Dimethoate in Drinking-Water
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Validation Report 20
EURL for Cereals and Feeding stuff National Food Institute Technical University of Denmark Validation Report 20 Determination of pesticide residues in rice baby food by GC-MS/MS and LC-MS/MS (QuEChERS method) Parvaneh Hajeb Susan Strange Herrmann Mette Erecius Poulsen December 2015 Page 2 of 18 CONTENT: 1. Introduction ...................................................................................................................................... 3 2. Principle of analysis......................................................................................................................... 3 3. Validation design ............................................................................................................................. 4 4. Chromatograms and calibration curves .......................................................................................... 5 5. Validation parameters...................................................................................................................... 9 6. Criteria for the acceptance of validation results ........................................................................... 10 7. Results and discussion ................................................................................................................... 10 8. Conclusions .................................................................................................................................... 12 9. References ..................................................................................................................................... -

Guide No. 1 – October 2020 2/12 the CONCEPT and IMPLEMENTATION of CPA GUIDANCE RESIDUE LEVELS
Cooperation Centre for Scientific Research Relative to Tobacco CORESTA GUIDE N° 1 The Concept and Implementation of CPA Guidance Residue Levels October 2020 Agro-Chemical Advisory Committee CORESTA TECHNICAL GUIDE N° 1 Title: The Concept and Implementation of CPA Guidance Residue Levels Status: Valid Note: This document will be periodically reviewed by CORESTA Document history: Date of review Information July 2003 Version 1 GRL for Pyrethrins () and Terbufos corrected. December 2003 CPA terminology corrected. June 2008 Version 2 – GRLs revised and residue definitions added Provisional GRL of 2.00 ppm for Cyfluthrin to replace previous June 2010 GRL of 0.50 ppm July 2013 Version 3 – GRLs revised October 2013 Note for Maleic Hydrazide revised Version 4 – GRLs revised + clarification that scope of GRLs July 2016 applies predominantly to the production of traditional cigarette tobaccos and GAP associated with their cultivation. June 2018 Fluopyram GRL of 5 ppm added to GRL list Version 5 – Nine new CPAs with GRL added to list. November 2019 Revision of GRLs for Chlorantraniliprole and Indoxacarb. Updated web links. October 2020 Version 6 – Flupyradifurone GRL of 21 ppm added to GRL list. CORESTA Guide No. 1 – October 2020 2/12 THE CONCEPT AND IMPLEMENTATION OF CPA GUIDANCE RESIDUE LEVELS Executive Summary • Guidance Residue Levels (GRLs) are in the remit of the Agro-Chemical Advisory Committee (ACAC) of CORESTA. Their development is a joint activity of all ACAC members, who represent the leaf production, processing and manufacturing sectors of the Tobacco Industry. The concept of GRLs and their implementation are described in this guide. • GRLs provide guidance to tobacco growers and assist with interpretation and evaluation of results from analyses of residues of Crop Protection Agents (CPAs*). -

Bayesian Nonparametric Model for Clustering Individual Co-Exposure to Pesticides Found in the French Diet
Bayesian nonparametric model for clustering individual co-exposure to pesticides found in the French diet. Amélie Crépet, Jessica Tressou To cite this version: Amélie Crépet, Jessica Tressou. Bayesian nonparametric model for clustering individual co-exposure to pesticides found in the French diet.. 2009. hal-00438796v2 HAL Id: hal-00438796 https://hal.archives-ouvertes.fr/hal-00438796v2 Preprint submitted on 12 Jan 2011 (v2), last revised 4 Feb 2011 (v3) HAL is a multi-disciplinary open access L’archive ouverte pluridisciplinaire HAL, est archive for the deposit and dissemination of sci- destinée au dépôt et à la diffusion de documents entific research documents, whether they are pub- scientifiques de niveau recherche, publiés ou non, lished or not. The documents may come from émanant des établissements d’enseignement et de teaching and research institutions in France or recherche français ou étrangers, des laboratoires abroad, or from public or private research centers. publics ou privés. Bayesian nonparametric model for clustering individual co-exposure to pesticides found in the French diet. Am´elieCr´epet a & Jessica Tressoub January 12, 2011 aANSES, French Agency for Food, Environmental and Occupational Health Safety, 27-31 Av. G´en´eralLeclerc, 94701 Maisons-Alfort, France bINRA-Met@risk, Food Risk Analysis Methodologies, National Institute for Agronomic Re- search, 16 rue Claude Bernard, 75231 Paris, France Keywords Dirichlet process; Bayesian nonparametric modeling; multivariate Normal mixtures; clustering; multivariate exposure; food risk analysis. Abstract This work introduces a specific application of Bayesian nonparametric statistics to the food risk analysis framework. The goal was to determine the cocktails of pesticide residues to which the French population is simultaneously exposed through its current diet in order to study their possible combined effects on health through toxicological experiments. -

