Mitochondrial Involvement and Erythronic Acid As a Novel Biomarker in Transaldolase Deficiency Udo F.H
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Supplementary Table S4. FGA Co-Expressed Gene List in LUAD
Supplementary Table S4. FGA co-expressed gene list in LUAD tumors Symbol R Locus Description FGG 0.919 4q28 fibrinogen gamma chain FGL1 0.635 8p22 fibrinogen-like 1 SLC7A2 0.536 8p22 solute carrier family 7 (cationic amino acid transporter, y+ system), member 2 DUSP4 0.521 8p12-p11 dual specificity phosphatase 4 HAL 0.51 12q22-q24.1histidine ammonia-lyase PDE4D 0.499 5q12 phosphodiesterase 4D, cAMP-specific FURIN 0.497 15q26.1 furin (paired basic amino acid cleaving enzyme) CPS1 0.49 2q35 carbamoyl-phosphate synthase 1, mitochondrial TESC 0.478 12q24.22 tescalcin INHA 0.465 2q35 inhibin, alpha S100P 0.461 4p16 S100 calcium binding protein P VPS37A 0.447 8p22 vacuolar protein sorting 37 homolog A (S. cerevisiae) SLC16A14 0.447 2q36.3 solute carrier family 16, member 14 PPARGC1A 0.443 4p15.1 peroxisome proliferator-activated receptor gamma, coactivator 1 alpha SIK1 0.435 21q22.3 salt-inducible kinase 1 IRS2 0.434 13q34 insulin receptor substrate 2 RND1 0.433 12q12 Rho family GTPase 1 HGD 0.433 3q13.33 homogentisate 1,2-dioxygenase PTP4A1 0.432 6q12 protein tyrosine phosphatase type IVA, member 1 C8orf4 0.428 8p11.2 chromosome 8 open reading frame 4 DDC 0.427 7p12.2 dopa decarboxylase (aromatic L-amino acid decarboxylase) TACC2 0.427 10q26 transforming, acidic coiled-coil containing protein 2 MUC13 0.422 3q21.2 mucin 13, cell surface associated C5 0.412 9q33-q34 complement component 5 NR4A2 0.412 2q22-q23 nuclear receptor subfamily 4, group A, member 2 EYS 0.411 6q12 eyes shut homolog (Drosophila) GPX2 0.406 14q24.1 glutathione peroxidase -

Table S1. Disease Classification and Disease-Reaction Association
Table S1. Disease classification and disease-reaction association Disorder class Associated reactions cross Disease Ref[Goh check et al. -

Protein Modification and Catabolic Fates of Lipid
PROTEIN MODIFICATION AND CATABOLIC FATES OF LIPID PEROXIDATION PRODUCTS by CHUAN SHI Submitted in partial fulfillment of the requirements for the Degree of Doctor of Philosophy Dissertation Advisor: Gregory P. Tochtrop, Ph.D. Department of Chemistry CASE WESTERN RESERVE UNIVERSITY January 2017 CASE WESTERN RESERVE UNIVERSITY SCHOOL OF GRADUATE STUDIES We hereby approve the dissertation of ______________________________________________________Chuan Shi candidate for the Doctor of Philosophy degree *. Rajesh Viswanathan (signed)_______________________________________________ (chair of the committee) Anthony Pearson ________________________________________________ Michael Zagorski ________________________________________________ Henri Brunengraber ________________________________________________ Gregory Tochtrop ________________________________________________ ________________________________________________ (date) _______________________Dec. 8, 2016 *We also certify that written approval has been obtained for any proprietary material contained therein. This thesis is dedicated to my parents in the deepest appreciation and gratitude for their unconditional love, endless support and continuous encouragement throughout every step in my life TABLE OF CONTENTS Table of Contents ................................................................................................................. i List of Figures ......................................................................................................................v List of Schemes ................................................................................................................ -

