Differential Expression of Wnt13 Isoforms During Leukemic Cell Differentiation
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Downloaded from the National Database for Autism Research (NDAR)
International Journal of Molecular Sciences Article Phenotypic Subtyping and Re-Analysis of Existing Methylation Data from Autistic Probands in Simplex Families Reveal ASD Subtype-Associated Differentially Methylated Genes and Biological Functions Elizabeth C. Lee y and Valerie W. Hu * Department of Biochemistry and Molecular Medicine, The George Washington University, School of Medicine and Health Sciences, Washington, DC 20037, USA; [email protected] * Correspondence: [email protected]; Tel.: +1-202-994-8431 Current address: W. Harry Feinstone Department of Molecular Microbiology and Immunology, y Johns Hopkins Bloomberg School of Public Health, Baltimore, MD 21205, USA. Received: 25 August 2020; Accepted: 17 September 2020; Published: 19 September 2020 Abstract: Autism spectrum disorder (ASD) describes a group of neurodevelopmental disorders with core deficits in social communication and manifestation of restricted, repetitive, and stereotyped behaviors. Despite the core symptomatology, ASD is extremely heterogeneous with respect to the severity of symptoms and behaviors. This heterogeneity presents an inherent challenge to all large-scale genome-wide omics analyses. In the present study, we address this heterogeneity by stratifying ASD probands from simplex families according to the severity of behavioral scores on the Autism Diagnostic Interview-Revised diagnostic instrument, followed by re-analysis of existing DNA methylation data from individuals in three ASD subphenotypes in comparison to that of their respective unaffected siblings. We demonstrate that subphenotyping of cases enables the identification of over 1.6 times the number of statistically significant differentially methylated regions (DMR) and DMR-associated genes (DAGs) between cases and controls, compared to that identified when all cases are combined. Our analyses also reveal ASD-related neurological functions and comorbidities that are enriched among DAGs in each phenotypic subgroup but not in the combined case group. -

Molecular Genetic Delineation of 2Q37.3 Deletion in Autism and Osteodystrophy: Report of a Case and of New Markers for Deletion Screening by PCR
UC Irvine UC Irvine Previously Published Works Title Molecular genetic delineation of 2q37.3 deletion in autism and osteodystrophy: report of a case and of new markers for deletion screening by PCR. Permalink https://escholarship.org/uc/item/83f0x61r Journal Cytogenetics and cell genetics, 94(1-2) ISSN 0301-0171 Authors Smith, M Escamilla, JR Filipek, P et al. Publication Date 2001 DOI 10.1159/000048775 License https://creativecommons.org/licenses/by/4.0/ 4.0 Peer reviewed eScholarship.org Powered by the California Digital Library University of California Original Article Cytogenet Cell Genet 94:15–22 (2001) Molecular genetic delineation of 2q37.3 deletion in autism and osteodystrophy: report of a case and of new markers for deletion screening by PCR M. Smith, J.R. Escamilla, P. Filipek, M.E. Bocian, C. Modahl, P. Flodman, and M.A. Spence Department of Pediatrics, University of California, Irvine CA (USA) Abstract. We recently studied a patient who meets criteria us to determine the parental origin of the deletion in our for autistic disorder and has a 2q37 deletion. Molecular cyto- patient. DNA from 8–13 unrelated individuals was used to genetic studies were carried out using DNA isolated from 22 determine heterozygosity estimates for these markers. We re- different 2q37 mapped BACs to more precisely define the view four genes deleted in our patient – genes whose known extent of the chromosome deletion. We also analyzed 2q37 functions and sites of expression in the brain and/or bone make mapped polymorphic markers. In addition DNA sequences of them candidates for involvement in autism and/or the osteo- BACs in the deletion region were scanned to identify microsa- dystrophy observed in patients with 2q37.3 deletions. -

