The Path of Carbon in Photosynthesis, XV. Ribulose and Sedoheptulose
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Postulated Physiological Roles of the Seven-Carbon Sugars, Mannoheptulose, and Perseitol in Avocado
J. AMER. SOC. HORT. SCI. 127(1):108–114. 2002. Postulated Physiological Roles of the Seven-carbon Sugars, Mannoheptulose, and Perseitol in Avocado Xuan Liu,1 James Sievert, Mary Lu Arpaia, and Monica A. Madore2 Department of Botany and Plant Sciences, University of California, Riverside, CA 92521 ADDITIONAL INDEX WORDS. ‘Hass’ avocado on ‘Duke 7’ rootstock, phloem transport, ripening, Lauraceae ABSTRACT. Avocado (Persea americana Mill.) tissues contain high levels of the seven-carbon (C7) ketosugar mannoheptulose and its polyol form, perseitol. Radiolabeling of intact leaves of ‘Hass’ avocado on ‘Duke 7’ rootstock indicated that both perseitol and mannoheptulose are not only primary products of photosynthetic CO2 fixation but are also exported in the phloem. In cell-free extracts from mature source leaves, formation of the C7 backbone occurred by condensation of a three-carbon metabolite (dihydroxyacetone-P) with a four-carbon metabolite (erythrose-4-P) to form sedoheptulose-1,7- bis-P, followed by isomerization to a phosphorylated D-mannoheptulose derivative. A transketolase reaction was also observed which converted five-carbon metabolites (ribose-5-P and xylulose-5-P) to form the C7 metabolite, sedoheptu- lose-7-P, but this compound was not metabolized further to mannoheptulose. This suggests that C7 sugars are formed from the Calvin Cycle, not oxidative pentose phosphate pathway, reactions in avocado leaves. In avocado fruit, C7 sugars were present in substantial quantities and the normal ripening processes (fruit softening, ethylene production, and climacteric respiration rise), which occurs several days after the fruit is picked, did not occur until levels of C7 sugars dropped below an apparent threshold concentration of ≈20 mg·g–1 fresh weight. -

United States Patent Office
- 2,926,180 United States Patent Office Patented Feb. 23, 1960 2 cycloalkyl, etc. These substituents R and R' may also be substituted with various groupings such as carboxyl 2,926,180 groups, sulfo groups, halogen atoms, etc. Examples of CONDENSATION OF AROMATIC KETONES WITH compounds which are included within the scope of this CARBOHYDRATES AND RELATED MATER ALS 5 general formula are acetophenone, propiophenone, benzo Carl B. Linn, Riverside, Ill., assignor, by mesne assign phenone, acetomesitylene, phenylglyoxal, benzylaceto ments, to Universal Oil Products Company, Des phenone, dypnone, dibenzoylmethane, benzopinacolone, Plaines, Ill., a corporation of Delaware dimethylaminobenzophenone, acetonaphthalene, benzoyl No Drawing. Application June 18, 1957 naphthalene, acetonaphthacene, benzoylnaphthacene, ben 10 zil, benzilacetophenone, ortho-hydroxyacetophenone, para Serial No. 666,489 hydroxyacetophenone, ortho - hydroxy-para - methoxy 5 Claims. (C. 260-345.9) acetophenone, para-hydroxy-meta-methoxyacetophenone, zingerone, etc. This application is a continuation-in-part of my co Carbohydrates which are condensed with aromatic pending application Serial No. 401,068, filed December 5 ketones to form a compound selected from the group 29, 1953, now Patent No. 2,798,079. consisting of an acylaryl-desoxy-alditol and an acylaryl This invention relates to a process for interacting aro desoxy-ketitol include simple sugars, their desoxy- and matic ketones with carbohydrates and materials closely omega-carboxy derivatives, compound sugars or oligo related to carbohydrates. The process relates more par saccharides, and polysaccharides. ticularly to the condensation of simple sugars, their 20 Simple sugars include dioses, trioses, tetroses, pentoses, desoxy- and their omega-carboxy derivatives, compound hexoses, heptoses, octoses, nonoses, and decoses. Com sugars or oligosaccharides, and polysaccharides with aro pound sugars include disaccharides, trisaccharides, and matic ketones in the presence of a hydrogen fluoride tetrasaccharides. -