Inborn Defects in the Antioxidant Systems of Human Red Blood Cells
Free Radical Biology and Medicine 67 (2014) 377–386 Contents lists available at ScienceDirect Free Radical Biology and Medicine journal homepage: www.elsevier.com/locate/freeradbiomed Review Article Inborn defects in the antioxidant systems of human red blood cells Rob van Zwieten a,n, Arthur J. Verhoeven b, Dirk Roos a a Laboratory of Red Blood Cell Diagnostics, Department of Blood Cell Research, Sanquin Blood Supply Organization, 1066 CX Amsterdam, The Netherlands b Department of Medical Biochemistry, Academic Medical Center, University of Amsterdam, Amsterdam, The Netherlands article info abstract Article history: Red blood cells (RBCs) contain large amounts of iron and operate in highly oxygenated tissues. As a result, Received 16 January 2013 these cells encounter a continuous oxidative stress. Protective mechanisms against oxidation include Received in revised form prevention of formation of reactive oxygen species (ROS), scavenging of various forms of ROS, and repair 20 November 2013 of oxidized cellular contents. In general, a partial defect in any of these systems can harm RBCs and Accepted 22 November 2013 promote senescence, but is without chronic hemolytic complaints. In this review we summarize the Available online 6 December 2013 often rare inborn defects that interfere with the various protective mechanisms present in RBCs. NADPH Keywords: is the main source of reduction equivalents in RBCs, used by most of the protective systems. When Red blood cells NADPH becomes limiting, red cells are prone to being damaged. In many of the severe RBC enzyme Erythrocytes deficiencies, a lack of protective enzyme activity is frustrating erythropoiesis or is not restricted to RBCs. Hemolytic anemia Common hereditary RBC disorders, such as thalassemia, sickle-cell trait, and unstable hemoglobins, give G6PD deficiency Favism rise to increased oxidative stress caused by free heme and iron generated from hemoglobin. -

Pentose Phosphate Pathway in Health and Disease: from Metabolic
UNIVERSIDADE DE LISBOA FACULDADE DE FARMÁCIA DEPARTAMENTO DE BIOQUÍMICA PENTOSE PHOSPHATE PATHWAY IN HEALTH AND DISEASE: FROM METABOLIC DYSFUNCTION TO BIOMARKERS Rúben José Jesus Faustino Ramos Orientador: Professora Doutora Maria Isabel Ginestal Tavares de Almeida Mestrado em Análises Clínicas 2013 Pentose Phosphate Pathway in health and disease: From metabolic dysfunction to biomarkers . Via das Pentoses Fosfato na saúde e na doença: Da disfunção metabólica aos biomarcadores Dissertação apresentada à Faculdade de Farmácia da Universidade de Lisboa para obtenção do grau de Mestre em Análises Clínicas Rúben José Jesus Faustino Ramos Lisboa 2013 Orientador: Professora Doutora Maria Isabel Ginestal Tavares de Almeida The studies presented in this thesis were performed at the Metabolism and Genetics group, iMed.UL (Research Institute for Medicines and Pharmaceutical Sciences), Faculdade de Farmácia da Universidade de Lisboa, Portugal, under the supervision of Prof. Maria Isabel Ginestal Tavares de Almeida, and in collaboration with the Department of Clinical Chemistry, VU University Medical Center, Amsterdam, The Netherlands, Dr. Mirjam Wamelink. De acordo com o disposto no ponto 1 do artigo nº 41 do Regulamento de Estudos Pós- Graduados da Universidade de Lisboa, deliberação nº 93/2006, publicada em Diário da Republica – II série nº 153 – de 5 julho de 2003, o autor desta dissertação declara que participou na conceção e execução do trabalho experimental, interpretação dos resultados obtidos e redação dos manuscritos. Para os meus pais e -

Pentose Phosphate Pathway Biochemistry, Metabolism and Inherited Defects
Pentose phosphate pathway biochemistry, metabolism and inherited defects Amsterdam 2008 Mirjam M.C. Wamelink The research described in this thesis was carried out at the Department of Clinical Chemistry, Metabolic Unit, VU University Medical Center, Amsterdam, The Netherlands. The publication of this thesis was financially supported by: Department of Clinical Chemistry, VU University Medical Center Amsterdam E.C. Noyons Stichting ter bevordering van de Klinische Chemie in Nederland J.E. Jurriaanse Stichting te Rotterdam Printed by: Printpartners Ipskamp BV, Enschede ISBN: 978-90-9023415-1 Cover: Representation of a pathway of sugar Copyright Mirjam Wamelink, Amsterdam, The Netherlands, 2008 2 VRIJE UNIVERSITEIT Pentose phosphate pathway biochemistry, metabolism and inherited defects ACADEMISCH PROEFSCHRIFT ter verkrijging van de graad Doctor aan de Vrije Universiteit Amsterdam, op gezag van de rector magnificus prof.dr. L.M. Bouter, in het openbaar te verdedigen ten overstaan van de promotiecommissie van de faculteit der Geneeskunde op donderdag 11 december 2008 om 13.45 uur in de aula van de universiteit, De Boelelaan 1105 door Mirjam Maria Catharina Wamelink geboren te Alkmaar 3 promotor: prof.dr.ir. C.A.J.M. Jakobs copromotor: dr. E.A. Struijs 4 Abbreviations 6PGD 6-phosphogluconate dehydrogenase ADP adenosine diphosphate ATP adenosine triphosphate CSF cerebrospinal fluid DHAP dihydroxyacetone phosphate G6PD glucose-6-phosphate dehydrogenase GA glyceraldehyde GAPDH glyceraldehyde-3-phosphate dehydrogenase GSG oxidized glutathione -

