Young Onset Dementia E L Sampson, J D Warren, M N Rossor
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-
AMYOTROPHIC LATERAL SCLEROSIS Spin21 (1)
AMYOTROPHIC LATERAL SCLEROSIS Spin21 (1) Amyotrophic Lateral Sclerosis (ALS) Synonyms: CHARCOT'S DISEASE (in Europe), LOU GEHRIG'S DISEASE (in USA) Last updated: April 22, 2019 ETIOPATHOPHYSIOLOGY, PATHOLOGY .................................................................................................. 1 GENETICS ............................................................................................................................................... 3 EPIDEMIOLOGY ........................................................................................................................................ 3 Genetics ............................................................................................................................................ 3 CLINICAL FEATURES ............................................................................................................................... 3 EL ESCORIAL WORLD FEDERATION OF NEUROLOGY CRITERIA .............................................................. 4 CLINICAL COURSE & PROGNOSIS ........................................................................................................... 4 VARIANTS .............................................................................................................................................. 5 DIAGNOSIS................................................................................................................................................ 5 TREATMENT ............................................................................................................................................ -

Myalgic Encephalomyelitis/Chronic Fatigue
2019 Science & Discovery Webinar Series ME/CFS in the Era of the Human Microbiome: Persistent Pathogens Drive Chronic Symptoms by Interfering With Host Metabolism, Gene Expression, and Immunity with Amy Proal, Ph.D. November 14, 2019 | 1:00 PM Eastern www.SolveME.org About Our Webinars • Welcome to the 2019 Webinar Series! • The audience is muted; use the question box to send us questions. Dr. Proal will address as many questions as time permits at the end of the webinar • Webinars are recorded and the recording is made available on our YouTube channel http://youtube.com/SolveCFS • The Solve ME/CFS Initiative does not provide medical advice www.SolveCFS.org 2019 Science & Discovery Webinar Series ME/CFS in the Era of the Human Microbiome: Persistent Pathogens Drive Chronic Symptoms by Interfering With Host Metabolism, Gene Expression, and Immunity with Amy Proal, Ph.D. November 14, 2019 | 1:00 PM Eastern www.SolveME.org Myalgic Encephalomyelitis/Chronic Fatigue Syndrome in the Era of the Human Microbiome: Persistent Pathogens Drive Chronic Symptoms by Interfering With Host Metabolism, Gene Expression, and Immunity Amy Proal, Autoimmunity Research Foundation/PolyBio Millions of patients across the globe are suffering with myalgic encephalomyelitis (ME/CFS) Currently there is no one disease-specific biomarker and severely ill patients are often wheelchair dependent, bedridden and unable to perform basic tasks of work or daily living. #millionsmissing Myalgic Encephalomeylitis (ME) = swelling of the brain • Unrelenting fatigue that does -

Neuromuscular Disorders Neurology in Practice: Series Editors: Robert A
Neuromuscular Disorders neurology in practice: series editors: robert a. gross, department of neurology, university of rochester medical center, rochester, ny, usa jonathan w. mink, department of neurology, university of rochester medical center,rochester, ny, usa Neuromuscular Disorders edited by Rabi N. Tawil, MD Professor of Neurology University of Rochester Medical Center Rochester, NY, USA Shannon Venance, MD, PhD, FRCPCP Associate Professor of Neurology The University of Western Ontario London, Ontario, Canada A John Wiley & Sons, Ltd., Publication This edition fi rst published 2011, ® 2011 by Blackwell Publishing Ltd Blackwell Publishing was acquired by John Wiley & Sons in February 2007. Blackwell’s publishing program has been merged with Wiley’s global Scientifi c, Technical and Medical business to form Wiley-Blackwell. Registered offi ce: John Wiley & Sons Ltd, The Atrium, Southern Gate, Chichester, West Sussex, PO19 8SQ, UK Editorial offi ces: 9600 Garsington Road, Oxford, OX4 2DQ, UK The Atrium, Southern Gate, Chichester, West Sussex, PO19 8SQ, UK 111 River Street, Hoboken, NJ 07030-5774, USA For details of our global editorial offi ces, for customer services and for information about how to apply for permission to reuse the copyright material in this book please see our website at www.wiley.com/wiley-blackwell The right of the author to be identifi ed as the author of this work has been asserted in accordance with the UK Copyright, Designs and Patents Act 1988. All rights reserved. No part of this publication may be reproduced, stored in a retrieval system, or transmitted, in any form or by any means, electronic, mechanical, photocopying, recording or otherwise, except as permitted by the UK Copyright, Designs and Patents Act 1988, without the prior permission of the publisher. -

