, a human disease characterized by microcephaly and dwarfism. Here we demonstrate that Cep63-deficient mice recapitulate Seckel syndrome pathology. The attrition of neural progenitor cells involves p53-dependent cell death, and brain size is rescued by the deletion of p53. Cell death is not the result of an aberrant DNA damage response but is triggered by centrosome-based mitotic errors. In addition, Cep63 loss severely impairs meiotic recombination, leading to profound male infertility. Cep63-deficient spermatocytes display numerical and structural centrosome aberrations, chromosome entanglements and defective telomere clustering, suggesting that a reduction in centrosome-mediated chromosome movements underlies recombination failure. Our results provide novel insight into the molecular pathology of microcephaly and establish a role for the centrosome in meiotic recombination. 1 Institute for Research in Biomedicine (IRB Barcelona), Barcelona 08028, Spain. 2 Division of Molecular Medicine, Rud–er Bosˇkovic´ Institute, Zagreb 10000, Croatia. 3 Departamento de Biologı´a, Edificio de Biolo´gicas, Universidad Auto´noma de Madrid, Madrid 28049, Spain. -

PCM1 Is Necessary for Focal Ciliary Integrity and Is a Candidate for Severe Schizophrenia
ARTICLE https://doi.org/10.1038/s41467-020-19637-5 OPEN PCM1 is necessary for focal ciliary integrity and is a candidate for severe schizophrenia Tanner O. Monroe 1,2, Melanie E. Garrett3, Maria Kousi4, Ramona M. Rodriguiz5,6, Sungjin Moon7, Yushi Bai8, Steven C. Brodar8, Karen L. Soldano3, Jeremiah Savage9, Thomas F. Hansen10,11, Donna M. Muzny12,13, Richard A. Gibbs12,13, Lawrence Barak8, Patrick F. Sullivan14,15,16, Allison E. Ashley-Koch 3, ✉ Akira Sawa 17,18,19,20, William C. Wetsel 5,6,8,21, Thomas Werge 10,11,22,23 & Nicholas Katsanis 1,2 1234567890():,; The neuronal primary cilium and centriolar satellites have functions in neurogenesis, but little is known about their roles in the postnatal brain. We show that ablation of pericentriolar material 1 in the mouse leads to progressive ciliary, anatomical, psychomotor, and cognitive abnormalities. RNAseq reveals changes in amine- and G-protein coupled receptor pathways. The physiological relevance of this phenotype is supported by decreased available dopamine D2 receptor (D2R) levels and the failure of antipsychotic drugs to rescue adult behavioral defects. Immunoprecipitations show an association with Pcm1 and D2Rs. Finally, we sequence PCM1 in two human cohorts with severe schizophrenia. Systematic modeling of all discovered rare alleles by zebrafish in vivo complementation reveals an enrichment for pathogenic alleles. Our data emphasize a role for the pericentriolar material in the postnatal brain, with progressive degenerative ciliary and behavioral phenotypes; and they support a contributory role for PCM1 in some individuals diagnosed with schizophrenia. 1 Department of Pediatrics, Northwestern University Feinberg School of Medicine, Chicago, IL 60611, USA. -
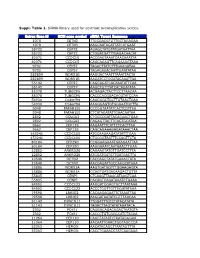
Suppl. Table 1