Diagnose a Broad Range of Metabolic Disorders with a Single Test, Global
PEDIATRIC Assessing or diagnosing a metabolic disorder commonly requires several tests. Global Metabolomic Assisted Pathway Screen, commonly known as Global MAPS, is a unifying test GLOBAL MAPS™ for analyzing hundreds of metabolites to identify changes Global Metabolomic or irregularities in biochemical pathways. Let Global MAPS Assisted Pathway Screen guide you to an answer. Diagnose a broad range of metabolic disorders with a single test, Global MAPS Global MAPS is a large scale, semi-quantitative metabolomic profiling screen that analyzes disruptions in both individual analytes and pathways related to biochemical abnormalities. Using state-of-the-art technologies, Global Metabolomic Assisted Pathway Screen (Global MAPS) provides small molecule metabolic profiling to identify >700 metabolites in human plasma, urine, or cerebrospinal fluid. Global MAPS identifies inborn errors of metabolism (IEMs) that would ordinarily require many different tests. This test defines biochemical pathway errors not currently detected by routine clinical or genetic testing. IEMs are inherited metabolic disorders that prevent the body from converting one chemical compound to another or from transporting a compound in or out of a cell. NORMAL PROCESS METABOLIC ERROR These processes are necessary for essentially all bodily functions. Most IEMs are caused by defects in the enzymes that help process nutrients, which result in an accumulation of toxic substances or a deficiency of substances needed for normal body function. Making a swift, accurate diagnosis -

