Opioid Prescription Drug Monitoring Surveillance Report 2015
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Opioids): SOR/2018-77 Canada Gazette, Part II, Volume 152, Number 9
Canada Gazette, Part 2, Volume 152, Number 9: Regulations Amendin... http://gazette.gc.ca/rp-pr/p2/2018/2018-05-02/html/sor-dors77-eng.html Home (http://www.canada.ca/en/index.html) How government works (http://www.canada.ca/en/government/system/index.html) Treaties, laws and regulations (https://www.canada.ca/en/government/system/laws.html) Canada Gazette (/accueil-home-eng.html) Publications (/rp-pr/publications-eng.html) Part II: Vol. 152 (2018) (/rp-pr/p2/2018/index-eng.html) May 2, 2018 (/rp-pr/p2/2018/2018-05-02/html/index-eng.html) Regulations Amending the Food and Drug Regulations (Opioids): SOR/2018-77 Canada Gazette, Part II, Volume 152, Number 9 Registration April 23, 2018 FOOD AND DRUGS ACT P.C. 2018-429 April 20, 2018 Her Excellency the Governor General in Council, on the recommendation of the Minister of Health, pursuant to section 30a of the Food and Drugs Act b, makes the annexed Regulations Amending the Food and Drug Regulations (Opioids). Regulations Amending the Food and Drug Regulations (Opioids) Amendments 1 Subsections C.01.005(2) and (3) of the Food and Drug Regulations 1 are replaced by the following: (2) Subsection (1) does not apply to (a) a drug in dosage form that is compounded by a pharmacist under a prescription or by a practitioner; or (b) a drug in dosage form that is sold under a prescription if the following information appears on the drug’s label: (i) the drug’s proper name, common name or brand name, (ii) the drug’s potency, and (iii) the name of the drug’s manufacturer. -

Oklahoma Drug Threat Assessment
2018 Oklahoma Drug Threat Assessment Oklahoma Bureau of Narcotics and Dangerous Drugs i John Scully, Director MARY FALLIN JOHN SCULLY Governor Director September 12, 2018 The Oklahoma Bureau of Narcotics has served the citizens of Oklahoma in the quest for a drug- free state since 1975. Our agency remains committed to working with lawmakers, law enforcement, public health officials, and the citizens of Oklahoma to develop comprehensive strategies to address drug abuse in communities across the state. While we know many factors contribute to drug abuse, OBN is committed to reducing the availability of illegal drugs in Oklahoma. Our agency works to eradicate illegal drugs in Oklahoma by enforcing drug laws, administering statewide programs, and providing continual outreach to our stakeholders – lawmakers, law enforcement, public health officials, and the citizens of Oklahoma. OBN agents enforce drug laws by utilizing aggressive investigative methods and administering statewide drug diversion programs. The Prescription Drug Monitoring Program is a valuable tool for practitioners, pharmacists, and law enforcement in the prevention and detection of the diversion and abuse of pharmaceutical controlled substances. As a service to our communities, OBN also administers the Safe Trips for Scripts Drug Prevention Program. This program provides citizens a safe way to discard unwanted medications by disposing of them in one of our 177 take back boxes located around the state. This drug threat assessment was created to help provide officials and citizens with helpful information about drug threats across our state. We will continue to collaborate with other agencies and the citizens to work toward a safer and healthier Oklahoma. -
3.2.2 Opioids and Abuse, Misuse and Dependence
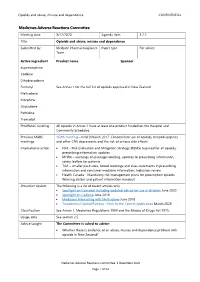
Opioids and abuse, misuse and dependence CONFIDENTIAL Medicines Adverse Reactions Committee Meeting date 3/12/2020 Agenda item 3.2.2 Title Opioids and abuse, misuse and dependence Submitted by Medsafe Pharmacovigilance Paper type For advice Team Active ingredient Product name Sponsor Buprenorphine Codeine Dihydrocodeine Fentanyl See Annex 1 for the full list of opioids approved in New Zealand Methadone Morphine Oxycodone Pethidine Tramadol PHARMAC funding All opioids in Annex 1 have at least one product funded on the Hospital and Community Schedules. Previous MARC 169th meeting – held 9 March 2017: Concomitant use of opioids, benzodiazepines meetings and other CNS depressants and the risk of serious side effects International action • FDA - Risk Evaluation and Mitigation Strategy (REMS) required for all opioids; prescribing information updates • MHRA – warnings on package labelling, updates to prescribing information, safety leaflets for patients • TGA – smaller pack sizes, boxed warnings and class statements in prescribing information and consumer medicine information, indication review • Health Canada – Mandatory risk management plans for prescription opioids, Warning sticker and patient information handout Prescriber Update The following is a list of recent articles only. • Spotlight on tramadol including updated advice for use in children June 2020 • Spotlight on Codeine June 2018 • Medicines Interacting with Methadone June 2018 • Transdermal Opioid Patches - Stick to the Correct Application March 2020 Classification See Annex 1. Medicines -