2020) XXX Draft COMMISSION REGULATION (EU
EUROPEAN COMMISSION Brussels, XXX SANTE/10482/2020 […](2020) XXX draft COMMISSION REGULATION (EU) …/… of XXX amending Annexes II, III and V to Regulation (EC) No 396/2005 of the European Parliament and of the Council as regards maximum residue levels for carbon tetrachloride, chlorothalonil, chlorpropham, dimethoate, ethoprophos, fenamidone, methiocarb, omethoate, propiconazole and pymetrozine in or on certain products (Text with EEA relevance) EN EN COMMISSION REGULATION (EU) …/… of XXX amending Annexes II, III and V to Regulation (EC) No 396/2005 of the European Parliament and of the Council as regards maximum residue levels for carbon tetrachloride, chlorothalonil, chlorpropham, dimethoate, ethoprophos, fenamidone, methiocarb, omethoate, propiconazole and pymetrozine in or on certain products (Text with EEA relevance) THE EUROPEAN COMMISSION, Having regard to the Treaty on the Functioning of the European Union, Having regard to Regulation (EC) No 396/2005 of the European Parliament and of the Council of 23 February 2005 on maximum residue levels of pesticides in or on food and feed of plant and animal origin and amending Council Directive 91/414/EEC1, and in particular Article 14(1)(a) and Article 18(1)(b) thereof, Whereas: (1) For chlorothalonil, chlorpropham, dimethoate, fenamidone, omethoate, propiconazole and pymetrozine maximum residue levels (MRLs) were set in Annex II to Regulation (EC) No 396/2005. For carbon tetrachloride, MRLs were set in Annex II and Part B of Annex III to that Regulation. For ethoprophos and methiocarb, MRLs were set in Part A of Annex III to that Regulation. (2) The approval of the active substance chlorothalonil was not renewed by Commission Implementing Regulation (EU) 2019/6772. -

Investigation on the Behavior of Pesticides in Atmosphere
Aerosol and Air Quality Research, 11: 783–790, 2011 Copyright © Taiwan Association for Aerosol Research ISSN: 1680-8584 print / 2071-1409 online doi: 10.4209/aaqr.2010.10.0085 Investigation on the Behavior of Pesticides in Atmosphere Pasquale Avino1, Giuseppe Cinelli2, Ivan Notardonato2, Mario V. Russo2* 1 DIPIA, INAIL (ex-ISPESL), via Urbana 167, 00184 Rome, Italy 2 Faculty of Agriculture, University of Molise, via De Sanctis, Campobasso, Italy ABSTRACT Although pesticides are widely used in agriculture, they and in particular the relative residues in foodstuffs, water and atmosphere, may cause remarkable sanitary problems due to the harmful effects (carcinogenic and mutagenic effects) on the human health. In fact, their spread in waters and atmosphere can produce undesired effects on various organisms and/or water contamination. This paper shows an analytical approach based on XAD-2 adsorbent and GC analysis for evaluating the pesticide trend in atmosphere: in particular, the pesticides investigated are omethoate, dicrotofos, disulfoton, dimethoate, parathion methyl, formothion, paraoxon ethyl, malaoxon, parathion ethyl, iodofenfos and triazofos. For the analytical methodology a linearity response was obtained (r2 = 0.9988) in GC-NPD whereas the limits of detection range between 2 and 5 pg/μL in GC-NPD with a Relative Standard Deviation below 9.5. Finally, this approach has been successfully applied to real samples: the results show that dimethoate concentration decreases with increasing distance from the sampling site but it is still persistent in atmosphere after few days from the pesticide spraying. Keywords: Pesticides; XAD-2 adsorbent; GC analysis; Atmosphere; Air quality. INTRODUCTION 2009). One of the most important OP reactions is water hydrolysis. -