Agonists Regulate the Expression of Stemness and Differentiation Genes in Brain Tumour Stem Cells
British Journal of Cancer (2012) 106, 1702–1712 & 2012 Cancer Research UK All rights reserved 0007 – 0920/12 www.bjcancer.com PPARg agonists regulate the expression of stemness and differentiation genes in brain tumour stem cells E Pestereva1, S Kanakasabai1 and JJ Bright*,1,2 1 Neuroscience Research Laboratory, Methodist Research Institute, Indiana University Health, 1800 North Capitol Avenue, Noyes Building E504C, 2 Indianapolis, IN 46202, USA; Department of Medicine, Indiana University School of Medicine, Indianapolis, IN, USA BACKGROUND: Brain tumour stem cells (BTSCs) are a small population of cancer cells that exhibit self-renewal, multi-drug resistance, and recurrence properties. We have shown earlier that peroxisome proliferator-activated receptor gamma (PPARg) agonists inhibit the expansion of BTSCs in T98G and U87MG glioma. In this study, we analysed the influence of PPARg agonists on the expression of stemness and differentiation genes in BTSCs. METHODS: The BTSCs were isolated from T98G and DB29 glioma cells, and cultured in neurobasal medium with epidermal growth factor þ basic fibroblast growth factor. Proliferation was measured by WST-1 (4-[3-(4-iodophenyl)-2-(4-nitrophenyl)-2 H-5- tetrazolio]-1,3-benzene disulphonate) and 3H thymidine uptake assays, and gene expression was analysed by quantitative reverse– transcription PCR and Taqman array. The expression of CD133, SRY box 2, and nanog homeobox (Nanog) was also evaluated by western blotting, immunostaining, and flow cytometry. 12,14 RESULTS: We found that PPARg agonists, ciglitazone and 15-deoxy-D -ProstaglandinJ2, inhibited cell viability and proliferation of þ T98G- and DB29-BTSCs. The PPARg agonists reduced the expansion of CD133 BTSCs and altered the expression of stemness and differentiation genes. -

PDF Download
Review Xatzipsalti Maria et al. Congenital Hypopituitarism: Various Genes, … Horm Metab Res 2018; 00: 00–00 Congenital Hypopituitarism: Various Genes, Various Phenotypes Authors Maria Xatzipsalti1, 2, Antonis Voutetakis1, Lela Stamoyannou2, George P. Chrousos1, Christina Kanaka-Gantenbein1 Affiliations ABSTRacT 1 Division of Endocrinology, Diabetes and Metabolism, The ontogenesis and development of the pituitary gland is a First Department of Pediatrics, Medical School, National highly complex process that depends on a cascade of transcrip- and Kapodistrian University of Athens, “Aghia Sofia” tion factors and signaling molecules. Spontaneous mutations Children's Hospital, Athens, Greece and transgenic murine models have demonstrated a role for 2 First Department of Pediatrics, “Aglaia Kyriakou” many of these factors, including HESX1, PROP1, PIT1, LHX3, Children's Hospital, Athens, Greece LHX4, SOX2, SOX3, OTX2, PAX6, FGFR1, SHH, GLI2, and FGF8 in the etiology of congenital hypopituitarism. Genetic muta- Key words tions in any of these factors can lead to congenital hypopitui- pituitary, combined pituitary hormone deficiency, congenital tarism, which is characterized by the deficiency in one or more hypopituitarism, transcription factors, syndromic hypopitui- pituitary hormones. The phenotype can be highly variable, tarism, non-syndromic hypopituitarism consisting of isolated hypopituitarism or more complex disor- ders. The same phenotype can be attributed to different gene received 27.03.2018 mutations; while a given gene mutation can -