Patterns of Opioid Prescribing in Minnesota: 2012 and 2015
ISSUE BRIEF | APRIL, 2018 Patterns of Opioid Prescribing in Minnesota: 2012 and 2015 Introduction Key Findings: Opioids are a class of drugs that include prescription • Overall rates of opioid prescribing declined in opioid medications for pain relief —such as oxycodone Minnesota from 2012 to 2015, but the morphine (OxyContin®), hydrocodone (Vicodin®), codeine, morphine, milligram equivalents (MME) per prescription and fentanyl—as well as illicitly produced drugs like heroin increased. and fentanyl-related substances (also called fentanyl analogs).1 While prescription opioids play a role in the • Medicare and Medicaid, where eligibility is determined management of some types of severe acute, cancer-related by age, disability status, and/or income, covered and end-of-life pain, increased opioid use since 1990, approximately one-third of Minnesotans with general including for chronic pain unrelated to cancer, has resulted health coverage and accounted for two-thirds of opioid in sharply rising opioid addiction and overdoses, as well as prescriptions filled in 2015. increased healthcare utilization and costs. Recent Centers • Nearly one in three Minnesotans with an opioid for Disease Control and Prevention (CDC) guidelines point prescription in 2015 had multiple prescribers. out the limitations of the evidence base in support of opioid therapy for pain, recommend non-opioid therapy • In both 2012 and 2015, 6 in 10 opioid prescriptions for chronic pain, and emphasize the risks associated with were filled within 15 days of the patient’s last medical opioid therapy.2 In Minnesota, opioids—both prescription visit; however, 1 in 10 opioid prescriptions were filled and illicit—were responsible for 336 overdose deaths without a medical visit in the past 90 days, suggesting in 2015, more than a six-fold increase since 2000.3 In closer patient-prescriber communication or opioid 2016, opioid use accounted for 395 overdose deaths in oversight may be needed in some cases. -

Perioperative Immunosuppression and Risk of Cancer Progression: the Impact of Opioids on Pain Management
Hindawi Pain Research and Management Volume 2018, Article ID 9293704, 8 pages https://doi.org/10.1155/2018/9293704 Review Article Perioperative Immunosuppression and Risk of Cancer Progression: The Impact of Opioids on Pain Management Renata Zaja˛czkowska ,1 Wojciech Leppert,2 Joanna Mika ,3 Magdalena Kocot-Ke˛pska,4 Jarosław Woron´ ,1 Anna Wrzosek ,1 and Jerzy Wordliczek 1 1Department of Interdisciplinary Intensive Care, Jagiellonian University Medical College, Krako´w, Poland 2Department of Palliative Medicine, Poznan University of Medical Sciences, Poznan, Poland 3Department of Pain Pharmacology, Institute of Pharmacology, Polish Academy of Sciences, Krak´ow, Poland 4Department of Pain Research and Treatment, Jagiellonian University Medical College, Krako´w, Poland Correspondence should be addressed to Anna Wrzosek; [email protected] Received 24 May 2018; Revised 5 August 2018; Accepted 19 August 2018; Published 19 September 2018 Academic Editor: Massimiliano Valeriani Copyright © 2018 Renata Zaja˛czkowska et al. -is is an open access article distributed under the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited. Opioids comprise an important group of drugs used in cancer pain pharmacotherapy. In recent years, more and more studies have emerged indicating the potentially immunosuppressive effects of opioid analgesics and their serious consequences, including the risk of cancer progression. -e identification of these risks has prompted a search for other effective, and most importantly, safer methods of perioperative analgesic management. Regional analgesia techniques, which allow for a significant reduction in opioid dosing and thus diminish the risk of immunosuppression associated with these drugs, seem to offer substantial hope in this respect. -