Palliative Care in Amyotrophic Lateral Sclerosis: from Diagnosis to Bereavement, 2Nd Edn, Pp
Palliative Care in Amyotrophic Lateral Sclerosis From Diagnosis to Bereavement THIRD EDITION Edited by David Oliver Consultant in Palliative Medicine Wisdom Hospice, Rochester and Honorary Reader University of Kent, UK Gian Domenico Borasio Chair in Palliative Medicine Director, Palliative Care Service Centre Hospitalier Universitaire Vaudois University of Lausanne, Switzerland Wendy Johnston Professor of Neurology Director, ALS Programme University of Alberta, Canada 1 1 Great Clarendon Street, Oxford, OX2 6DP, United Kingdom Oxford University Press is a department of the University of Oxford. It furthers the University’s objective of excellence in research, scholarship, and education by publishing worldwide. Oxford is a registered trade mark of Oxford University Press in the UK and in certain other countries © Oxford University Press 2014 The moral rights of the authors have been asserted Second Edition published in 2006 9780199212934 HB 9780198570486 PB First Edition published in 2000 9780192631664 Impression: 1 All rights reserved. No part of this publication may be reproduced, stored in a retrieval system, or transmitted, in any form or by any means, without the prior permission in writing of Oxford University Press, or as expressly permitted by law, by licence or under terms agreed with the appropriate reprographics rights organization. Enquiries concerning reproduction outside the scope of the above should be sent to the Rights Department, Oxford University Press, at the address above You must not circulate this work in any other form and you must impose this same condition on any acquirer Published in the United States of America by Oxford University Press 198 Madison Avenue, New York, NY 10016, United States of America British Library Cataloguing in Publication Data Data available Library of Congress Control Number: 2013950545 ISBN 978–0–19–968602–5 Printed and bound by CPI Group (UK) Ltd, Croydon, CR0 4YY Oxford University Press makes no representation, express or implied, that the drug dosages in this book are correct. -

African Meningitis Belt
WHO/EMC/BAC/98.3 Control of epidemic meningococcal disease. WHO practical guidelines. 2nd edition World Health Organization Emerging and other Communicable Diseases, Surveillance and Control This document has been downloaded from the WHO/EMC Web site. The original cover pages and lists of participants are not included. See http://www.who.int/emc for more information. © World Health Organization This document is not a formal publication of the World Health Organization (WHO), and all rights are reserved by the Organization. The document may, however, be freely reviewed, abstracted, reproduced and translated, in part or in whole, but not for sale nor for use in conjunction with commercial purposes. The views expressed in documents by named authors are solely the responsibility of those authors. The mention of specific companies or specific manufacturers' products does no imply that they are endorsed or recommended by the World Health Organization in preference to others of a similar nature that are not mentioned. CONTENTS CONTENTS ................................................................................... i PREFACE ..................................................................................... vii INTRODUCTION ......................................................................... 1 1. MAGNITUDE OF THE PROBLEM ........................................................3 1.1 REVIEW OF EPIDEMICS SINCE THE 1970S .......................................................................................... 3 Geographical distribution -

