Elemental Analysis and Phenolic Profiles of Selected Italian Wines
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-
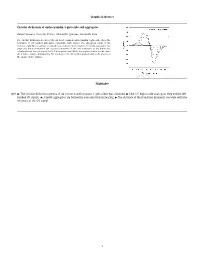
Graphical Abstract Circular Dichroism of Anthocyanidin 3-Glucoside Self
Graphical abstract Circular dichroism of anthocyanidin 3-glucoside self-aggregates Raquel Gavara, Vesselin Petrov, Alexandre Quintas, Fernando Pina * The circular dichroism spectra of the six most common anthocyanidin 3-glucoside show the formation of left handed aggregates compatible with dimers. The absorption bands of the monomer split by increasing concentration according to the formation of H and J aggregates. The angle and distance between the transition moments of the two monomers in the dimer was calculated from the splitting of the 0–0 absorption band. While the angle is similar for the series the distance changes dramatically. The intensity of the CD signal is proportional to the inverse of the square of the distance. Highlights Q10 " The circular dichroism spectra of six common anthocyanins 3-glucosides was obtained. " Like 3,5-diglucoside analogous they exhibit left- handed CD signals. " J and H aggregates are formed by concentration increasing. " The distance of the transition moments correlate with the intensity of the CD signal. 1 1 2 Circular dichroism of anthocyanidin 3-glucoside self-aggregates a a b a,⇑ 3 Q1 Raquel Gavara , Vesselin Petrov , Alexandre Quintas , Fernando Pina 4 a REQUIMTE, Departamento de Química, Faculdade de Ciências e Tecnologia, Universidade Nova de Lisboa, 2829516 Monte de Caparica, Portugal 5 b Instituto Superior de Ciencias da Saude Egas Moniz, Centro de Investigação Interdisciplinar Egas Moniz, P-2829511 Monte de Caparica, Caparica, Portugal 6 7 article info a b s t r a c t 1 9 10 Self-association constants for the flavylium cations of the six most common anthocyanidin 3-glucosides 20 11 were determined by circular dichroism (CD) and UV–Vis spectroscopy. -

A Combined Experimental and Computational Study of Chrysanthemin As a Pigment for Dye-Sensitized Solar Cells
molecules Article A Combined Experimental and Computational Study of Chrysanthemin as a Pigment for Dye-Sensitized Solar Cells Atoumane Ndiaye 1,2 , Alle Dioum 1, Corneliu I. Oprea 2 , Anca Dumbrava 3,* , Jeanina Lungu 2, Adrian Georgescu 2, Florin Moscalu 2, Mihai A. Gîr¸tu 2,* , Aboubaker Chedikh Beye 1 and Issakha Youm 1,* 1 Department of Physics, Cheikh Anta Diop University of Dakar, 5005 Dakar-Fann, Senegal; [email protected] (A.N.); [email protected] (A.D.); [email protected] (A.C.B.) 2 Department of Physics, Ovidius University of Constanta, 900527 Constanta, Romania; [email protected] (C.I.O.); [email protected] (J.L.); [email protected] (A.G.); fl[email protected] (F.M.) 3 Department of Chemistry and Chemical Engineering, Ovidius University of Constanta, 900527 Constanta, Romania * Correspondence: [email protected] (A.D.); [email protected] (M.A.G.); [email protected] (I.Y.) Abstract: The theoretical study of chrysanthemin (cyanidin 3-glucoside) as a pigment for TiO2-based dye-sensitized solar cells (DSSCs) was performed with the GAUSSSIAN 09 simulation. The electronic spectra of neutral and anionic chrysanthemin molecules were calculated by density functional theory with B3LYP functional and DGDZVP basis set. A better energy level alignment was found for partially deprotonated molecules of chrysanthemin, with the excited photoelectron having enough energy in order to be transferred to the conduction band of TiO2 semiconductor in DSSCs. In addition, we used the raw aqueous extracts of roselle (Hibiscus sabdariffa) calyces as the source of chrysanthemin Citation: Ndiaye, A.; Dioum, A.; and the extracts with various pH values were tested in DSSCs. -