FOS Rescues Neuronal Differentiation of Sox2-Deleted Neural Stem Cells by Genome-Wide Regulation of Common SOX2 and AP1(FOS-JUN) Target Genes
cells Article FOS Rescues Neuronal Differentiation of Sox2-Deleted Neural Stem Cells by Genome-Wide Regulation of Common SOX2 and AP1(FOS-JUN) Target Genes Miriam Pagin 1,† , Mattias Pernebrink 2,3,†, Mattia Pitasi 1, Federica Malighetti 1, Chew-Yee Ngan 4, Sergio Ottolenghi 1, Giulio Pavesi 5, Claudio Cantù 2,3,* and Silvia K. Nicolis 1,* 1 Department of Biotechnology and Biosciences, University of Milano-Bicocca, Piazza della Scienza 2, 20126 Milano, Italy; [email protected] (M.P.); [email protected] (M.P.); [email protected] (F.M.); [email protected] (S.O.) 2 Wallenberg Centre for Molecular Medicine, Linköping University, SE-581 83 Linköping, Sweden; [email protected] 3 Department of Biomedical and Clinical Sciences, Division of Molecular Medicine and Virology, Faculty of Medicine and Health Sciences, Linköping University, SE-581 83 Linköping, Sweden 4 The Jackson Laboratory for Genomic Medicine, Farmington, CT 06032, USA; [email protected] 5 Department of Biosciences, University of Milano, Via Celoria 26, 20134 Milano, Italy; [email protected] * Correspondence: [email protected] (C.C.); [email protected] (S.K.N.) † Joint first authors. Abstract: The transcription factor SOX2 is important for brain development and for neural stem Citation: Pagin, M.; Pernebrink, M.; cells (NSC) maintenance. Sox2-deleted (Sox2-del) NSC from neonatal mouse brain are lost after Pitasi, M.; Malighetti, F.; Ngan, C.-Y.; few passages in culture. Two highly expressed genes, Fos and Socs3, are strongly downregulated in Ottolenghi, S.; Pavesi, G.; Cantù, C.; Sox2-del NSC; we previously showed that Fos or Socs3 overexpression by lentiviral transduction fully Nicolis, S.K. -
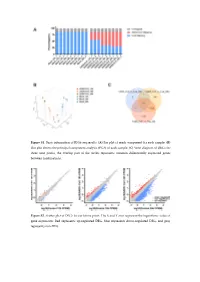
Figure S1. Basic Information of RNA-Seq Results. (A) Bar Plot of Reads Component for Each Sample
Figure S1. Basic information of RNA-seq results. (A) Bar plot of reads component for each sample. (B) Dot plot shows the principal component analysis (PCA) of each sample. (C) Venn diagram of DEGs for three time points, the overlap part of the circles represents common differentially expressed genes between combinations. Figure S2. Scatter plot of DEGs for each time point. The X and Y axes represent the logarithmic value of gene expression. Red represents up-regulated DEG, blue represents down-regulated DEG, and gray represents non-DEG. Table S1. Primers used for quantitative real-time PCR analysis of DEGs. Gene Primer Sequence Forward 5’-CTACGAGTGGATGGTCAAGAGC-3’ FOXO1 Reverse 5’-CCAGTTCCTTCATTCTGCACACG-3’ Forward 5’-GACGTCCGGCATCAGAGAAA-3’ IRS2 Reverse 5’-TCCACGGCTAATCGTCACAG-3’ Forward 5’-CACAACCAGGACCTCACACC-3’ IRS1 Reverse 5’-CTTGGCACGATAGAGAGCGT-3’ Forward 5’-AGGATACCACTCCCAACAGACCT-3’ IL6 Reverse 5’-CAAGTGCATCATCGTTGTTCATAC-3’ Forward 5’-TCACGTTGTACGCAGCTACC-3’ CCL5 Reverse 5’-CAGTCCTCTTACAGCCTTTGG-3’ Forward 5’-CTGTGCAGCCGCAGTGCCTACC-3’ BMP7 Reverse 5’-ATCCCTCCCCACCCCACCATCT-3’ Forward 5’-CTCTCCCCCTCGACTTCTGA-3’ BCL2 Reverse 5’-AGTCACGCGGAACACTTGAT-3’ Forward 5’-CTGTCGAACACAGTGGTACCTG-3’ FGF7 Reverse 5’-CCAACTGCCACTGTCCTGATTTC-3’ Forward 5’-GGGAGCCAAAAGGGTCATCA-3’ GAPDH Reverse 5’-CGTGGACTGTGGTCATGAGT-3’ Supplementary material: Differentially expressed genes log2(SADS-CoV_12h/ Qvalue (SADS-CoV _12h/ Gene Symbol Control_12h) Control_12h) PTGER4 -1.03693 6.79E-04 TMEM72 -3.08132 3.66E-04 IFIT2 -1.02918 2.11E-07 FRAT2 -1.09282 4.66E-05 -

Mouse Thalamic Differentiation: Gli-Dependent Pattern and Gli-Independent Prepattern