Suppl. Table 1. SiRNA library used for centriole overduplication screen. Entrez Gene Id NCBI gene symbol siRNA Target Sequence 1070 CETN3 TTGCGACGTGTTGCTAGAGAA 1070 CETN3 AAGCAATAGATTATCATGAAT 55722 CEP72 AGAGCTATGTATGATAATTAA 55722 CEP72 CTGGATGATTTGAGACAACAT 80071 CCDC15 ACCGAGTAAATCAACAAATTA 80071 CCDC15 CAGCAGAGTTCAGAAAGTAAA 9702 CEP57 TAGACTTATCTTTGAAGATAA 9702 CEP57 TAGAGAAACAATTGAATATAA 282809 WDR51B AAGGACTAATTTAAATTACTA 282809 WDR51B AAGATCCTGGATACAAATTAA 55142 CEP27 CAGCAGATCACAAATATTCAA 55142 CEP27 AAGCTGTTTATCACAGATATA 85378 TUBGCP6 ACGAGACTACTTCCTTAACAA 85378 TUBGCP6 CACCCACGGACACGTATCCAA 54930 C14orf94 CAGCGGCTGCTTGTAACTGAA 54930 C14orf94 AAGGGAGTGTGGAAATGCTTA 5048 PAFAH1B1 CCCGGTAATATCACTCGTTAA 5048 PAFAH1B1 CTCATAGATATTGAACAATAA 2802 GOLGA3 CTGGCCGATTACAGAACTGAA 2802 GOLGA3 CAGAGTTACTTCAGTGCATAA 9662 CEP135 AAGAATTTCATTCTCACTTAA 9662 CEP135 CAGCAGAAAGAGATAAACTAA 153241 CCDC100 ATGCAAGAAGATATATTTGAA 153241 CCDC100 CTGCGGTAATTTCCAGTTCTA 80184 CEP290 CCGGAAGAAATGAAGAATTAA 80184 CEP290 AAGGAAATCAATAAACTTGAA 22852 ANKRD26 CAGAAGTATGTTGATCCTTTA 22852 ANKRD26 ATGGATGATGTTGATGACTTA 10540 DCTN2 CACCAGCTATATGAAACTATA 10540 DCTN2 AACGAGATTGCCAAGCATAAA 25886 WDR51A AAGTGATGGTTTGGAAGAGTA 25886 WDR51A CCAGTGATGACAAGACTGTTA 55835 CENPJ CTCAAGTTAAACATAAGTCAA 55835 CENPJ CACAGTCAGATAAATCTGAAA 84902 CCDC123 AAGGATGGAGTGCTTAATAAA 84902 CCDC123 ACCCTGGTTGTTGGATATAAA 79598 LRRIQ2 CACAAGAGAATTCTAAATTAA 79598 LRRIQ2 AAGGATAATATCGTTTAACAA 51143 DYNC1LI1 TTGGATTTGTCTATACATATA 51143 DYNC1LI1 TAGACTTAGTATATAAATACA 2302 FOXJ1 CAGGACAGACAGACTAATGTA -

Pericentrin Polyclonal Antibody Catalog Number:22271-1-AP 1 Publications
For Research Use Only Pericentrin Polyclonal antibody Catalog Number:22271-1-AP 1 Publications www.ptglab.com Catalog Number: GenBank Accession Number: Purification Method: Basic Information 22271-1-AP NM_006031 Antigen affinity purification Size: GeneID (NCBI): Recommended Dilutions: 150UL , Concentration: 800 μg/ml by 5116 IF 1:50-1:500 Nanodrop and 360 μg/ml by Bradford Full Name: method using BSA as the standard; pericentrin Source: Calculated MW: Rabbit 378 kDa Isotype: IgG Applications Tested Applications: Positive Controls: IF,ELISA IF : MDCK cells, Cited Applications: IF Species Specificity: human, Canine Cited Species: human PCNT, also named as KIAA0402, PCNT2, Pericentrin-B and Kendrin, is an integral component of the filamentous Background Information matrix of the centrosome involved in the initial establishment of organized microtubule arrays in both mitosis and meiosis. PCNT plays a role, together with DISC1, in the microtubule network formation. Is an integral component of the pericentriolar material (PCM). PCNT is a marker of centrosome. Defects in PCNT are the cause of microcephalic osteodysplastic primordial dwarfism type 2 (MOPD2). The antibody is specific to PCNT, and it can recognize isoform 1 and 2. Notable Publications Author Pubmed ID Journal Application Won-Jing Wang 26609813 Elife IF Storage: Storage Store at -20°C. Stable for one year after shipment. Storage Buffer: PBS with 0.02% sodium azide and 50% glycerol pH 7.3. Aliquoting is unnecessary for -20ºC storage For technical support and original validation data for this product please contact: This product is exclusively available under Proteintech T: 1 (888) 4PTGLAB (1-888-478-4522) (toll free E: [email protected] Group brand and is not available to purchase from any in USA), or 1(312) 455-8498 (outside USA) W: ptglab.com other manufacturer. -

PCM1 Monoclonal Antibody (CL0206) Catalog Number MA5-24589 Product Data Sheet
Lot Number: 02985 Website: thermofisher.com Customer Service (US): 1 800 955 6288 ext. 1 Technical Support (US): 1 800 955 6288 ext. 441 thermofisher.com/contactus PCM1 Monoclonal Antibody (CL0206) Catalog Number MA5-24589 Product Data Sheet Details Species Reactivity Size 100 µl Tested species reactivity Human Host / Isotype Mouse IgG1 Tested Applications Dilution * Class Monoclonal Immunohistochemistry (IHC) Assay-Dependent Type Antibody Western Blot (WB) 1 µg/mL Clone CL0206 * Suggested working dilutions are given as a guide only. It