Chemistry and Pharmacology of Kinkéliba (Combretum
CHEMISTRY AND PHARMACOLOGY OF KINKÉLIBA (COMBRETUM MICRANTHUM), A WEST AFRICAN MEDICINAL PLANT By CARA RENAE WELCH A Dissertation submitted to the Graduate School-New Brunswick Rutgers, The State University of New Jersey in partial fulfillment of the requirements for the degree of Doctor of Philosophy Graduate Program in Medicinal Chemistry written under the direction of Dr. James E. Simon and approved by ______________________________ ______________________________ ______________________________ ______________________________ New Brunswick, New Jersey January, 2010 ABSTRACT OF THE DISSERTATION Chemistry and Pharmacology of Kinkéliba (Combretum micranthum), a West African Medicinal Plant by CARA RENAE WELCH Dissertation Director: James E. Simon Kinkéliba (Combretum micranthum, Fam. Combretaceae) is an undomesticated shrub species of western Africa and is one of the most popular traditional bush teas of Senegal. The herbal beverage is traditionally used for weight loss, digestion, as a diuretic and mild antibiotic, and to relieve pain. The fresh leaves are used to treat malarial fever. Leaf extracts, the most biologically active plant tissue relative to stem, bark and roots, were screened for antioxidant capacity, measuring the removal of a radical by UV/VIS spectrophotometry, anti-inflammatory activity, measuring inducible nitric oxide synthase (iNOS) in RAW 264.7 macrophage cells, and glucose-lowering activity, measuring phosphoenolpyruvate carboxykinase (PEPCK) mRNA expression in an H4IIE rat hepatoma cell line. Radical oxygen scavenging activity, or antioxidant capacity, was utilized for initially directing the fractionation; highlighted subfractions and isolated compounds were subsequently tested for anti-inflammatory and glucose-lowering activities. The ethyl acetate and n-butanol fractions of the crude leaf extract were fractionated leading to the isolation and identification of a number of polyphenolic ii compounds. -

Anthocyanins, Vibrant Color Pigments, and Their Role in Skin Cancer Prevention
biomedicines Review Anthocyanins, Vibrant Color Pigments, and Their Role in Skin Cancer Prevention 1,2, , 2,3, 4,5 3 Zorit, a Diaconeasa * y , Ioana S, tirbu y, Jianbo Xiao , Nicolae Leopold , Zayde Ayvaz 6 , Corina Danciu 7, Huseyin Ayvaz 8 , Andreea Stanilˇ aˇ 1,2,Madˇ alinaˇ Nistor 1,2 and Carmen Socaciu 1,2 1 Faculty of Food Science and Technology, University of Agricultural Sciences and Veterinary Medicine, 400372 Cluj-Napoca, Romania; [email protected] (A.S.); [email protected] (M.N.); [email protected] (C.S.) 2 Institute of Life Sciences, University of Agricultural Sciences and Veterinary Medicine, Calea Mănă¸stur3-5, 400372 Cluj-Napoca, Romania; [email protected] 3 Faculty of Physics, Babes, -Bolyai University, Kogalniceanu 1, 400084 Cluj-Napoca, Romania; [email protected] 4 Institute of Chinese Medical Sciences, State Key Laboratory of Quality Research in Chinese Medicine, University of Macau, Taipa, Macau 999078, China; [email protected] 5 International Research Center for Food Nutrition and Safety, Jiangsu University, Zhenjiang 212013, China 6 Faculty of Marine Science and Technology, Department of Marine Technology Engineering, Canakkale Onsekiz Mart University, 17100 Canakkale, Turkey; [email protected] 7 Victor Babes University of Medicine and Pharmacy, Department of Pharmacognosy, 2 Eftimie Murgu Sq., 300041 Timisoara, Romania; [email protected] 8 Department of Food Engineering, Engineering Faculty, Canakkale Onsekiz Mart University, 17020 Canakkale, Turkey; [email protected] * Correspondence: [email protected]; Tel.: +40-751-033-871 These authors contributed equally to this work. y Received: 31 July 2020; Accepted: 25 August 2020; Published: 9 September 2020 Abstract: Until today, numerous studies evaluated the topic of anthocyanins and various types of cancer, regarding the anthocyanins’ preventative and inhibitory effects, underlying molecular mechanisms, and such. -