The Main Tea Eta a El Mattitauli Mali Malta
THE MAIN TEA ETA USA 20180169172A1EL MATTITAULI MALI MALTA ( 19 ) United States (12 ) Patent Application Publication ( 10) Pub . No. : US 2018 /0169172 A1 Kariman (43 ) Pub . Date : Jun . 21 , 2018 ( 54 ) COMPOUND AND METHOD FOR A61K 31/ 437 ( 2006 .01 ) REDUCING APPETITE , FATIGUE AND PAIN A61K 9 / 48 (2006 .01 ) (52 ) U . S . CI. (71 ) Applicant : Alexander Kariman , Rockville , MD CPC . .. .. .. .. A61K 36 / 74 (2013 .01 ) ; A61K 9 / 4825 (US ) (2013 . 01 ) ; A61K 31/ 437 ( 2013 . 01 ) ; A61K ( 72 ) Inventor: Alexander Kariman , Rockville , MD 31/ 4375 (2013 .01 ) (US ) ( 57 ) ABSTRACT The disclosed invention generally relates to pharmaceutical (21 ) Appl . No. : 15 /898 , 232 and nutraceutical compounds and methods for reducing appetite , muscle fatigue and spasticity , enhancing athletic ( 22 ) Filed : Feb . 16 , 2018 performance , and treating pain associated with cancer, trauma , medical procedure , and neurological diseases and Publication Classification disorders in subjects in need thereof. The disclosed inven ( 51 ) Int. Ci. tion further relates to Kratom compounds where said com A61K 36 / 74 ( 2006 .01 ) pound contains at least some pharmacologically inactive A61K 31/ 4375 ( 2006 .01 ) component. pronuPatent Applicationolan Publication manu saJun . decor21, 2018 deSheet les 1 of 5 US 2018 /0169172 A1 reta Mitragynine 7 -OM - nitragynine *** * *momoda W . 00 . Paynantheine Speciogynine **** * * * ! 1000 co Speclociliatine Corynartheidine Figure 1 Patent Application Publication Jun . 21, 2018 Sheet 2 of 5 US 2018 /0169172 A1 -

Methadone & EDDP by Liquid/Liquid Extraction and Gas
OFFICE OF CHIEF MEDICAL EXAMINER CITY OF NEW YORK METHADONE & EDDP BY LIQUID/LIQUID EXTRACTION AND GAS CHROMATOGRAPHY/MASS SPECTROMETRY (SELECTIVE ION MONITORING) PRINCIPLE Methadone (Symoron, Dolophine, Amidone, Methadose, Physeptone, Heptadon and many other names) is a synthetic opioid developed in Germany in 1937. It is used medically as an analgesic and also as a maintenance anti-addictive and reductive preparation for use by patients with opioid dependence. Because it is structurally an acyclic analog of morphine, methadone also acts upon the same opioid receptors, and thus has many of the same effects. Because of its long duration of action, strong analgesic effects, methadone is also used in managing severe chronic pain. Eli Lilly and Company introduced methadone into the United States in 1947. The abuse of methadone results in approximately 5,000 overdose deaths per year in the United States. Methadone is mainly used in the treatment of opioid dependence. It has cross-tolerance with other opioids including morphine, codeine and heroin, and offers very similar effects but with a longer duration. Oral doses of methadone can stabilize patients by mitigating opioid withdrawal syndrome or making it more tolerable. Higher doses can block the euphoric effects of heroin and morphine. Methadone is produced and distributed by a number of pharmaceutical companies. The racemic hydrochloride is the only form available in most countries, such as the Netherlands, Belgium, France and in the United States (as of March 2008). The dextrorotary enantiomer of methadone, dextromethadone, is an NMDA antagonist rather than an opiate agonist. As a result, methadone medications used for opiate addiction occasionally will contain only levomethadone, the levorotary enantiomer. -