Proquest Dissertations
Feeding and oviposition behavior of tobacco hornworms, Manduca sexta, in relation to Myo-inositol Item Type text; Thesis-Reproduction (electronic) Authors Nelson, Nancy Marie, 1968- Publisher The University of Arizona. Rights Copyright © is held by the author. Digital access to this material is made possible by the University Libraries, University of Arizona. Further transmission, reproduction or presentation (such as public display or performance) of protected items is prohibited except with permission of the author. Download date 07/10/2021 15:39:03 Link to Item http://hdl.handle.net/10150/278543 INFORMATION TO USERS This manuscript has been reproduced from the microfilm master. UMI films the text directly from the original or copy submitted. Thus, some thesis and dissertation copies are in typewriter face, while others may be from any type of computer printer. The quality of this reproduction is dependent upon the quality of the copy submitted. Broken or indistinct print, colored or poor quality illustrations and photographs, print bleedthrough, substandard margins, and improper alignment can adversely affect reproduction. In the unlikely event that the author did not send UMI a complete manuscript and there are missing pages, these will be noted. Also, if unauthorized copyright material had to be removed, a note will indicate the deletion. Oversize materials (e.g., maps, drawings, charts) are reproduced by sectioning the original, beginning at the upper left-hand comer and continuing from left to right in equal sections with small overlaps. Each original is also photographed in one exposure and is included in reduced form at the back of the book. -

PENTOSE PHOSPHATE PATHWAY — Restricted for Students Enrolled in MCB102, UC Berkeley, Spring 2008 ONLY
Metabolism Lecture 5 — PENTOSE PHOSPHATE PATHWAY — Restricted for students enrolled in MCB102, UC Berkeley, Spring 2008 ONLY Bryan Krantz: University of California, Berkeley MCB 102, Spring 2008, Metabolism Lecture 5 Reading: Ch. 14 of Principles of Biochemistry, “Glycolysis, Gluconeogenesis, & Pentose Phosphate Pathway.” PENTOSE PHOSPHATE PATHWAY This pathway produces ribose from glucose, and it also generates 2 NADPH. Two Phases: [1] Oxidative Phase & [2] Non-oxidative Phase + + Glucose 6-Phosphate + 2 NADP + H2O Ribose 5-Phosphate + 2 NADPH + CO2 + 2H ● What are pentoses? Why do we need them? ◦ DNA & RNA ◦ Cofactors in enzymes ● Where do we get them? Diet and from glucose (and other sugars) via the Pentose Phosphate Pathway. ● Is the Pentose Phosphate Pathway just about making ribose sugars from glucose? (1) Important for biosynthetic pathways using NADPH, and (2) a high cytosolic reducing potential from NADPH is sometimes required to advert oxidative damage by radicals, e.g., ● - ● O2 and H—O Metabolism Lecture 5 — PENTOSE PHOSPHATE PATHWAY — Restricted for students enrolled in MCB102, UC Berkeley, Spring 2008 ONLY Two Phases of the Pentose Pathway Metabolism Lecture 5 — PENTOSE PHOSPHATE PATHWAY — Restricted for students enrolled in MCB102, UC Berkeley, Spring 2008 ONLY NADPH vs. NADH Metabolism Lecture 5 — PENTOSE PHOSPHATE PATHWAY — Restricted for students enrolled in MCB102, UC Berkeley, Spring 2008 ONLY Oxidative Phase: Glucose-6-P Ribose-5-P Glucose 6-phosphate dehydrogenase. First enzymatic step in oxidative phase, converting NADP+ to NADPH. Glucose 6-phosphate + NADP+ 6-Phosphoglucono-δ-lactone + NADPH + H+ Mechanism. Oxidation reaction of C1 position. Hydride transfer to the NADP+, forming a lactone, which is an intra-molecular ester. -