Effects of Anthocyanins on the Ahr–CYP1A1 Signaling Pathway in Human
Toxicology Letters 221 (2013) 1–8 Contents lists available at SciVerse ScienceDirect Toxicology Letters jou rnal homepage: www.elsevier.com/locate/toxlet Effects of anthocyanins on the AhR–CYP1A1 signaling pathway in human hepatocytes and human cancer cell lines a b c d Alzbeta Kamenickova , Eva Anzenbacherova , Petr Pavek , Anatoly A. Soshilov , d e e a,∗ Michael S. Denison , Michaela Zapletalova , Pavel Anzenbacher , Zdenek Dvorak a Department of Cell Biology and Genetics, Faculty of Science, Palacky University, Slechtitelu 11, 783 71 Olomouc, Czech Republic b Institute of Medical Chemistry and Biochemistry, Faculty of Medicine and Dentistry, Palacky University, Hnevotinska 3, 775 15 Olomouc, Czech Republic c Department of Pharmacology and Toxicology, Charles University in Prague, Faculty of Pharmacy in Hradec Kralove, Heyrovskeho 1203, Hradec Kralove 50005, Czech Republic d Department of Environmental Toxicology, University of California, Meyer Hall, One Shields Avenue, Davis, CA 95616-8588, USA e Institute of Pharmacology, Faculty of Medicine and Dentistry, Palacky University, Hnevotinska 3, 775 15 Olomouc, Czech Republic h i g h l i g h t s • Food constituents may interact with drug metabolizing pathways. • AhR–CYP1A1 pathway is involved in drug metabolism and carcinogenesis. • We examined effects of 21 anthocyanins on AhR–CYP1A1 signaling. • Human hepatocytes and cell lines HepG2 and LS174T were used as the models. • Tested anthocyanins possess very low potential for food–drug interactions. a r t i c l e i n f o a b s t r a c t -

Compositions of Anthocyanin and Other Flavonoids in Cultured Cells of Rabbiteye Blueberry (Vaccinium Ashei Reade Cv
Food Sci. Technol Res., 10 (3), 239-246, 2004 Compositions of Anthocyanin and Other Flavonoids in Cultured Cells of Rabbiteye Blueberry (Vaccinium ashei Reade cv. Tiiblue) Shioka HAMAMATSU,Kimiko YABE and Yoshihiko NAWA National Food Research Institute, 2-1-12 Kannondai, Tsukuba, Ibaraki 305-8642, Japan Received August 18, 2003 ; Accepted April 2, 2004 Our blueberry cultured cells produce anthocyanins in high quantity (Nawa et al., Biosci. Biotech. Biochem., 57, 770-774, 1993). Using high performance liquid chromatography/mass spectrometry (LC-MS), 14 anthocyanins in the red cells derived from leaves of rabbiteye blueberry cv. Tifblue (Vaccinium ashei Reade) were identified as 3-galactoside (gal), 3-glucoside (glc) and 3-arabinoside (ara) of cyanidin (Cy), delphinidin (Dp), petunidin (Pe), peonidin (Pn) and malvidin (Mv), except for Mv 3-ara. The fruits additionally contained 15 anthocyanins and Mv 3-ara, as a main anthocyanin. The autumn red leaves contained Cy 3-gal, Cy 3-ara and Cy 3-glc. In the red cells and the leaves, derivatives of Cy were the most abundant at 85.7% (Cy 3-gal 49.1%, Cy 3-glc 5.8%, Cy 3-ara 30.9%) and 100% (Cy 3-gal 51.0%, Cy 3-glc 6.5%, Cy 3-ara 42.5%) of the total anthocyanins, respectively. In contrast, the fruits contained Mv derivatives as main components that amounted to 50.9% of the total anthocyanins; Mv 3-gal, Mv 3-ara and Mv 3-glc constituted 21.2, 15.3 and 14.4%, respectively. Cy derivatives in fruit anthocyanins amounted to 18.0% . The red cells produced 107 mg of anthocyaninlIOO g fr wt, which was slightly lower than that of the fruits (129 mg). -