ORIGINAL RESEARCH ARTICLE published: 22 February 2012 doi: 10.3389/fnins.2012.00027 Mouse thalamic differentiation: Gli-dependent pattern and Gli-independent prepattern Roberta Haddad-Tóvolli 1, Michael Heide 1, Xunlei Zhou 1, Sandra Blaess 2 and Gonzalo Alvarez-Bolado1* 1 Department of Neuroanatomy, Institute of Anatomy and Cell Biology, University of Heidelberg, Heidelberg, Germany 2 Institute of Reconstructive Neurobiology, Life and Brain Center, University of Bonn, Bonn, Germany Edited by: Sonic hedgehog (Shh) signaling is essential for thalamic development.The Gli transcription Steffen Scholpp, Karlsruhe Institute of factors act downstream of Shh – while Gli2 is the major activator (GliA), Gli3 acts primarily Technology, Germany as a repressor (GliR). The thalamus is remarkable among dorsal structures because of its Reviewed by: Tomomi Shimogori, RIKEN Brain proximity to the mid-diencephalic organizer, a unique dorsal Shh source. This lends com- Science Institute, Japan plexity to the interactions between Shh, Gli2, and Gli3, suggesting the presence of a dorsal David J. Price, University of Gli activator which elsewhere is found only ventrally, and making the dissection of thala- Edinburgh, UK mic Gli functions particularly interesting. A current model based on mutant phenotypes in *Correspondence: telencephalon and midbrain postulates a degree of reciprocal antagonism of Shh and Gli3 Gonzalo Alvarez-Bolado, Department of Neuroanatomy, Institute of in dorsal brain regions. To approach the role of Gli factors in thalamic specification we first Anatomy and Cell Biology, University analyzed mice deficient in Gli2 or Gli3.InGli2 mutants, the thalamus is small and poorly of Heidelberg, Im Neuenheimer Feld differentiated with the exception of the medial and intralaminar nuclei which, in contrast, 307, 69120 Heidelberg, Germany. -

Zeb2 Regulates Cell Fate at the Exit from Epiblast State In
View metadata, citation and similar papers at core.ac.uk EMBRYONIC STEM CELLS/INDUCEDbrought to you by CORE PLURIPOTENT STEMprovidedC byELLS Ghent University Academic Bibliography Zeb2 Regulates Cell Fate at the Exit from Epiblast State in Mouse Embryonic Stem Cells aDepartment of Development and Regeneration, KU a a,b c,d,e,f a b Leuven, Leuven 3000, AGATA STRYJEWSKA, RUBEN DRIES, TIM PIETERS, GRIET VERSTAPPEN, ANDREA CONIDI, a a a b,g Belgium; bDepartment of Cell KATHLEEN CODDENS, ANNICK FRANCIS, LIEVE UMANS, WILFRED F. J. VAN IJCKEN, g d,e h b c,d,e,i Biology, Center for Biomics, GEERT BERX, LEO A. VAN GRUNSVEN, FRANK G. GROSVELD, STEVEN GOOSSENS, c,d,i a,b Erasmus University Medical JODY J. HAIGH, DANNY HUYLEBROECK Center, Rotterdam 3015 CN, c The Netherlands; VIB Key Words. Cell differentiation • DNA-methylation • Embryonic stem cells • Pluripotent stem cells Inflammation Research • Repressors • RNA-sequencing • Transcription factors • Transcriptom Center (IRC), Unit Vascular Cell Biology, dDepartment of Biomedical Molecular Biology, eVIB-IRC, Unit ABSTRACT Molecular and Cellular In human embryonic stem cells (ESCs) the transcription factor Zeb2 regulates neuroectoderm ver- Oncology, Ghent University, sus mesendoderm formation, but it is unclear how Zeb2 affects the global transcriptional regulato- f Ghent 9052, Belgium; Center ry network in these cell-fate decisions. We generated Zeb2 knockout (KO) mouse ESCs, subjected for Medical Genetics, Ghent them as embryoid bodies (EBs) to neural and general differentiation and carried out temporal University Hospital, Ghent h RNA-sequencing (RNA-seq) and reduced representation bisulfite sequencing (RRBS) analysis in neu- 9000, Belgium; Department ral differentiation. -

Molecular Regulation in Dopaminergic Neuron Development