THE CLINICAL ASSESSMENT of the PATIENT with EARLY DEMENTIA S Cooper, J D W Greene V15
J Neurol Neurosurg Psychiatry: first published as 10.1136/jnnp.2005.081133 on 16 November 2005. Downloaded from THE CLINICAL ASSESSMENT OF THE PATIENT WITH EARLY DEMENTIA S Cooper, J D W Greene v15 J Neurol Neurosurg Psychiatry 2005;76(Suppl V):v15–v24. doi: 10.1136/jnnp.2005.081133 ementia is a clinical state characterised by a loss of function in at least two cognitive domains. When making a diagnosis of dementia, features to look for include memory Dimpairment and at least one of the following: aphasia, apraxia, agnosia and/or disturbances in executive functioning. To be significant the impairments should be severe enough to cause problems with social and occupational functioning and the decline must have occurred from a previously higher level. It is important to exclude delirium when considering such a diagnosis. When approaching the patient with a possible dementia, taking a careful history is paramount. Clues to the nature and aetiology of the disorder are often found following careful consultation with the patient and carer. A focused cognitive and physical examination is useful and the presence of specific features may aid in diagnosis. Certain investigations are mandatory and additional tests are recommended if the history and examination indicate particular aetiologies. It is useful when assessing a patient with cognitive impairment in the clinic to consider the following straightforward questions: c Is the patient demented? c If so, does the loss of function conform to a characteristic pattern? c Does the pattern of dementia conform to a particular pattern? c What is the likely disease process responsible for the dementia? An understanding of cognitive function and its anatomical correlates is necessary in order to ascertain which brain areas are affected. -

Motor Neurone Disease
Neurology Motor neurone disease Margaret Zoing Matthew Kiernan Caring for the patient in general practice Motor neurone disease (MND) is a progressive Background neurodegenerative disease. It is characterised by motor Motor neurone disease is a neurodegenerative disease that systems failure that results in the death of nerves responsible leads to progressive disability – and eventually death – for all voluntary movements, leading to limb paralysis, within 2–3 years. weakness of the muscles of speech and swallowing, and Objective ultimately respiratory failure. Typically MND strikes patients This article describes the role of the general practitioner in at the prime of adult life, usually in the fifth to sixth decades, caring for patients with motor neurone disease. and has a short trajectory from diagnosis with an average life Discussion expectancy of less than 3 years.1 Current estimates are that The diagnosis of motor neurone disease relies on the 1400 people are living with MND in Australia at any time, presence of upper and lower motor neurone features. There with 370 newly diagnosed patients each year.2 More than one is currently no pathognomic test for motor neurone disease Australian dies every day from this most pernicious disease. and it largely remains a diagnosis of exclusion following an accurate clinical history, combined with basic screening The cause of MND remains unknown but appears heterogeneous. blood investigations and structural imaging of the brain Environmental factors may trigger an underlying susceptibility – toxins, and spinal cord. Neuro-physiological studies may be useful chemicals, metals and trauma have all been proposed.1 Most cases as an ancillary diagnostic tool. -
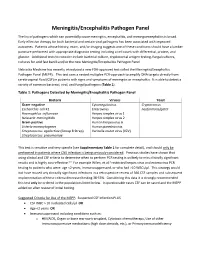
Meningitis/Encephalitis Pathogen Panel
Meningitis/Encephalitis Pathogen Panel The list of pathogens which can potentially cause meningitis, encephalitis, and meningoencephalitis is broad. Early effective therapy for both bacterial and certain viral pathogens has been associated with improved outcomes. Patients whose history, exam, and/or imaging suggests one of these conditions should have a lumber puncture performed with appropriate diagnostic testing including a cell count with differential, protein, and glucose. Additional tests to consider include bacterial culture, cryptococcal antigen testing, fungal cultures, cultures for acid fast bacilli and/or the new Meningitis/Encephalitis Pathogen Panel. Nebraska Medicine has recently introduced a new FDA-approved test called the Meningitis/Encephalitis Pathogen Panel (MEPP). This test uses a nested multiplex PCR-approach to amplify DNA targets directly from cerebrospinal fluid (CSF) in patients with signs and symptoms of meningitis or encephalitis. It is able to detect a variety of common bacterial, viral, and fungal pathogens (Table 1). Table 1: Pathogens Detected by Meningitis/Encephalitis Pathogen Panel Bacteria Viruses Yeast Gram-negative Cytomegalovirus Cryptococcus Escherichia coli K1 Enterovirus neoformans/gattii Haemophilus influenzae Herpes simplex virus 1 Neisseria meningitidis Herpes simplex virus 2 Gram-positive Human herpesvirus 6 Listeria monocytogenes Human parechovirus Streptococcus agalactiae (Group B Strep) Varicella zoster virus (VZV) Streptococcus pneumoniae This test is sensitive and very specific (see Supplementary Table 1 for complete detail), and should only be performed in patients where CNS infection is being seriously considered. Previous studies have shown that using clinical and CSF criteria to determine when to perform PCR testing is unlikely to miss clinically significant results and is highly cost-effective.1-3 For example Wilen, et al.3 restricted herpes virus and enterovirus PCR testing to patients who were: age <2 years, immunosuppressed, or who had >10 WBCs/µl. -