1 SUPPLEMENTAL DATA Figure S1. Poly I:C Induces IFN-Β Expression
SUPPLEMENTAL DATA Figure S1. Poly I:C induces IFN-β expression and signaling. Fibroblasts were incubated in media with or without Poly I:C for 24 h. RNA was isolated and processed for microarray analysis. Genes showing >2-fold up- or down-regulation compared to control fibroblasts were analyzed using Ingenuity Pathway Analysis Software (Red color, up-regulation; Green color, down-regulation). The transcripts with known gene identifiers (HUGO gene symbols) were entered into the Ingenuity Pathways Knowledge Base IPA 4.0. Each gene identifier mapped in the Ingenuity Pathways Knowledge Base was termed as a focus gene, which was overlaid into a global molecular network established from the information in the Ingenuity Pathways Knowledge Base. Each network contained a maximum of 35 focus genes. 1 Figure S2. The overlap of genes regulated by Poly I:C and by IFN. Bioinformatics analysis was conducted to generate a list of 2003 genes showing >2 fold up or down- regulation in fibroblasts treated with Poly I:C for 24 h. The overlap of this gene set with the 117 skin gene IFN Core Signature comprised of datasets of skin cells stimulated by IFN (Wong et al, 2012) was generated using Microsoft Excel. 2 Symbol Description polyIC 24h IFN 24h CXCL10 chemokine (C-X-C motif) ligand 10 129 7.14 CCL5 chemokine (C-C motif) ligand 5 118 1.12 CCL5 chemokine (C-C motif) ligand 5 115 1.01 OASL 2'-5'-oligoadenylate synthetase-like 83.3 9.52 CCL8 chemokine (C-C motif) ligand 8 78.5 3.25 IDO1 indoleamine 2,3-dioxygenase 1 76.3 3.5 IFI27 interferon, alpha-inducible -
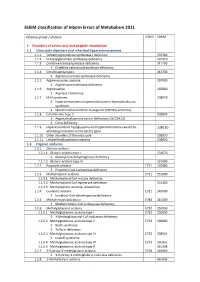
SSIEM Classification of Inborn Errors of Metabolism 2011
SSIEM classification of Inborn Errors of Metabolism 2011 Disease group / disease ICD10 OMIM 1. Disorders of amino acid and peptide metabolism 1.1. Urea cycle disorders and inherited hyperammonaemias 1.1.1. Carbamoylphosphate synthetase I deficiency 237300 1.1.2. N-Acetylglutamate synthetase deficiency 237310 1.1.3. Ornithine transcarbamylase deficiency 311250 S Ornithine carbamoyltransferase deficiency 1.1.4. Citrullinaemia type1 215700 S Argininosuccinate synthetase deficiency 1.1.5. Argininosuccinic aciduria 207900 S Argininosuccinate lyase deficiency 1.1.6. Argininaemia 207800 S Arginase I deficiency 1.1.7. HHH syndrome 238970 S Hyperammonaemia-hyperornithinaemia-homocitrullinuria syndrome S Mitochondrial ornithine transporter (ORNT1) deficiency 1.1.8. Citrullinemia Type 2 603859 S Aspartate glutamate carrier deficiency ( SLC25A13) S Citrin deficiency 1.1.9. Hyperinsulinemic hypoglycemia and hyperammonemia caused by 138130 activating mutations in the GLUD1 gene 1.1.10. Other disorders of the urea cycle 238970 1.1.11. Unspecified hyperammonaemia 238970 1.2. Organic acidurias 1.2.1. Glutaric aciduria 1.2.1.1. Glutaric aciduria type I 231670 S Glutaryl-CoA dehydrogenase deficiency 1.2.1.2. Glutaric aciduria type III 231690 1.2.2. Propionic aciduria E711 232000 S Propionyl-CoA-Carboxylase deficiency 1.2.3. Methylmalonic aciduria E711 251000 1.2.3.1. Methylmalonyl-CoA mutase deficiency 1.2.3.2. Methylmalonyl-CoA epimerase deficiency 251120 1.2.3.3. Methylmalonic aciduria, unspecified 1.2.4. Isovaleric aciduria E711 243500 S Isovaleryl-CoA dehydrogenase deficiency 1.2.5. Methylcrotonylglycinuria E744 210200 S Methylcrotonyl-CoA carboxylase deficiency 1.2.6. Methylglutaconic aciduria E712 250950 1.2.6.1. Methylglutaconic aciduria type I E712 250950 S 3-Methylglutaconyl-CoA hydratase deficiency 1.2.6.2. -

12) United States Patent (10
US007635572B2 (12) UnitedO States Patent (10) Patent No.: US 7,635,572 B2 Zhou et al. (45) Date of Patent: Dec. 22, 2009 (54) METHODS FOR CONDUCTING ASSAYS FOR 5,506,121 A 4/1996 Skerra et al. ENZYME ACTIVITY ON PROTEIN 5,510,270 A 4/1996 Fodor et al. MICROARRAYS 5,512,492 A 4/1996 Herron et al. 5,516,635 A 5/1996 Ekins et al. (75) Inventors: Fang X. Zhou, New Haven, CT (US); 5,532,128 A 7/1996 Eggers Barry Schweitzer, Cheshire, CT (US) 5,538,897 A 7/1996 Yates, III et al. s s 5,541,070 A 7/1996 Kauvar (73) Assignee: Life Technologies Corporation, .. S.E. al Carlsbad, CA (US) 5,585,069 A 12/1996 Zanzucchi et al. 5,585,639 A 12/1996 Dorsel et al. (*) Notice: Subject to any disclaimer, the term of this 5,593,838 A 1/1997 Zanzucchi et al. patent is extended or adjusted under 35 5,605,662 A 2f1997 Heller et al. U.S.C. 154(b) by 0 days. 5,620,850 A 4/1997 Bamdad et al. 5,624,711 A 4/1997 Sundberg et al. (21) Appl. No.: 10/865,431 5,627,369 A 5/1997 Vestal et al. 5,629,213 A 5/1997 Kornguth et al. (22) Filed: Jun. 9, 2004 (Continued) (65) Prior Publication Data FOREIGN PATENT DOCUMENTS US 2005/O118665 A1 Jun. 2, 2005 EP 596421 10, 1993 EP 0619321 12/1994 (51) Int. Cl. EP O664452 7, 1995 CI2O 1/50 (2006.01) EP O818467 1, 1998 (52) U.S. -