Citation Classics and Trends in the Field of Opioids: a Bibliometric Analysis
Open Access Original Article DOI: 10.7759/cureus.5055 Citation Classics and Trends in the Field of Opioids: A Bibliometric Analysis Hira F. Akbar 1 , Khadijah Siddiq 2 , Salman Nusrat 3 1. Internal Medicine, Dow Medical College, Dow University of Health Sciences, Karachi, PAK 2. Internal Medicine, Civil Hospital Karachi, Dow University of Health Sciences, Karachi, PAK 3. Gasteroenterology, University of Oklahoma Health Sciences Center, Oklahoma City, USA Corresponding author: Hira F. Akbar, [email protected] Abstract Introduction Bibliometric analysis is one of the emerging and latest statistical study type used to examine and keep a systemic record of the research done on a particular topic of a certain field. A number of such bibliometric studies are conducted on various topics of the medical science but none existed on the vast topic of pharmacology - opioids. Hence, we present a bibliometric analysis of the ‘Citation Classics’ of opioids. Method The primary database chosen to extract the citation classics of opioids was Scopus. Top 100 citation classics were arranged according to the citation count and then analyzed. Results The top 100 citation classics were published between 1957 and 2013, among which seventy-two were published from 1977 to 1997. Among all nineteen countries that contributed to these citation classics, United States of America alone produced sixty-three classics. The top three journals of the list were multidisciplinary and contained 36 citation classics. Endogenous opioids were the most studied (n=35) class of opioids among the citation classes and the most studied subject was of the neurosciences. Conclusion The subject areas of neurology and analgesic aspects of opioids are well established and endogenous and synthetic opioids were the most studied classes of opioids. -

OMAT (Opioid Medication Assisted Treatment Options for Opioid Substance Use Disorders Crossroads’ Turning Points, Inc
OMAT (Opioid Medication Assisted Treatment Options for Opioid Substance Use Disorders Crossroads’ Turning Points, Inc. Opioid = Pain Killer • Pain-killing agents that originate from the poppy flower and its product opium. www.endthepain.org/learning/Glossary.html • Originally, a term denoting synthetic narcotics resembling opiates, but increasingly used to refer to both opiates and synthetic narcotics. www.prostate-cancer.org/resource/glosso.html • Medications made from, artificially developed from, or containing opium, a potent pain-killing substance that is derived from poppy pods. www.ichelp.com/AboutIC/FAQs/Terminology/tabid/288/Default.asp x • The opioids include natural opioids (eg morphine), semisynthetics (eg heroin), and synthetics with morphine-like action (eg codeine, hydromorphone, methadone, oxycodone, fentanyl). www.cdag.cochrane.org/en/glossary.html List of Opioids • Heroin • Morphine • Codeine • Fentanyl • Hydrocodone (Vicodin) • Hydromorphone (Dilaudid) (more potent than Morphine) • Oxycodone (Percodan; Percocet) • Oxycontin • Methadone • Naltrexone (Vivitrol) • Buprenorphine • Others Conditions for which Opioids are prescribed: • Medical procedures • Dental procedures • Automobile and other accidents • Sports injuries • Acute pain • Chronic pain • Opioid substance use disorder Co-occurring Disorders (Substance Use and Mental Health disorders) • Anxiety Disorders • Depressive Disorders • Post-traumatic Stress Disorders • Schizophrenia • Eating, sleeping, adjustment, impulse-control, personality Disorders • Others Treatment -