Collagen-Based Matrix Matrix Auf Der Basis Von Kollagen Matrice À Base De Collagène
Europäisches Patentamt *EP000693523B1* (19) European Patent Office Office européen des brevets (11) EP 0 693 523 B1 (12) EUROPEAN PATENT SPECIFICATION (45) Date of publication and mention (51) Int Cl.7: C08H 1/06, A61L 27/00, of the grant of the patent: A61L 31/00 20.11.2002 Bulletin 2002/47 (21) Application number: 95111260.6 (22) Date of filing: 18.07.1995 (54) Collagen-based matrix Matrix auf der Basis von Kollagen Matrice à base de collagène (84) Designated Contracting States: • Noff, Matityahu AT BE CH DE DK ES FR GB GR IE IT LI LU MC NL Rehovot 76228 (IL) PT SE (74) Representative: Grünecker, Kinkeldey, (30) Priority: 19.07.1994 IL 11036794 Stockmair & Schwanhäusser Anwaltssozietät Maximilianstrasse 58 (43) Date of publication of application: 80538 München (DE) 24.01.1996 Bulletin 1996/04 (56) References cited: (73) Proprietor: COL-BAR R & D LTD. DE-A- 4 302 708 FR-A- 2 679 778 Ramat-Gan 52290 (IL) US-A- 4 971 954 (72) Inventors: Remarks: • Pitaru, Sandu The file contains technical information submitted Tel-Aviv 62300 (IL) after the application was filed and not included in this specification Note: Within nine months from the publication of the mention of the grant of the European patent, any person may give notice to the European Patent Office of opposition to the European patent granted. Notice of opposition shall be filed in a written reasoned statement. It shall not be deemed to have been filed until the opposition fee has been paid. (Art. 99(1) European Patent Convention). EP 0 693 523 B1 Printed by Jouve, 75001 PARIS (FR) EP 0 693 523 B1 Description [0001] The present invention concerns a collagen-based matrix and devices comprising this matrix. -

Pyrolysis of the Simplest Carbohydrate, Glycolaldehyde (CHO-CH2OH), and Glyoxal in a Heated Micro-Reactor
Pyrolysis of the Simplest Carbohydrate, Glycolaldehyde (CHO-CH2OH), and Glyoxal in a Heated Micro-Reactor Jessica Porterfield1, Joshua H. Baraban1, Tyler P. Troy2, Musahid Ahmed2, Michael C. McCarthy3, Kathleen M. Morgan4, John W. Daily5, Thanh Lam Nguyen6, John F. Stanton6, G. Barney Ellison1 1Department of Chemistry and Biochemistry, University of Colorado, Boulder, 80309- 0215, U.S.A., 2Chemical Sciences Division, Lawrence Berkeley National Laboratories, Berkeley, CA 94720, U.S.A., 3Harvard-Smithsonian Center for Astrophysics, Cambridge, MA 02138, 4Department of Chemistry , Xavier University of Louisiana , New Orleans, LA 70125-1098, 5Center for Combustion and Environmental Research, Department of Mechanical Engineering, University of Colorado, Boulder, CO 80309-0427, U.S.A., and 6Department of Chemistry; University of Texas at Austin; Austin, TX 78712. E-mail: [email protected] E-mail: [email protected] E-mail: [email protected] E-mail: [email protected] E-mail: [email protected] E-mail: [email protected] E-mail: [email protected] E-mail: [email protected] E-mail: [email protected] Email: [email protected] J. Physical Chemistry A (revised, March 2016) - 2 - Abstract Both glycolaldehyde and glyoxal have been pyrolyzed in a set of flash- pyrolysis micro-reactors. The pyrolysis products resulting from CHO-CH2OH and HCO-CHO were detected and identified by VUV photoionization mass spectrometry. Complementary product identification was provided by argon matrix infrared absorption spectroscopy. Pyrolysis pressures in the micro- reactor were roughly 100 Torr and contact times with the micro-reactors were approximately 100 µsec. At 1200 K, the products of glycolaldehyde pyrolysis are: H atoms, CO, CH2=O, CH2=C=O, HCO-CHO. -