Dema and Faust Et Al., Suppl. Material 2020.02.03
Supplementary Materials Cyclin-dependent kinase 18 controls trafficking of aquaporin-2 and its abundance through ubiquitin ligase STUB1, which functions as an AKAP Dema Alessandro1,2¶, Dörte Faust1¶, Katina Lazarow3, Marc Wippich3, Martin Neuenschwander3, Kerstin Zühlke1, Andrea Geelhaar1, Tamara Pallien1, Eileen Hallscheidt1, Jenny Eichhorst3, Burkhard Wiesner3, Hana Černecká1, Oliver Popp1, Philipp Mertins1, Gunnar Dittmar1, Jens Peter von Kries3, Enno Klussmann1,4* ¶These authors contributed equally to this work 1Max Delbrück Center for Molecular Medicine in the Helmholtz Association (MDC), Robert- Rössle-Strasse 10, 13125 Berlin, Germany 2current address: University of California, San Francisco, 513 Parnassus Avenue, CA 94122 USA 3Leibniz-Forschungsinstitut für Molekulare Pharmakologie (FMP), Robert-Rössle-Strasse 10, 13125 Berlin, Germany 4DZHK (German Centre for Cardiovascular Research), Partner Site Berlin, Oudenarder Strasse 16, 13347 Berlin, Germany *Corresponding author Enno Klussmann Max Delbrück Center for Molecular Medicine Berlin in the Helmholtz Association (MDC) Robert-Rössle-Str. 10, 13125 Berlin Germany Tel. +49-30-9406 2596 FAX +49-30-9406 2593 E-mail: [email protected] 1 Content 1. CELL-BASED SCREENING BY AUTOMATED IMMUNOFLUORESCENCE MICROSCOPY 3 1.1 Screening plates 3 1.2 Image analysis using CellProfiler 17 1.4 Identification of siRNA affecting cell viability 18 1.7 Hits 18 2. SUPPLEMENTARY TABLE S4, FIGURES S2-S4 20 2 1. Cell-based screening by automated immunofluorescence microscopy 1.1 Screening plates Table S1. Genes targeted with the Mouse Protein Kinases siRNA sub-library. Genes are sorted by plate and well. Accessions refer to National Center for Biotechnology Information (NCBI, BLA) entries. The siRNAs were arranged on three 384-well microtitre platres. -

1 a Clinical Approach to Inherited Metabolic Diseases
1 A Clinical Approach to Inherited Metabolic Diseases Jean-Marie Saudubray, Isabelle Desguerre, Frédéric Sedel, Christiane Charpentier Introduction – 5 1.1 Classification of Inborn Errors of Metabolism – 5 1.1.1 Pathophysiology – 5 1.1.2 Clinical Presentation – 6 1.2 Acute Symptoms in the Neonatal Period and Early Infancy (<1 Year) – 6 1.2.1 Clinical Presentation – 6 1.2.2 Metabolic Derangements and Diagnostic Tests – 10 1.3 Later Onset Acute and Recurrent Attacks (Late Infancy and Beyond) – 11 1.3.1 Clinical Presentation – 11 1.3.2 Metabolic Derangements and Diagnostic Tests – 19 1.4 Chronic and Progressive General Symptoms/Signs – 24 1.4.1 Gastrointestinal Symptoms – 24 1.4.2 Muscle Symptoms – 26 1.4.3 Neurological Symptoms – 26 1.4.4 Specific Associated Neurological Abnormalities – 33 1.5 Specific Organ Symptoms – 39 1.5.1 Cardiology – 39 1.5.2 Dermatology – 39 1.5.3 Dysmorphism – 41 1.5.4 Endocrinology – 41 1.5.5 Gastroenterology – 42 1.5.6 Hematology – 42 1.5.7 Hepatology – 43 1.5.8 Immune System – 44 1.5.9 Myology – 44 1.5.10 Nephrology – 45 1.5.11 Neurology – 45 1.5.12 Ophthalmology – 45 1.5.13 Osteology – 46 1.5.14 Pneumology – 46 1.5.15 Psychiatry – 47 1.5.16 Rheumatology – 47 1.5.17 Stomatology – 47 1.5.18 Vascular Symptoms – 47 References – 47 5 1 1.1 · Classification of Inborn Errors of Metabolism 1.1 Classification of Inborn Errors Introduction of Metabolism Inborn errors of metabolism (IEM) are individually rare, but collectively numerous.