Environmental Health Criteria 63 ORGANOPHOSPHORUS
Environmental Health Criteria 63 ORGANOPHOSPHORUS INSECTICIDES: A GENERAL INTRODUCTION Please note that the layout and pagination of this web version are not identical with the printed version. Organophophorus insecticides: a general introduction (EHC 63, 1986) INTERNATIONAL PROGRAMME ON CHEMICAL SAFETY ENVIRONMENTAL HEALTH CRITERIA 63 ORGANOPHOSPHORUS INSECTICIDES: A GENERAL INTRODUCTION This report contains the collective views of an international group of experts and does not necessarily represent the decisions or the stated policy of the United Nations Environment Programme, the International Labour Organisation, or the World Health Organization. Published under the joint sponsorship of the United Nations Environment Programme, the International Labour Organisation, and the World Health Organization World Health Orgnization Geneva, 1986 The International Programme on Chemical Safety (IPCS) is a joint venture of the United Nations Environment Programme, the International Labour Organisation, and the World Health Organization. The main objective of the IPCS is to carry out and disseminate evaluations of the effects of chemicals on human health and the quality of the environment. Supporting activities include the development of epidemiological, experimental laboratory, and risk-assessment methods that could produce internationally comparable results, and the development of manpower in the field of toxicology. Other activities carried out by the IPCS include the development of know-how for coping with chemical accidents, coordination -

IMDG Code (Amendment 37-14) Index Korean Register of Shipping
IMDG Code (Amendment 37-14) Index Substance, material or article MP Class UN No. ACETAL - 3 1088 ACETALDEHYDE - 3 1089 ACETALDEHYDE AMMONIA - 9 1841 Acetaldehyde diethyl acetal, see - 3 1088 ACETALDEHYDE OXIME - 3 2332 Acetaldol, see - 6.1 2839 beta-Acetaldoxime, see - 3 2332 ACETIC ACID, GLACIAL - 8 2789 ACETIC ACID SOLUTION more than 10% and less than 50% acid, - 8 2790 by mass ACETIC ACID SOLUTION not less than 50% but no more than 80% - 8 2790 acid, by mass ACETIC ACID SOLUTION more than 80% acid, by mass - 8 2789 Acetic aldehyde, see - 3 1089 ACETIC ANHYDRIDE - 8 1715 Acetic oxide, see - 8 1715 Acetoin, see - 3 2621 ACETONE - 3 1090 ACETONE CYANOHYDRIN, STABILIZED P 6.1 1541 Acetone hexafluoride, see - 2.3 2420 ACETONE OILS - 3 1091 Acetone-pyrogallol copolymer 2-diazo-1-naphthol-5-sulphonate - 4.1 3228 ACETONITRILE - 3 1648 3-Acetoxypropene, see - 3 2333 Acetylacetone, see - 3 2310 Acetyl acetone peroxide (concentration ≤32%, as a paste), see - 5.2 3106 Acetyl acetone peroxide (concentration ≤42%, with diluent Type A, and - 5.2 3105 water, available oxygen ≤4.7%), see ACETYL BROMIDE - 8 1716 ACETYL CHLORIDE - 3 1717 Acetyl cyclohexanesulphonyl peroxide - 5.2 3115 (concentration ≤32%, with diluent Type B), see Acetyl cyclohexanesulphonyl peroxide - 5.2 3112 (concentration ≤82%, with water), see Acetylene dichloride, see - 3 1150 ACETYLENE, DISSOLVED - 2.1 1001 Acetylene, ethylene and propylene mixtures, refrigerated liquid, see - 2.1 3138 ACETYLENE, SOLVENT FREE - 2.1 3374 Acetylene tetrabromide, see P 6.1 2504 Acetylene tetrachloride, see P 6.1 1702 ACETYL IODIDE - 8 1898 Acetyl ketene, stabilized, see - 6.1 2521 ACETYL METHYL CARBINOL - 3 2621 Acid butyl phosphate, see - 8 1718 Acid mixture, hydrofluoric and sulphuric, see - 8 1786 Acid mixture, nitrating acid, see - 8 1796 Korean Register of Shipping IMDG Code (Amendment 37-14) Index Substance, material or article MP Class UN No. -