Case Amyotrophic Lateral Sclerosis
Case Amyotrophic Lateral Sclerosis: First Case Report in Department of Neurosurgery, Faculty of Medicine, Universitas Padjadjaran, Bandung Ahmad Faried, Priandana Adya Eka Saputra, Alief Dhuha, Muhammad Zafrullah Arifin Department of Neurosurgery, Faculty of Medicine, Universitas Padjadjaran-Dr. Hasan Sadikin Hospital, Bandung Abstract Objective: Amyotrophic lateral sclerosis (ALS) is a neurodegenerative disease that is incurable and results in paralysis of the muscles. Electromyography (EMG) is used to diagnosis amyotrophic lateral sclerosis. Although recently several years and is helping those who are newly diagnosed. there is no cure for ALS, knowledge has increased significantly in the past Methods: Neurosurgery, Faculty of Medicine, Universitas Padjadjaran-Dr. Hasan Sadikin GeneralThis Hospital,study reported Bandung afor 58-year the firstold mantime who in wasDepartment presented inof the institution with a history of weakness of both lower extremities for four months preceded by weakness of both upper extremities since the previous month. There was no history of any medical illness or any chronic medication. The patient then underwent EMG studies, followed by muscle biopsy. Results: diagnosis of ALS. Electromyography and histopathological results confirmed a Received: Conclusions: This case was so exceptional, since ALS occurrence in April 24, 2018 Neurosurgery Centre is extremely rare, and the diagnosis can only be established through EMG and histopathological of muscle biopsy studies. Revised: December 20, 2018 Keywords: electromyography -

Motor Neuron Disease Motor Neuron Disease
Motor Neuron Disease Motor Neuron Disease • Incidence: 2-4 per 100 000 • Onset: usually 50-70 years • Pathology: – Degenerative condition – anterior horn cells and upper motor neurons in spinal cord, resulting in mixed upper and lower motor neuron signs • Cause unknown – 10% familial (SOD-1 mutation) – ? Related to athleticism Presentation • Several variations in onset, but progress to the same endpoint • Motor nerves only affected • May be just UMN or just LMN at onset, but other features will appear over time • Main patterns: – Amyotrophic lateral sclerosis – Bulbar presentaion – Primary lateral sclerosis (UMN onset) – Progressive muscular atrophy (LMN onset) Questions Wasting Classification • Amyotrophic Lateral Sclerosis • Progressive Bulbar Palsy • Progressive Muscular Atrophy • Primary Lateral Sclerosis • Multifocal Motor Neuropathy • Spinal Muscular Atrophy • Kennedy’s Disease • Monomelic Amyotrophy • Brachial Amyotrophic Diplegia El Escorial Criteria for Diagnosis Tongue fasiculations Amyotrophic lateral sclerosis • ‘Typical’ presentation (60%+) • Usually one limb initially – Foot drop – Clumsy weak hand – May complain of cramps • Gradual progression over months • May be some wasting at presentation • Usually fasiculations (often more widespread) • Brisk reflexes, extensor plantars • No sensory signs; MAY occasionally be mild symptoms • Relentless progression, noticable over weeks/ months Bulbar MND • Approximately 30% of cases • Onset with dysarthria, dysphagia • Bulbar and pseudobulbar symptoms • On examination – Dysarthria – -