A Biochemical Survey of Some Mendelian Factors for Flower Colour
A BIOCHEMICAL 8UP~VEY OF SOME MENDELIAN FACTOI%S FO].~ FLOWEP~ COLOU~. BY ROSE SCOTT-MONCI~IEFF. (John Inncs Horticultural Institution, London.) (With One Text-figure.) CONTENTS. PAGE P~rb I. Introductory ].17 (a) The plastid 1)igmenl~s ] 21 (b) The a,n~hoxan~hius: i~heir backgromld, co-pigment and interaction effecbs upon flower-colour v~ri~bion 122 (c) The ani~hocyauins ] 25 (c) Col[oidM condition . 131 (f) Anthoey~nins as indic~bors 132 (g) The source of tim ~nl;hoey~nins 133 ]?ar[ II, Experimental 134 A. i~ecen~ investigations: (a) 2Prim,ula si,sensis 134- (b) Pa,l)aver Rhoeas 14.1 (c) Primuln aca.ulis 147 (d) Chc.l)ranth'ss Chci,rl 148 (e) ltosa lmlyanlha . 149 (f) Pelargonium zomdc 149 (g) Lalh,ymts odor~,l,us 150 (h) Vcrbom, hybrids 153 (i) Sl;'e2)loca~'])uG hybrids 15~ (j) T'rol)aeolu,m ,majors ] 55 ]3. B,eviews of published remflts of bhe t~u~horand o~hers.. (a) Dahlia variabilis (Lawreuce and Scol,~-Monerieff) 156 (b) A.nlb'rhinum majors (Wheklalo-Onslow, :Basseb~ a,nd ,~cobb- M.oncrieff ) 157 (c) Pharbilis nil (I-Iagiwam) . 158 (d) J/it& (Sht'itl.er it,lid Anderson) • . 159 (e) Zect d]f.ctys (~&udo, Miiner trod 8borl/lall) 159 Par~, III. The generM beh~wiour of Mendelian £acbors rot' flower colour . 160 Summary . 167 tLefermmes 168 I)AI~T I. II~TI~O])UOTOnY. Slm~C~ Onslow (1914) m~de the first sfudy of biochemica] chal~ges in- volved in flower-eolour va,riadon, our pro'ely chemical knowledge of bhe 118 A Bio&emical Su~'vey oI' Factor's fo~ • Flowe~' Colou~' anthocya.nin pigments has been considerably advanced by the work of Willstgtter, P~obinson, Karrer and their collaborators. -

Effects of Ph on the Stability of Cyanidin and Cyanidin 3-O-Β-Glucopyranoside in Aqueous Solution
Effects of pH on the stability of cyanidin and cyanidin 3-O-β-glucopyranoside in aqueous solution Violeta P. Rakić1, Mihaela A. Skrt2, Milena N. Miljković3, Danijela A. Kostić3, Dušan T. Sokolović4, Nataša E. Poklar Ulrih2,5 1College of Agriculture and Food Technology, Prokuplje, Serbia 2Department of Food Science and Technology, Biotechnical Faculty, University of Ljubljana, Ljubljana, Slovenia 3Department of Chemistry, Faculty of Science and Mathematics, University of Niš, Niš, Serbia 4Faculty of Medicine, University of Niš, Niš, Serbia 5Centre of Excellence for Integrated Approaches in Chemistry and Biology of Proteins (CipKeBiP), Ljubljana, Slovenia Abstract The colour variation, colour intensity and stability at various pH values (2.0, 4.0, 7.0 and SCIENTIFIC PAPER 9.0) of cyanidin 3-O-β-glucopyranoside (Cy3Glc) and its aglycone cyanidin were inves- tigated during a period of 8 hours storage at 25 °C. Our data showed that pH of aqueous UDC 547.973:667.777:543.42:544 solution had impact on spectroscopic profile of cyanidin and Cy3Glc. Beginning with the most acidic solutions, increasing the pH induce bathochromic shifts of absorbance maxi- mum in the visible range for all examined pH values (with the exception pH 4.0 for cya- Hem. Ind. 69 (5) 511–522 (2015) nidin), while the presence of the 3-glucosidic substitution induce hypsochromic shift. Com- pared to cyanidin, Cy3Glc has higher colour intensity and higher stability in the whole pH doi: 10.2298/HEMIND140711072R range, except at pH 7.0. The 3-glucosidic substitution influences on the colour intensity of Cy3Glc in the alkaline region. After 8-hour incubation of Cy3Glc and cyanidin at pH 2.0 and 25 °C, 99% of Cy3Glc and only 27% of cyanidin remained unchanged. -