Pesticide Residues : Maximum Residue Limits
THAI AGRICULTURAL STANDARD TAS 9002-2013 PESTICIDE RESIDUES : MAXIMUM RESIDUE LIMITS National Bureau of Agricultural Commodity and Food Standards Ministry of Agriculture and Cooperatives ICS 67.040 ISBN UNOFFICAL TRANSLATION THAI AGRICULTURAL STANDARD TAS 9002-2013 PESTICIDE RESIDUES : MAXIMUM RESIDUE LIMITS National Bureau of Agricultural Commodity and Food Standards Ministry of Agriculture and Cooperatives 50 Phaholyothin Road, Ladyao, Chatuchak, Bangkok 10900 Telephone (662) 561 2277 Fascimile: (662) 561 3357 www.acfs.go.th Published in the Royal Gazette, Announcement and General Publication Volume 131, Special Section 32ง (Ngo), Dated 13 February B.E. 2557 (2014) (2) Technical Committee on the Elaboration of the Thai Agricultural Standard on Maximum Residue Limits for Pesticide 1. Mrs. Manthana Milne Chairperson Department of Agriculture 2. Mrs. Thanida Harintharanon Member Department of Livestock Development 3. Mrs. Kanokporn Atisook Member Department of Medical Sciences, Ministry of Public Health 4. Mrs. Chuensuke Methakulawat Member Office of the Consumer Protection Board, The Prime Minister’s Office 5. Ms. Warunee Sensupa Member Food and Drug Administration, Ministry of Public Health 6. Mr. Thammanoon Kaewkhongkha Member Office of Agricultural Regulation, Department of Agriculture 7. Mr. Pisan Pongsapitch Member National Bureau of Agricultural Commodity and Food Standards 8. Ms. Wipa Thangnipon Member Office of Agricultural Production Science Research and Development, Department of Agriculture 9. Ms. Pojjanee Paniangvait Member Board of Trade of Thailand 10. Mr. Charoen Kaowsuksai Member Food Processing Industry Club, Federation of Thai Industries 11. Ms. Natchaya Chumsawat Member Thai Agro Business Association 12. Mr. Sinchai Swasdichai Member Thai Crop Protection Association 13. Mrs. Nuansri Tayaputch Member Expert on Method of Analysis 14. -

Part II Poisons
Poisons to Which Part II of the Poisons List Applies The following poisons are listed in Part II of the Poisons List: • aldicarb • alpha-chloralose • ammonia • the following arsenic compounds-calcium arsenites, copper acetoarsenite, copper arsenates, copper arsenites, lead arsenates • the following salts of barium-barium carbonate, barium silicofluoride • carbofuran • cycloheximide • dinitrocresols (DNOC), their compounds with a metal or a base • dinoseb, its compounds with a metal or a base • dinoterb • drazoxolon and its salts • endosulfan • endothal and its salts • endrin • compounds of fentin • formaldehyde • formic acid • hydrochloric acid • hydrofluoric acid, alkali metal bifluorides, ammonium bifluoride, alkali metal fluorides, ammonium fluoride, sodium silicofluoride • mercuric chloride, mercuric iodide, organic compounds of mercury except compounds which contain a methyl (CH3) group directly linked to the mercury atom • metallic oxalates • methomyl • nicotine and its salts and quaternary compounds • nitric acid • nitrobenzene • oxamyl • paraquat and its salts • phenols (as defined in part I of the poisons list) in substances containing less than 60% weight in weight, of phenols and compound of phenols with a metal in substances containing less than the equivalent of 60% weight in weight, of phenols • phosphoric acid • the following phosphorus compounds:- azinphos-methyl, chlorfenvinphos, demphion, demeton-S-methyl sulphone, dialifos, dichlorvos, dioxathion, disulfoton, fonofos, mecarbam, mephosfolan, methidathion, mevinphos, omethoate, -

1 of 3 GC+LC-USA
Updated: 07/18/2016 1 of 3 GC+LC-USA Limit of Quantitation (LOQ): 0.010 mg/kg (ppm) Sample Types: Low Fat Content Samples Minimum Sample Size: 100 grams (~1/4 pound). Certain products require more for better sample representation. Instrument: GC-MS/MS and LC-MS/MS Turnaround: 24-48 hours Accreditation: Part of AGQ USA's ISO/IEC 17025 Accreditation Scope 4,4'-Dichlorobenzophenone Bupirimate Cyantraniliprole Diflufenican Abamectin Buprofezin Cyazofamid Dimethoate Acephate Butachlor Cycloate Dimethoate (Sum) Acequinocyl Butocarboxim Cycloxydim Dimethomorph Acetamiprid Butralin Cyflufenamid Diniconazole Acetochlor Cadusafos Cyfluthrin Dinocap Acrinathrin Captafol Cymoxanil Dinotefuran Alachlor Captan Cyproconazole Diphenylamine Aldicarb Captan (Sum) Cyprodinil Disulfoton Aldicarb (Sum) Carbaryl Cyromazine Disulfoton (Sum) Aldicarb-sulfone Carbofuran DDD-o,p Disulfoton-sulfone Aldicarb-sulfoxide Carbofuran-3-hydroxy DDD-p,p +DDT-o,p Disulfoton-sulfoxide Aldrin Carbophenothion DDE-o,p Ditalimfos Ametryn Carbosulfan DDE-p,p Diuron Amitraz Carboxine DDT (Sum) Dodemorph Atrazine Carfentrazone-ethyl DDT-p,p Dodine Azadirachtin Chinomethionat DEET Emamectin Benzoate Azamethiphos Chlorantraniliprole Deltamethrin Endosulfan (A+B+Sulf) Azinphos-ethyl Chlordane Demeton Endosulfan Alfa Azinphos-methyl Chlordane Trans Demeton-S-methyl-sulfone Endosulfan Beta Azoxystrobin Chlorfenapyr Desmedipham Endosulfan Sulfate Benalaxyl Chlorfenson Diafenturion Endrin Ben-Carb-TPM (Sum) Chlorfenvinphos Dialifos EPN Bendiocarb Chlorfluazuron Diazinon Epoxiconazole -