Separation and Extraction of Proteins and Polysaccharides from the Seaweed Palmaria Palmata Using Enzyme Digestion
Separation and Extraction of Proteins and Polysaccharides from the Seaweed Palmaria Palmata using Enzyme Digestion by Anna Nilsson Supervisor: Björn Viðar Aðalbjörnsson Co-supervisor: Ph.D. Johan Svensson Bonde Examiner: Professor Leif Bülow 2017 Department of Pure and Applied Biochemistry Faculty of Science Lund University SE-221 00 Lund, Sweden Copywright © 2017 Anna Nilsson All rights reserved Printed in Sweden by Media-Tryck, Lund, 2017 DEPARTMENT OF CHEMICAL ENGINEERING LUND UNIVERSITY P.O. BOX 124 SE-221 00 LUND, SWEDEN ii Organization Document name Lund University Master thesis Date of issue April 2017 Author Anna Nilsson Title and subtitle Separation and Extraction of Proteins and Polysaccharides from the Seaweed Palmaria Palmata using Enzyme Digestion Key words Palmaria palmata, dulse, enzyme digestion, separation, protease, xylan, xylose, seaweed, xylanase Language English, Swedish Recipient’s notes Number of pages 67 iii PREFACE This master thesis was possible thanks to Matís, a food and biotechnology R&D institute located in Reykjavik, Iceland. It commenced 1st of February 2016 and ended with a presentation held the 27/4 2017. The purpose of the project was to find a method of enzymatic extraction of polysaccharides and proteins in Palmaria palmata (P. palmata) a red seaweed. The project focuses mainly on extraction using proteases, which catalyse the hydrolysis of proteins, and xylanase, that catalyse the hydrolysis of xylan. Xylan is the main polysaccharide in P. palmata. The findings intend to be beneficial and useful within the human food and animal feed industry. These new methods and ways to utilize this large ocean resource may lead to more sustainable and environmental solutions compared to already existing terrestrial options such as soy and other grains. -

Sedoheptulose in Photosynthesis by Plants
TWO-WEEK LOAN COPY This is a Library Circulating Copy which may be borrowed for two weeks. For a personal retention copy, call Tech. Info. Division, Ext. 5545 UCRW.268 Unclassified - Biology Distribution UNIVERSITY OF CALIFOmIA Radiation Laboratory Contract No. W-7405-eng-48 SEDOHEPT[JLOSE IN PHOTOSYNTHESIS BY PLANTS A. A. Benson, J. A. Bassham, and Me Calvin May 1, 1951 Berkeley, California A. A. Benson, 6. A, Bassham, and M, Calvin Radiation Laboratory and Department of Ohemistry University of Calif omia, Berkeley May 1, 1951 Although its function has not been ascertained, the general 1 occurrence of sedohep~ulose, D-altroheptuLose, in the succulent plants is well established. This sugar has not been identified in the majority of the members of the plat lringdom, but it now appears possibae that its phosphate esters may perform a vital function during photosynthesis. We haw isolated labeled sedoheptulose monopho sphate in cl% 2 photosynthesis products of all the plants thus far studied in this laboratory (~lorelh,Scenedesmus, Rhodospfr5.llyn rubn~,and the leaves of barley seedlings, soy bean, alf~lfa,sugar beet, spinach and geranium). It is invariably found as monophosphate esters. At least two such esters have been observed in radiograms of C1'-labeled Scenedesmus. The mjor one is associated with fructose monophosphztte while the minor one is inseparable, as yet, from glucose monophosphate. Sedoheptulose may be liberated e~zymticallyfrom its phosphates during the lrilling of the plan-i;,'but it has not been observed to accwnulate In amounts exceeding tbe steady state concentrations of -these phosphates. .--LL- * This work was sponsored bp the United States Atomic Energy Cormnission, -- This suggests its participation only as a phosphate in most plznts. -