Cyanidin-3-O-Glucoside: Physical-Chemistry, Foodomics and Health Effects
molecules Review Cyanidin-3-O-glucoside: Physical-Chemistry, Foodomics and Health Effects Francisco J. Olivas-Aguirre 1, Joaquín Rodrigo-García 1, Nina del R. Martínez-Ruiz 1, Arely I. Cárdenas-Robles 2, Sandra O. Mendoza-Díaz 2, Emilio Álvarez-Parrilla 1, Gustavo A. González-Aguilar 3, Laura A. de la Rosa 1, Arnulfo Ramos-Jiménez 1 and Abraham Wall-Medrano 1,* 1 Instituto de Ciencias Biomédicas, Departamento de Ciencias Químico-Biológicas, Universidad Autónoma de Ciudad Juárez, Anillo Envolvente del PRONAF y Estocolmo s/n, Ciudad Juárez 32310, Chihuahua, Mexico; [email protected] (F.J.O.-A.); [email protected] (J.R.-G.); [email protected] (N.d.R.M.-R.); [email protected] (E.Á.-P.); [email protected] (L.A.d.l.R.); [email protected] (A.R.-J.) 2 Departamento de Investigación y Posgrado en Alimentos, Facultad de Química, Universidad Autónoma de Querétaro, Cerro de las Campanas s/n, Querétaro 76010, Querétaro, Mexico; [email protected] (A.I.C.-R.); [email protected] (S.O.M.-D.) 3 Coordinación de Tecnología de Alimentos de Origen Vegetal, Centro de Investigación en Alimentación y Desarrollo, AC. Carretera a la Victoria km. 0.6, AP 1735, Hermosillo 83000, Sonora, Mexico; [email protected] * Correspondence: [email protected]; Tel.: +52-(656)-688-1821; Fax: +52-(656)-688-1800 Academic Editors: Celestino Santos-Buelga and Arturo San Feliciano Received: 15 August 2016; Accepted: 13 September 2016; Published: 21 September 2016 Abstract: Anthocyanins (ACNs) are plant secondary metabolites from the flavonoid family. Red to blue fruits are major dietary sources of ACNs (up to 1 g/100 g FW), being cyanidin-3-O-glucoside (Cy3G) one of the most widely distributed. -

Color and Pigment Analyses in Fruit Products
Station Bulletin 624 Reprinted May 1993 Color and Pigment Analyses in Fruit Products Agriculiural Experiment Station Oregon State University For additional copies of this publication, write: Publications Orders Agricultural Communications Oregon State University Administrative Services A422 Corvallis, OR 97331-2119 Agricultural Experiment Station Oregon State University Station Bulletin 624 Reprinted May 1993 Color and Pigment Analyses in Fruit Products Ronald E. Wroistad Professor of Food Science and Technology Oregon State University In July 1992 the warehouse storing Oregon Agricultural Experiment Station publications was destroyed by fire. SB 624, originally published in October, 1976, is hereby reprinted in its entirety. COLOR AND PIGMENT ANALYSES IN FRUIT PRODUCTS Ronald E. Wroistad ABSTRACT Methods are described for making the following deter- minations in fruits and processed fruit products: anthocyanin pigment content, color density, polymeric color, browning, turbidity, and anthocyanin degrada- tion index. Tables list the anthocyanins of common fruits, their molecular weights, molar absorbances, and wavelengths of maximum absorption. Thirty-one references are given. KEY WORDS: color analyses, anthocyanin pigment, browning, fruits Introduction Measurement of pigment content, browning, color density, and haze are analytical problems confronting the food technologist working with fruit concentrates, juices, wines, jams and other processedpro- ducts. This publication includes some of the methods we have found practical in determining these parameters. We also have included tables which list anthocyanin pigments of fruits likelyto be processed in the Northwest and the molar absorption of the common occurring anthocyanin pigments. We have not included procedures of pigment extraction, isolation, and identification or methods for tristimulus colorimetry.Tristimulus colorimeters, such as the Hunter color-difference meter and the Lovi- bond tintometer, give the best measurements for visualappearance of a product. -