NMP-Free Formulations of Neonicotinoids
(19) & (11) EP 2 266 400 A1 (12) EUROPEAN PATENT APPLICATION (43) Date of publication: (51) Int Cl.: 29.12.2010 Bulletin 2010/52 A01N 43/40 (2006.01) A01N 43/86 (2006.01) A01N 47/40 (2006.01) A01N 51/00 (2006.01) (2006.01) (2006.01) (21) Application number: 09305544.0 A01P 7/00 A01N 25/02 (22) Date of filing: 15.06.2009 (84) Designated Contracting States: (72) Inventors: AT BE BG CH CY CZ DE DK EE ES FI FR GB GR • Gasse, Jean-Jacques HR HU IE IS IT LI LT LU LV MC MK MT NL NO PL 27600 Saint-Aubin-Sur-Gaillon (FR) PT RO SE SI SK TR • Duchamp, Guillaume Designated Extension States: 92230 Gennevilliers (FR) AL BA RS • Cantero, Maria 92230 Gennevilliers (FR) (71) Applicant: NUFARM 92233 Gennevelliers (FR) (74) Representative: Cabinet Plasseraud 52, rue de la Victoire 75440 Paris Cedex 09 (FR) (54) NMP-free formulations of neonicotinoids (57) The invention relates to NMP-free liquid formulation comprising at least one nicotinoid and at least one aprotic polar component selected from the group comprising the compounds of formula I, II or III below, and mixtures thereof, wherein R1 and R2 independently represent H or an alkyl group having less than 5 carbons, preferably a methyl group, and n represents an integer ranging from 0 to 5, and to their applications. EP 2 266 400 A1 Printed by Jouve, 75001 PARIS (FR) EP 2 266 400 A1 Description Technical Field of the invention 5 [0001] The invention relates to novel liquid formulations of neonicotinoids and to their use for treating plants, for protecting plants from pests and/or for controlling pests infestation. -
Method Description
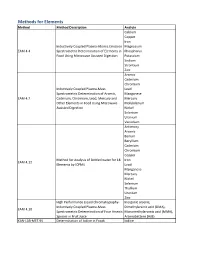
Methods for Elements Method Method Description Analyte Calcium Copper Iron Inductively Coupled Plasma-Atomic Emission Magnesium EAM 4.4 Spectrometric Determination of Elements in Phosphorus Food Using Microwave Assisted Digestion Potassium Sodium Strontium Zinc Arsenic Cadmium Chromium Inductively Coupled Plasma-Mass Lead Spectrometric Determination of Arsenic, Manganese EAM 4.7 Cadmium, Chromium, Lead, Mercury and Mercury Other Elements in Food Using Microwave Molybdenum Assisted Digestion Nickel Selenium Uranium Vanadium Antimony Arsenic Barium Beryllium Cadmium Chromium Copper Method for Analysis of Bottled water for 18 Iron EAM 4.12 Elements by ICPMS Lead Manganese Mercury Nickel Selenium Thallium Uranium Zinc High Performance Liquid Chromatography- Inorganic arsenic, Inductively Coupled Plasma-Mass Dimethylarsinic acid (DMA), EAM 4.10 Spectrometric Determination of Four Arsenic Monomethylarsonic acid (MMA), Species in Fruit Juice Arsenobetaine (AsB) KAN-LAB-MET.95 Determination of Iodine in Foods Iodine Methods for Radionuclides Method Method Description Analyte Determination of Strontium-90 in Foods by WEAC.RN.METHOD.2.0 Strontium-90 Internal Gas-Flow Proportional Counting Americium-241 Cesium-134 Cesium-137 Determination of Gamma-Ray Emitting Cobalt-60 WEAC.RN.METHOD.3.0 Radionuclides in Foods by High-Purity Potassium-40 Germanium Spectrometry Radium-226 Ruthenium-103 Ruthenium-106 Thorium-232 Methods for Pesticides/Industrial Chemicals Method Method Description Analyte Extraction Method: Analysis of Pesticides KAN-LAB-PES.53 and