Expression and Structure-Functional Studies of Human Apolipoproteinciii
Haiqun Liu EXPRESSION AND STRUCTURE-FUNCTIONAL STUDIES OF HUMAN APOLIPOPROTEINCra A thesis submitted in accordance with the regulations of the University of London for the degree of Doctor of Philosophy October 2000 Centre for the Genetics of Cardiovascular Disorders Department of Medicine The Rayne Institute University College London ProQuest Number: U642127 All rights reserved INFORMATION TO ALL USERS The quality of this reproduction is dependent upon the quality of the copy submitted. In the unlikely event that the author did not send a complete manuscript and there are missing pages, these will be noted. Also, if material had to be removed, a note will indicate the deletion. uest. ProQuest U642127 Published by ProQuest LLC(2015). Copyright of the Dissertation is held by the Author. All rights reserved. This work is protected against unauthorized copying under Title 17, United States Code. Microform Edition © ProQuest LLC. ProQuest LLC 789 East Eisenhower Parkway P.O. Box 1346 Ann Arbor, Ml 48106-1346 ABSTRACT Apolipoprotein (Apo) CHI plays a key role in triglyceride (TG)-rich lipoprotein metabolism and is a risk factor for coronary heart disease (CHD). The study involved in this thesis is the first in vitro structure-functional study using recombinant apoCni proteins. The physicochemical properties of recombinant wild type and A23T, a naturally occurring mutation that is associated with apoCIII deficiency and lower plasma TG levels, as well as three site-directed mutants of apoCIII, designed by molecular modelling and implicated in lipid binding (L9T/T20L, F64A/W65A) or lipoprotein lipase (LPL) inhibition (K21A), were compared. Relative lipid binding efficiencies of each apoCIII variants to 1.2-dimyristoyl-sn-glycero-3- phosphatidylcholine (DMPC) were: L9T/T20L>WT>K21A>A23T>F64A/W65A with an inverse correlation with size of the discoidal complexes formed. -
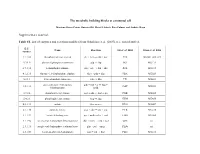
The Metabolic Building Blocks of a Minimal Cell Supplementary
The metabolic building blocks of a minimal cell Mariana Reyes-Prieto, Rosario Gil, Mercè Llabrés, Pere Palmer and Andrés Moya Supplementary material. Table S1. List of enzymes and reactions modified from Gabaldon et. al. (2007). n.i.: non identified. E.C. Name Reaction Gil et. al. 2004 Glass et. al. 2006 number 2.7.1.69 phosphotransferase system glc + pep → g6p + pyr PTS MG041, 069, 429 5.3.1.9 glucose-6-phosphate isomerase g6p ↔ f6p PGI MG111 2.7.1.11 6-phosphofructokinase f6p + atp → fbp + adp PFK MG215 4.1.2.13 fructose-1,6-bisphosphate aldolase fbp ↔ gdp + dhp FBA MG023 5.3.1.1 triose-phosphate isomerase gdp ↔ dhp TPI MG431 glyceraldehyde-3-phosphate gdp + nad + p ↔ bpg + 1.2.1.12 GAP MG301 dehydrogenase nadh 2.7.2.3 phosphoglycerate kinase bpg + adp ↔ 3pg + atp PGK MG300 5.4.2.1 phosphoglycerate mutase 3pg ↔ 2pg GPM MG430 4.2.1.11 enolase 2pg ↔ pep ENO MG407 2.7.1.40 pyruvate kinase pep + adp → pyr + atp PYK MG216 1.1.1.27 lactate dehydrogenase pyr + nadh ↔ lac + nad LDH MG460 1.1.1.94 sn-glycerol-3-phosphate dehydrogenase dhp + nadh → g3p + nad GPS n.i. 2.3.1.15 sn-glycerol-3-phosphate acyltransferase g3p + pal → mag PLSb n.i. 2.3.1.51 1-acyl-sn-glycerol-3-phosphate mag + pal → dag PLSc MG212 acyltransferase 2.7.7.41 phosphatidate cytidyltransferase dag + ctp → cdp-dag + pp CDS MG437 cdp-dag + ser → pser + 2.7.8.8 phosphatidylserine synthase PSS n.i. cmp 4.1.1.65 phosphatidylserine decarboxylase pser → peta PSD n.i. -