Anthocyanin Pigments in Redbud (Cercis Spp) Flowers
Veazie et al. J Hortic Sci Res 2017, 1(1):13-18 DOI: 10.36959/745/393 | Volume 1 | Issue 1 Journal of Horticultural Science and Research Research Article Open Access Anthocyanin Pigments in Redbud (Cercis spp) Flowers Penelope Perkins-Veazie*, Guoying Ma and Dennis Werner Department of Horticultural Science, Plants for Human Health Institute, North Carolina State University, USA Abstract Redbud (Cercis spp.) is used as a spring flowering ornamental tree and is found wild in much of North America. Typically flowers are light purple although there are selected cultigens that are white, rose, or red-purple. Flowers from cultigens common to the eastern U.S. and from wild Eastern redbud (C. canadensis) were collected and tested for color and anthocyanin pigment composition. The anthocyanins cyanidin 3-glucoside, petunidin 3-glucoside, peonidin 3-glucoside, and malvidin 3-glucoside were most aboundant in purple, rose, and red-purple redbud flowers and total anthocyanin content was 2263 to 8730 mg.kg DW-1. Small amounts of delphinidin, cyanidin, and petunidin 3, diglucosides were also present. Most of the typical purple-flowered redbuds contained cyanidin 3-glucoside as the dominant pigment, while the red-purple flowered ‘Appalachian Red’ and ‘Crosswicks Red’ contained malvidin 3,5-diglucoside as the dominant anthocyanin. An unknown anthocyanin was present in all redbud flowers, and was higher in the red-purple flowered phenotypes. These results show that the color of redbud flowers is from anthocyanins, predominantly cyanidin 3-glucoside and malvidin 3,5-diglucoside, with malvidin 3,5-diglucoside as the primary pigment in red-purple flowers and cyanidin 3-glucoside dominant in purple flowers. -

Supplementary Materials
Supplementary Materials Table S1. Metals with stdev W1 W2 W3 W4 W5 W6 W7 W8 W9 W10 W11 W12 W13 As <LOD <LOD 19.84±0.03 <LOD <LOD <LOD <LOD <LOD <LOD <LOD <LOD <LOD <LOD Ba 32.6±0.3 33.8±0.4 26.1±0.2 123.3±0.9 79.8±0.8 79.1±0.7 83.3±0.6 239.7±0.8 58.1±0.4 77.7±0.3 32.1±0.3 31.7±0.5 122.1±0.8 Cd 0.365±0.004 1.553±0.002 0.939±0.002 1.254±0.003 0.386±0.005 1.367±0.008 0.733±0.004 0.050±0.002 0.976±0.002 1.644±0.006 <LOD 0.593±0.004 0.375±0.003 Co 3.64±0.04 2.09±0.02 0.98±0.03 4.40±0.06 2.77±0.02 1.83±0.02 2.42±0.02 2.68±0.03 7.34±0.09 7.78±0.07 2.12±0.04 1.61±0.02 2.60±0.03 Cr <LOD <LOD <LOD <LOD 4.322±0.005 3.030±0004 <LOD 6.417±0.006 15.41±0.02 14.82±0.05 11.12±0.04 9.15±0.02 3.111±0.004 Pb <LOD 56.0±0.8 <LOD 20.8±0.9 80±2 11.7±0.6 2.20±0.05 175±3 45.0±0.8 <LOD 0.830±0.007 0.538±0.006 14.9±0.3 Sb 32.8±0.5 8.59±0.09 19.1±0.2 18.±0.2 67.5±0.5 37.9±0.2 9.58±0.05 46.5±0.3 44.8±0.4 18.50±0.08 7.26±0.03 44.3±0.2 29.6±0.4 Se <LOD <LOD <LOD <LOD 1.45±0.09 0.30±0.05 0.23±0.08 25.7±0.4 2.87±0.2 2.06±0.04 2.91±0.8 2.38±0.07 2.99±0.06 Ni 4.17±0.05 46.0±0.2 0.569±0.005 30.9±0.3 15.3±0.2 5.77±0.02 35.2±0.3 13.86±0.04 50.6±0.7 17.9±0.2 9.99±0.03 0.197±0.007 5.00±0.02 Al 566±5 260.1±0.5 155.3±0.4 720±5 447±4 290.7±2 26.5±0.4 155.4±0.9 444±4 329±3 1027±10 567.7±8 236±3 Cu 97.5±0.8 469±3 36.3±0.9 10.07±0.05 <LOD <LOD <LOD <LOD 21.19±0.07 7.73±0.03 0.813±0.05 49.3±0.8 50.2±1.0 Mn 735±30 613±10 642±6 1016±6 762±3 743±5 839±8 1634±30 1732±20 1472±20 887±10 771±6 1299±10 V 4.18±0.02 1.894±0.007 0.574±0.002 <LOD 1.530±0.005 <LOD 1.948±0.006 3.179±0.004 3.235±0.007