Part Ii – Neurological Disorders
Part ii – Neurological Disorders CHAPTER 14 MOVEMENT DISORDERS AND MOTOR NEURONE DISEASE Dr William P. Howlett 2012 Kilimanjaro Christian Medical Centre, Moshi, Kilimanjaro, Tanzania BRIC 2012 University of Bergen PO Box 7800 NO-5020 Bergen Norway NEUROLOGY IN AFRICA William Howlett Illustrations: Ellinor Moldeklev Hoff, Department of Photos and Drawings, UiB Cover: Tor Vegard Tobiassen Layout: Christian Bakke, Division of Communication, University of Bergen E JØM RKE IL T M 2 Printed by Bodoni, Bergen, Norway 4 9 1 9 6 Trykksak Copyright © 2012 William Howlett NEUROLOGY IN AFRICA is freely available to download at Bergen Open Research Archive (https://bora.uib.no) www.uib.no/cih/en/resources/neurology-in-africa ISBN 978-82-7453-085-0 Notice/Disclaimer This publication is intended to give accurate information with regard to the subject matter covered. However medical knowledge is constantly changing and information may alter. It is the responsibility of the practitioner to determine the best treatment for the patient and readers are therefore obliged to check and verify information contained within the book. This recommendation is most important with regard to drugs used, their dose, route and duration of administration, indications and contraindications and side effects. The author and the publisher waive any and all liability for damages, injury or death to persons or property incurred, directly or indirectly by this publication. CONTENTS MOVEMENT DISORDERS AND MOTOR NEURONE DISEASE 329 PARKINSON’S DISEASE (PD) � � � � � � � � � � � -

Bacterial Meningitis and Neurological Complications in Adults
OCUSED EVIEW Parunyou J. et al. Bacterial meningitis F R Bacterial meningitis and neurological complications in adults Parunyou Julayanont MD, Doungporn Ruthirago MD, John C. DeToledo MD ABSTRACT Bacterial meningitis is a leading cause of death from infectious disease worldwide. The neurological complications secondary to bacterial meningitis contribute to the high mortality rate and to disability among the survivors. Cerebrovascular complications, including infarc- tion and hemorrhage, are common. Inflammation and increased pressure in the subarach- noid space result in cranial neuropathy. Seizures occur in either the acute or delayed phase after the infection and require early detection and treatment. Spreading of infection to other intracranial structures, including the subdural space, brain parenchyma, and ventricles, in- creases morbidity and mortality in survivors. Infection can also spread to the spinal canal causing spinal cord abscess, epidural abscess, polyradiculitis, and spinal cord infarction secondary to vasculitis of the spinal artery. Hypothalamic-pituitary dysfunction is also an un- common complication after bacterial meningitis. Damage to cerebral structures contributes to cognitive and neuropsychiatric problems. Being aware of these complications leads to early detection and treatment and improves mortality and outcomes in patients with bacte- rial meningitis. Key words: meningitis; meningitis, bacterial; central nervous system bacterial infection; nervous system diseases INTRODUCTION prove recovery and outcomes. Bacterial meningitis is a leading cause of death In this article, we present a case of bacterial men- from infectious disease worldwide. Despite the avail- ingitis complicated by an unusual number of neuro- ability of increasingly effective antibiotics and inten- logical complications that occurred in spite of a timely sive neurological care, the overall mortality remains diagnosis, adequate treatment, and intensive neuro- high, with 17-34% of the survivors having unfavorable logical monitoring.