Chapter 3: the Chemical Basis for Life Lesson 2: Organic Compounds
Chapter 3: The Chemical Basis for Life Lesson 2: Organic Compounds We have already learned that water is the primary substance for life on Earth, but we will now explore the element found in most of the molecules from which living organisms are made. That element is carbon and it is found in all organic compounds. The picture above is a graphic depiction of the organic compounds: carbohydrates, proteins, lipids, and nucleic acids. These are all large complex molecules that have contributed to the great diversity of life on Earth. Lesson Objectives • Define and explain elements and compounds; the relationships from atom to molecule to macromolecule. • Explain why carbon is essential to life on Earth and uniquely suited to form biological macromolecules. • Describe and compare the structure and function of the four major types of organic compounds. Vocabulary • atom biochemical conversion biological macromolecule carbohydrate DNA (deoxyribonucleic acid) isomers lipids macromolecules molecule monomer nucleic acid organic molecule polymer protein 63 INTRODUCTION If you look at your hand, what do you see? Of course, you see skin, which consists of cells. But what are skin cells made of? Like all living cells, they are made of matter. In fact, all things are made of matter. Matter is anything that takes up space and has mass. Matter, in turn, is made up of chemical substances. In this lesson you will learn about the chemical substances that make up living things. CHEMICAL SUBSTANCES A chemical substance is matter that has a definite composition. It also has the same composition throughout. A chemical substance may be either an element or a compound. -

Nucleotide Sugars in Chemistry and Biology
molecules Review Nucleotide Sugars in Chemistry and Biology Satu Mikkola Department of Chemistry, University of Turku, 20014 Turku, Finland; satu.mikkola@utu.fi Academic Editor: David R. W. Hodgson Received: 15 November 2020; Accepted: 4 December 2020; Published: 6 December 2020 Abstract: Nucleotide sugars have essential roles in every living creature. They are the building blocks of the biosynthesis of carbohydrates and their conjugates. They are involved in processes that are targets for drug development, and their analogs are potential inhibitors of these processes. Drug development requires efficient methods for the synthesis of oligosaccharides and nucleotide sugar building blocks as well as of modified structures as potential inhibitors. It requires also understanding the details of biological and chemical processes as well as the reactivity and reactions under different conditions. This article addresses all these issues by giving a broad overview on nucleotide sugars in biological and chemical reactions. As the background for the topic, glycosylation reactions in mammalian and bacterial cells are briefly discussed. In the following sections, structures and biosynthetic routes for nucleotide sugars, as well as the mechanisms of action of nucleotide sugar-utilizing enzymes, are discussed. Chemical topics include the reactivity and chemical synthesis methods. Finally, the enzymatic in vitro synthesis of nucleotide sugars and the utilization of enzyme cascades in the synthesis of nucleotide sugars and oligosaccharides are briefly discussed. Keywords: nucleotide sugar; glycosylation; glycoconjugate; mechanism; reactivity; synthesis; chemoenzymatic synthesis 1. Introduction Nucleotide sugars consist of a monosaccharide and a nucleoside mono- or diphosphate moiety. The term often refers specifically to structures where the nucleotide is attached to the anomeric carbon of the sugar component.