International Journal of Molecular Sciences Review Molecular Regulation in Dopaminergic Neuron Development. Cues to Unveil Molecular Pathogenesis and Pharmacological Targets of Neurodegeneration Floriana Volpicelli 1 , Carla Perrone-Capano 1,2 , Gian Carlo Bellenchi 2,3, Luca Colucci-D’Amato 4,* and Umberto di Porzio 2 1 Department of Pharmacy, University of Naples Federico II, 80131 Naples, Italy; fl[email protected] (F.V.); [email protected] (C.P.C.) 2 Institute of Genetics and Biophysics “Adriano Buzzati Traverso”, CNR, 80131 Rome, Italy; [email protected] (G.C.B.); [email protected] (U.d.P.) 3 Department of Systems Medicine, University of Rome Tor Vergata, 00133 Rome, Italy 4 Department of Environmental, Biological and Pharmaceutical Sciences and Technologies, University of Campania “Luigi Vanvitelli”, 81100 Caserta, Italy * Correspondence: [email protected]; Tel.: +39-0823-274577 Received: 28 April 2020; Accepted: 1 June 2020; Published: 3 June 2020 Abstract: The relatively few dopaminergic neurons in the mammalian brain are mostly located in the midbrain and regulate many important neural functions, including motor integration, cognition, emotive behaviors and reward. Therefore, alteration of their function or degeneration leads to severe neurological and neuropsychiatric diseases. Unraveling the mechanisms of midbrain dopaminergic (mDA) phenotype induction and maturation and elucidating the role of the gene network involved in the development and maintenance of these neurons is of pivotal importance to rescue or substitute these cells in order to restore dopaminergic functions. Recently, in addition to morphogens and transcription factors, microRNAs have been identified as critical players to confer mDA identity. The elucidation of the gene network involved in mDA neuron development and function will be crucial to identify early changes of mDA neurons that occur in pre-symptomatic pathological conditions, such as Parkinson’s disease. -

Zeb2 Regulates Cell Fate at the Exit from Epiblast State in Mouse
EMBRYONIC STEM CELLS/INDUCED PLURIPOTENT STEM CELLS Zeb2 Regulates Cell Fate at the Exit from Epiblast State in Mouse Embryonic Stem Cells aDepartment of Development and Regeneration, KU a a,b c,d,e,f a b Leuven, Leuven 3000, AGATA STRYJEWSKA, RUBEN DRIES, TIM PIETERS, GRIET VERSTAPPEN, ANDREA CONIDI, a a a b,g Belgium; bDepartment of Cell KATHLEEN CODDENS, ANNICK FRANCIS, LIEVE UMANS, WILFRED F. J. VAN IJCKEN, g d,e h b c,d,e,i Biology, Center for Biomics, GEERT BERX, LEO A. VAN GRUNSVEN, FRANK G. GROSVELD, STEVEN GOOSSENS, c,d,i a,b Erasmus University Medical JODY J. HAIGH, DANNY HUYLEBROECK Center, Rotterdam 3015 CN, c The Netherlands; VIB Key Words. Cell differentiation • DNA-methylation • Embryonic stem cells • Pluripotent stem cells Inflammation Research • Repressors • RNA-sequencing • Transcription factors • Transcriptom Center (IRC), Unit Vascular Cell Biology, dDepartment of Biomedical Molecular Biology, eVIB-IRC, Unit ABSTRACT Molecular and Cellular In human embryonic stem cells (ESCs) the transcription factor Zeb2 regulates neuroectoderm ver- Oncology, Ghent University, sus mesendoderm formation, but it is unclear how Zeb2 affects the global transcriptional regulato- f Ghent 9052, Belgium; Center ry network in these cell-fate decisions. We generated Zeb2 knockout (KO) mouse ESCs, subjected for Medical Genetics, Ghent them as embryoid bodies (EBs) to neural and general differentiation and carried out temporal University Hospital, Ghent h RNA-sequencing (RNA-seq) and reduced representation bisulfite sequencing (RRBS) analysis in neu- 9000, Belgium; Department ral differentiation. This shows that Zeb2 acts preferentially as a transcriptional repressor associated of Cell Biology, Liver Cell with developmental progression and that Zeb2 KO ESCs can exit from their na€ıve state. -