is recommended that the user titrate the product for use in their own experiment using appropriate negative and positive controls. Immunogen Recombinant protein corresponding to Human PCM1 Conjugate Unconjugated Form Liquid Concentration 0.7mg/ml Purification Protein A Storage Buffer PBS, pH 7.2, with 40% glycerol Contains 0.02% sodium azide Store at 4°C short term. For long Storage Conditions term storage, store at -20°C, avoiding freeze/thaw cycles. Product Specific Information Immunogen sequence: TIYSEVATLI SQNESRPHFL IELFHELQLL NTDYLRQRAL YALQDIVSRH ISESHEKGEN VKSVNSGTWI ASNSELTPSE SLATTDDETF EKNFE Highest antigen sequence identity to the following orthologs: Mouse - 94%, Rat - 95%. Binds to an epitope located within the peptide sequence RQRALYALQD as determined by overlapping synthetic peptides. Background/Target Information PCM-1 is required for centrosome assembly and function. This protein is essential for the correct localization of several centrosomal proteins including CEP250, CETN3, PCNT and NEK2. The protein is required to anchor microtubules to the centrosome. For Research Use Only. Not for use in diagnostic procedures. Not for resale without express authorization. For Research Use Only. Not for use in diagnostic procedures. -

PCM1 Recruits Plk1 to the Pericentriolar Matrix to Promote
Research Article 1355 PCM1 recruits Plk1 to the pericentriolar matrix to promote primary cilia disassembly before mitotic entry Gang Wang1,*, Qiang Chen1,*, Xiaoyan Zhang1, Boyan Zhang1, Xiaolong Zhuo1, Junjun Liu2, Qing Jiang1 and Chuanmao Zhang1,` 1MOE Key Laboratory of Cell Proliferation and Differentiation and State Key Laboratory of Biomembrane and Membrane Biotechnology, College of Life Sciences, Peking University, Beijing 100871, China 2Department of Biological Sciences, California State Polytechnic University, Pomona, CA 91768, USA *These authors contributed equally to this work `Author for correspondence ([email protected]) Accepted 11 December 2012 Journal of Cell Science 126, 1355–1365 ß 2013. Published by The Company of Biologists Ltd doi: 10.1242/jcs.114918 Summary Primary cilia, which emanate from the cell surface, exhibit assembly and disassembly dynamics along the progression of the cell cycle. However, the mechanism that links ciliary dynamics and cell cycle regulation remains elusive. In the present study, we report that Polo- like kinase 1 (Plk1), one of the key cell cycle regulators, which regulate centrosome maturation, bipolar spindle assembly and cytokinesis, acts as a pivotal player that connects ciliary dynamics and cell cycle regulation. We found that the kinase activity of centrosome enriched Plk1 is required for primary cilia disassembly before mitotic entry, wherein Plk1 interacts with and activates histone deacetylase 6 (HDAC6) to promote ciliary deacetylation and resorption. Furthermore, we showed that pericentriolar material 1 (PCM1) acts upstream of Plk1 and recruits the kinase to pericentriolar matrix (PCM) in a dynein-dynactin complex-dependent manner. This process coincides with the primary cilia disassembly dynamics at the onset of mitosis, as depletion of PCM1 by shRNA dramatically disrupted the pericentriolar accumulation of Plk1. -

Modifier Genes in Microcephaly: a Report on WDR62, CEP63, RAD50