Health Canada's Approach to Prescription Opioid Risk Management
Health Canada’s Approach to Prescription Opioid Risk Management RADARS System 13th Annual Scientific Meeting Silver Spring, Maryland May 15, 2019 Andrew Slot – Health Canada Overview • High-level description of select Health Canada initiatives to address problematic opioid use • Specific Health Canada actions on prescription opioids related to safe use, prescribing and advertising • Prescription opioid Risk Management Plans • Challenges and next steps 2 Overview of Canadian Federal Actions on Opioids • Regulatory Actions – Supporting the establishment of supervised consumption sites and drug checking technologies – Regulatory amendments to facilitate methadone prescribing and use of medical diacetylmorphine – Regulatory amendments to facilitate access to drugs for urgent public health needs – Proposed regulatory amendments to schedule tramadol – Intent to make all low-dose codeine products prescription- only medications – Enacted legislation to protect individuals who seek emergency assistance for overdose 3 Overview of Canadian Federal Actions on Opioids • Non-regulatory actions – Key funding initiatives through the Substance Use and Addictions Program (SUAP) – Supporting the development of treatment guidelines for Opioid Use Disorder (OUD) – Facilitated access to naloxone – Coordinating national data collection and publishing quarterly reports 4 Health Canada Actions on Prescription Opioids in the Post-market Space 1. Updated product monographs for opioids 2. Mandatory pharmacy stickers and patient information handouts 3. Stronger stance -

Amended Application for Authorization to Institute a Class Action Canada
Amended Application for authorization to institute a class action Canada Province of Quebec District of Montreal No. 500-06-001004-197 (Class Action) Superior Court (…) RICCARDO CAMARDA, with an elected domicile for the purpose hereof at 1250 René-Lévesque Boulevard West, Suite 4100, Montreal, Quebec H3B 4W8 Plaintiff v. ABBOTT LABORATORIES, LIMITED, a legal person, having its principal place of business at 75 boul. Pierre-Roux Est, CP 307, Victoriaville, Quebec G6P 6S9 and APOTEX INC., a legal person, having a place of business at 2970 André Avenue, Dorval, Quebec H9P 2P2 and ARALEZ PHARMACEUTICALS CANADA INC., a legal person having a place of business at 7100 West Credit Avenue, Suite 101, Mississauga, Ontario L5N 0E4 and BGP PHARMA ULC, a legal person, having a place of business at 1959 Upper Water Street, Suite 900, Halifax, Nova Scotia B3J 2X2 and BOEHRINGER INGELHEIM (CANADA) LTD., a legal person, having a place of business at 5180 South Service Road, Burlington, Ontario L7L 5H4 and BRISTOL-MYERS SQUIBB CANADA CO., a legal person, having its principal place of business at 2344 Alfred-Nobel Boulevard, Montreal, Quebec H4S 0A4 and CHURCH & DWIGHT CANADA CORP., a legal person, having its principal place of business at 5485 Ferrier Street, Mont-Royal, Quebec H4P 1M6 and COBALT PHARMACEUTICALS INC., a legal person, having a place of business at 17800 Lapointe Street, Mirabel, Quebec J7J 1P3 and ETHYPHARM INC., a legal person, having a place of business at 1000 De La Gauchetière, Suite 2400, Montreal, Quebec H3B 4W5 and GLAXOSMITHKLINE INC., a legal person, having its principal place of business at 245 Armand-Frappier Boulevard, Laval, Quebec H7V 4A7 and HIKMA LABS INC., a legal person, having a place of business at 1809 North Wilson Road, Hilliard, Ohio 43026, U.S.A. -

Use of Chronic Opioid Therapy in Chronic Noncancer Pain CHRONIC NONCANCER PAINNONCANCER CHRONIC Evidence Review
CLINICAL GUIDELINE FOR THE USE OF CHRONIC OPIOID THERAPY IN CLINICAL GUIDELINE FOR THE USE OF CHRONIC OPIOID THERAPY IN GUIDELINE FOR THE Use of Chronic Opioid Therapy in Chronic Noncancer Pain CHRONIC NONCANCER PAIN Evidence Review The American Pain Society in Conjunction with The American Academy of Pain Medicine EVIDENCE REVIEW APS-AAPM Clinical Guidelines for the Use of Chronic Opioid Therapy in Chronic Noncancer Pain TABLE OF CONTENTS Page Introduction 1 Purpose of evidence review ...................................................................... 1 Background 1 Previous guidelines ................................................................................... 2 Scope of evidence review 3 Key questions............................................................................................ 3 Populations................................................................................................ 7 Interventions.............................................................................................. 8 Outcomes.................................................................................................. 8 Conflict of interest............................................................................................. 10 Methods 10 Literature search and strategy................................................................... 10 Inclusion and exclusion criteria.................................................................. 11 Data extraction and synthesis ..................................................................