Highly Dynamic Chromatin Interactions Drive Neurogenesis Through Gene
bioRxiv preprint doi: https://doi.org/10.1101/303842; this version posted April 18, 2018. The copyright holder for this preprint (which was not certified by peer review) is the author/funder, who has granted bioRxiv a license to display the preprint in perpetuity. It is made available under aCC-BY-NC-ND 4.0 International license. Highly dynamic chromatin interactions drive neurogenesis through gene regulatory networks Valeriya Malysheva1,2,4, Marco Antonio Mendoza-Parra1, Matthias Blum1,3 and Hinrich Gronemeyer1,4 1 Institut de Génétique et de Biologie Moléculaire et Cellulaire (IGBMC), Equipe Labellisée Ligue Contre le Cancer, Centre National de la Recherche Scientifique UMR 7104, Institut National de la Santé et de la Recherche Médicale U964, University of Strasbourg, Illkirch, France. 2 Present address: The Babraham Institute, Babraham Research Campus, Cambridge, UK 3 Present address: European Molecular Biology Laboratory, European Bioinformatics Institute (EMBL-EBI), Wellcome Trust Genome Campus, Hinxton, Cambridge, UK 4 Corresponding authors: Valeriya Malysheva Email: [email protected] Phone: +(44) 1223 49 6505 Hinrich Gronemeyer Email: [email protected] Phone: +(33) 3 88 65 34 73 bioRxiv preprint doi: https://doi.org/10.1101/303842; this version posted April 18, 2018. The copyright holder for this preprint (which was not certified by peer review) is the author/funder, who has granted bioRxiv a license to display the preprint in perpetuity. It is made available under aCC-BY-NC-ND 4.0 International license. Cell fate transitions are fundamental processes in the ontogeny of multicellular organisms1 and aberrations can generate pathologies2. Cell fate acquisition is a highly complex phenomenon which involves a plethora of intrinsic and extrinsic instructive signals that direct the lineage progression of pluripotent cells. -

Predict AID Targeting in Non-Ig Genes Multiple Transcription Factor
Downloaded from http://www.jimmunol.org/ by guest on September 26, 2021 is online at: average * The Journal of Immunology published online 20 March 2013 from submission to initial decision 4 weeks from acceptance to publication Multiple Transcription Factor Binding Sites Predict AID Targeting in Non-Ig Genes Jamie L. Duke, Man Liu, Gur Yaari, Ashraf M. Khalil, Mary M. Tomayko, Mark J. Shlomchik, David G. Schatz and Steven H. Kleinstein J Immunol http://www.jimmunol.org/content/early/2013/03/20/jimmun ol.1202547 Submit online. Every submission reviewed by practicing scientists ? is published twice each month by http://jimmunol.org/subscription Submit copyright permission requests at: http://www.aai.org/About/Publications/JI/copyright.html Receive free email-alerts when new articles cite this article. Sign up at: http://jimmunol.org/alerts http://www.jimmunol.org/content/suppl/2013/03/20/jimmunol.120254 7.DC1 Information about subscribing to The JI No Triage! Fast Publication! Rapid Reviews! 30 days* Why • • • Material Permissions Email Alerts Subscription Supplementary The Journal of Immunology The American Association of Immunologists, Inc., 1451 Rockville Pike, Suite 650, Rockville, MD 20852 Copyright © 2013 by The American Association of Immunologists, Inc. All rights reserved. Print ISSN: 0022-1767 Online ISSN: 1550-6606. This information is current as of September 26, 2021. Published March 20, 2013, doi:10.4049/jimmunol.1202547 The Journal of Immunology Multiple Transcription Factor Binding Sites Predict AID Targeting in Non-Ig Genes Jamie L. Duke,* Man Liu,†,1 Gur Yaari,‡ Ashraf M. Khalil,x Mary M. Tomayko,{ Mark J. Shlomchik,†,x David G.