G C A T T A C G G C A T genes Article Modifier Genes in Microcephaly: A Report on WDR62, CEP63, RAD50 and PCNT Variants Exacerbating Disease Caused by Biallelic Mutations of ASPM and CENPJ Ehtisham Ul Haq Makhdoom 1,2,3,†, Syeda Seema Waseem 1,4,†, Maria Iqbal 1,2,4, Uzma Abdullah 5 , Ghulam Hussain 3, Maria Asif 1,4, Birgit Budde 1 , Wolfgang Höhne 1, Sigrid Tinschert 6, Saadia Maryam Saadi 2 , Hammad Yousaf 2, Zafar Ali 7, Ambrin Fatima 8, Emrah Kaygusuz 9 , Ayaz Khan 2 , Muhammad Jameel 2, Sheraz Khan 2 , Muhammad Tariq 2 , Iram Anjum 10 , Janine Altmüller 1, Holger Thiele 1, Stefan Höning 4, Shahid Mahmood Baig 2,8,11, Peter Nürnberg 1,12 and Muhammad Sajid Hussain 1,4,12,* 1 Cologne Center for Genomics (CCG), Faculty of Medicine and University Hospital Cologne, University of Cologne, 50931 Cologne, Germany; [email protected] (E.U.H.M.); [email protected] (S.S.W.); [email protected] (M.I.); [email protected] (M.A.); [email protected] (B.B.); [email protected] (W.H.); [email protected] (J.A.); [email protected] (H.T.); [email protected] (P.N.) 2 Human Molecular Genetics Laboratory, Health Biotechnology Division, National Institute for Biotechnology and Genetic Engineering (NIBGE) College, PIEAS, Faisalabad 38000, Pakistan; [email protected] (S.M.S.); [email protected] (H.Y.); [email protected] (A.K.); [email protected] (M.J.); [email protected] (S.K.); [email protected] (M.T.); [email protected] (S.M.B.) 3 Citation: Makhdoom, E.U.H.; Neurochemicalbiology and Genetics Laboratory (NGL), Department of Physiology, Faculty of Life Sciences, Waseem, S.S.; Iqbal, M.; Abdullah, U.; Government College University, Faisalabad 38000, Pakistan; [email protected] 4 Hussain, G.; Asif, M.; Budde, B.; Institute of Biochemistry I, Medical Faculty, University of Cologne, 50931 Cologne, Germany; [email protected] Höhne, W.; Tinschert, S.; Saadi, 5 University Institute of Biochemistry and Biotechnology (UIBB), PMAS-Arid Agriculture University, S.M.; et al. -

C-Nap1 Mutation Affects Centriole Cohesion and Is Associated with a Seckel-Like Syndrome in Cattle
ARTICLE Received 27 Jul 2014 | Accepted 11 Mar 2015 | Published 23 Apr 2015 DOI: 10.1038/ncomms7894 OPEN C-Nap1 mutation affects centriole cohesion and is associated with a Seckel-like syndrome in cattle Sandrine Floriot1, Christine Vesque2,3,4, Sabrina Rodriguez1,5, Florence Bourgain-Guglielmetti2,3,6, Anthi Karaiskou2,3, Mathieu Gautier1,7, Amandine Duchesne1, Sarah Barbey8,Se´bastien Fritz1,9, Alexandre Vasilescu10, Maud Bertaud1, Mohammed Moudjou11, Sophie Halliez11, Vale´rie Cormier-Daire12, Joyce E.L. Hokayem12, Erich A. Nigg13, Luc Manciaux14,w, Raphae¨l Guatteo15,16, Nora Cesbron15,16, Geraldine Toutirais17, Andre´ Eggen1, Sylvie Schneider-Maunoury2,3,4, Didier Boichard1, Joelle Sobczak-The´pot2,3,6 & Laurent Schibler1,9 Caprine-like Generalized Hypoplasia Syndrome (SHGC) is an autosomal-recessive disorder in Montbe´liarde cattle. Affected animals present a wide range of clinical features that include the following: delayed development with low birth weight, hind limb muscular hypoplasia, caprine-like thin head and partial coat depigmentation. Here we show that SHGC is caused by a truncating mutation in the CEP250 gene that encodes the centrosomal protein C-Nap1. This mutation results in centrosome splitting, which neither affects centriole ultrastructure and duplication in dividing cells nor centriole function in cilium assembly and mitotic spindle organization. Loss of C-Nap1-mediated centriole cohesion leads to an altered cell migration phenotype. This discovery extends the range of loci that constitute the spectrum of autosomal primary recessive microcephaly (MCPH) and Seckel-like syndromes. 1 Institut National de la Recherche Agronomique (INRA), Unite´ Mixte de Recherche 1313—Ge´ne´tique Animale et Biologie Inte´grative (UMR1313—GABI), F-78352 Jouy-en-Josas, France.